Advanced Benzofuranone Manufacturing: Scalable Carbonylation Technology for Global Supply Chains
The global demand for high-performance fine chemical intermediates continues to escalate, driven by the expanding pharmaceutical and agrochemical sectors. In this context, Patent CN111153878A introduces a transformative methodology for synthesizing benzofuranone, a critical building block for novel antioxidants and fungicides like azoxystrobin. This technical disclosure outlines a robust five-step sequence that begins with the readily available o-nitrotoluene, leveraging transition metal-catalyzed carbonylation to establish the carbon framework efficiently. Unlike traditional pathways that suffer from prohibitive costs and environmental hazards, this innovation prioritizes mild reaction conditions and high atom economy. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a competitive edge in the supply of high-purity agrochemical intermediate materials. The process demonstrates exceptional potential for commercial scale-up, offering a viable solution to the longstanding bottlenecks in benzofuranone production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of benzofuranone has been plagued by significant economic and technical inefficiencies that hinder large-scale adoption. The most prevalent conventional route relies on o-chlorophenylacetic acid as the starting material, a precursor that is not only expensive but also difficult to source in bulk quantities with consistent quality. As illustrated in the reaction pathway below, this method necessitates harsh hydrolysis and cyclization conditions that often lead to substantial material degradation and low overall yields.  Furthermore, the reliance on concentrated sulfuric acid in alternative traditional methods introduces severe corrosion risks to manufacturing equipment, escalating maintenance costs and safety liabilities. The operational complexity of these legacy processes, characterized by multi-step purifications and difficult-to-control exothermic reactions, creates unpredictable supply chain vulnerabilities. Consequently, manufacturers facing these constraints often struggle to meet the stringent purity specifications required by downstream pharmaceutical clients, leading to batch rejections and delayed time-to-market for critical drug substances.
Furthermore, the reliance on concentrated sulfuric acid in alternative traditional methods introduces severe corrosion risks to manufacturing equipment, escalating maintenance costs and safety liabilities. The operational complexity of these legacy processes, characterized by multi-step purifications and difficult-to-control exothermic reactions, creates unpredictable supply chain vulnerabilities. Consequently, manufacturers facing these constraints often struggle to meet the stringent purity specifications required by downstream pharmaceutical clients, leading to batch rejections and delayed time-to-market for critical drug substances.
The Novel Approach
In stark contrast to these legacy challenges, the novel approach detailed in the patent utilizes o-nitrotoluene, a commodity chemical with superior availability and cost-effectiveness, to drive the synthesis forward. This methodology employs a direct carbonylation reaction under the influence of a transition metal complex, enabling the formation of the key ester intermediate in a single, streamlined step. By operating at moderate temperatures ranging from 80°C to 150°C and controlled carbon monoxide pressures, the process mitigates the safety risks associated with high-energy reactions while maintaining high conversion rates. The strategic integration of alkaline hydrolysis followed by precise acidification ensures that the intermediate o-nitrophenylacetic acid is isolated with exceptional purity, setting a strong foundation for subsequent transformations. This shift in synthetic strategy not only simplifies the operational workflow but also aligns with modern green chemistry principles by reducing waste generation and eliminating the need for corrosive mineral acids in the initial stages.
