Advanced Manufacturing of Dabigatran Etexilate Intermediates via Optimized Esterification and Coupling
Introduction to Next-Generation Anticoagulant Synthesis
The pharmaceutical landscape for direct thrombin inhibitors has evolved significantly, driven by the demand for safer, more scalable manufacturing processes for critical medications like dabigatran etexilate. Patent CN103275065A introduces a transformative synthetic methodology that addresses the longstanding bottlenecks associated with producing this vital anticoagulant prodrug. Unlike traditional approaches that rely on hazardous high-pressure hydrogenation and generate substantial oily impurities, this novel route leverages a strategic sequence of esterification, selective reduction, and coupling reactions. The process begins with readily available raw materials, specifically methylamino-m-nitrobenzoate, and proceeds through seven