Mechanistic Insights into Transition Metal-Catalyzed Carbonylation
The core of this technological breakthrough lies in the sophisticated catalytic cycle that facilitates the carbonylation of the methyl group on the o-nitrotoluene substrate. The reaction mechanism involves the coordination of carbon monoxide and the alcohol solvent to the transition metal center, typically palladium, ruthenium, or copper, activated by specialized phosphine ligands such as Xantphos or DPPF. This catalytic system promotes the insertion of the carbonyl group into the carbon-hydrogen bond of the methyl group, a transformation that is traditionally difficult to achieve with high selectivity. 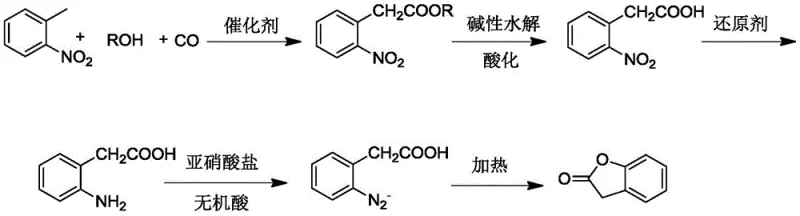 The presence of an oxidant, such as tert-butyl peroxy-ether or hydrogen peroxide, plays a critical role in regenerating the active catalytic species, ensuring sustained turnover numbers throughout the reaction duration. For R&D teams, optimizing the ligand-to-metal ratio and the partial pressure of carbon monoxide is paramount to maximizing the yield of the o-nitroacetophenone acetate intermediate. The mechanistic elegance of this system allows for the recycling of redundant raw materials and the catalyst itself, significantly enhancing the overall process mass intensity and reducing the environmental footprint of the manufacturing operation.
The presence of an oxidant, such as tert-butyl peroxy-ether or hydrogen peroxide, plays a critical role in regenerating the active catalytic species, ensuring sustained turnover numbers throughout the reaction duration. For R&D teams, optimizing the ligand-to-metal ratio and the partial pressure of carbon monoxide is paramount to maximizing the yield of the o-nitroacetophenone acetate intermediate. The mechanistic elegance of this system allows for the recycling of redundant raw materials and the catalyst itself, significantly enhancing the overall process mass intensity and reducing the environmental footprint of the manufacturing operation.
Beyond the primary catalytic cycle, the control of impurity profiles during the reduction and cyclization phases is vital for meeting the rigorous standards of the pharmaceutical industry. The reduction of the nitro group to an amino group is carefully managed using reducing agents like iron powder or hydrazine hydrate, which are selected for their ability to minimize side reactions that could generate difficult-to-remove byproducts. Subsequent diazotization at low temperatures, typically between -5°C and 5°C, ensures the stable formation of the diazonium salt without premature decomposition. The final hydrolytic cyclization step, conducted under acidic conditions at elevated temperatures, drives the ring closure to form the benzofuranone lactone structure with high fidelity. This meticulous control over each reaction parameter ensures that the final product exhibits a clean impurity spectrum, reducing the burden on downstream purification processes and ensuring consistent batch-to-batch quality for sensitive applications.
How to Synthesize Benzofuranone Efficiently
Implementing this synthesis route requires a disciplined approach to process engineering and reaction control to fully realize its commercial potential. The procedure begins with the charging of o-nitrotoluene, alcohol, and the oxidant into a pressure reactor equipped with efficient stirring and temperature control systems to manage the exothermic nature of the carbonylation. Following the formation of the ester intermediate, the process transitions to hydrolysis and acidification, where precise pH control is essential to precipitate the acid intermediate without co-precipitating impurities. The detailed standardized synthesis steps, including specific molar ratios, reaction times, and workup procedures, are outlined in the structured guide below to ensure reproducibility and safety during technology transfer.
- Perform methyl carbonylation on o-nitrotoluene with CO and alcohol under transition metal catalysis to form o-nitroacetophenone acetate.
- Execute alkaline hydrolysis and acidification to convert the ester intermediate into o-nitrophenylacetic acid.
- Reduce the nitro group to an amino group, followed by diazotization and acidic hydrolytic cyclization to finalize the benzofuranone structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers profound strategic advantages that extend beyond mere technical feasibility. The shift to o-nitrotoluene as a feedstock fundamentally alters the cost structure of benzofuranone production, leveraging a raw material that is produced on a massive global scale for other industries, thereby ensuring price stability and supply continuity. The elimination of expensive and hard-to-source precursors like o-chlorophenylacetic acid removes a significant bottleneck from the supply chain, reducing the risk of production stoppages due to raw material shortages. Furthermore, the ability to recycle catalysts and solvents within this process framework contributes to a drastic simplification of the waste management workflow, lowering the operational expenditures associated with environmental compliance and disposal. These factors combine to create a resilient manufacturing model that can withstand market volatility and deliver consistent value to downstream partners.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of high-cost specialty chemicals with commodity-grade o-nitrotoluene, which significantly lowers the direct material cost per kilogram of the final product. Additionally, the recyclability of the transition metal catalyst system means that the consumption of precious metals is minimized, leading to substantial savings on catalyst procurement over the lifecycle of the plant. The mild reaction conditions also reduce energy consumption for heating and cooling, further optimizing the utility costs associated with large-scale production. By streamlining the number of unit operations and avoiding complex purification steps required by older methods, the overall processing time is reduced, enhancing asset utilization and throughput capacity without the need for capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: Sourcing o-nitrotoluene is inherently more stable than relying on niche intermediates, as it is a standard chemical available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions allows for flexible production scheduling, as the process is less sensitive to minor fluctuations in ambient conditions or raw material quality compared to harsher traditional methods. This reliability translates into shorter lead times for order fulfillment, enabling suppliers to respond more agilely to sudden spikes in demand from the agrochemical or pharmaceutical sectors. Moreover, the simplified logistics of handling safer, less corrosive reagents reduce the regulatory burden on transportation and storage, ensuring smoother inbound and outbound logistics operations.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard pressure reactors and separation equipment that are readily available in existing fine chemical manufacturing facilities, facilitating a seamless transition from pilot to commercial scale. The reduction in hazardous waste generation, particularly the avoidance of large volumes of acidic waste from concentrated sulfuric acid processes, aligns with increasingly stringent global environmental regulations. This compliance advantage reduces the risk of regulatory fines and operational shutdowns, ensuring long-term business continuity. The efficient atom economy of the carbonylation step ensures that raw materials are converted into product rather than waste, supporting corporate sustainability goals and enhancing the brand reputation of manufacturers adopting this green chemistry approach.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzofuranone synthesis technology, based on the specific data and claims within the patent documentation. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this route for potential licensing or procurement partnerships. Understanding these details is crucial for making informed decisions about integrating this technology into existing production portfolios to enhance competitiveness and efficiency.
Q: What are the primary advantages of the o-nitrotoluene route over traditional o-chlorophenylacetic acid methods?
A: The o-nitrotoluene route utilizes significantly cheaper and more accessible raw materials compared to o-chlorophenylacetic acid. Furthermore, it avoids harsh synthesis conditions and complex steps, resulting in higher overall yields and reduced environmental pollution pressure.
Q: How does the transition metal catalyst system impact the scalability of this process?
A: The use of recyclable transition metal complexes, such as palladium or copper catalysts with phosphine ligands, allows for efficient catalytic turnover. This recyclability minimizes catalyst consumption costs and simplifies post-reaction processing, which is critical for large-scale industrial production.
Q: Is this synthesis method suitable for producing intermediates for azoxystrobin?
A: Yes, benzofuranone is a key intermediate for synthesizing azoxystrobin, a broad-spectrum agricultural fungicide. This patented method provides a high-purity supply of benzofuranone, directly supporting the manufacturing needs of the agrochemical sector.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuranone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in maintaining a competitive supply chain for high-value intermediates like benzofuranone. Our CDMO expertise allows us to adapt complex pathways, such as the transition metal-catalyzed carbonylation described in Patent CN111153878A, to meet the rigorous demands of global markets. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of benzofuranone meets the exacting standards required for pharmaceutical and agrochemical applications, providing you with a partner who understands both the chemistry and the commerce of fine chemicals.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to validate the performance of our benzofuranone against your internal benchmarks. By partnering with us, you gain access to a reliable agrochemical intermediate supplier committed to innovation, quality, and long-term supply security.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
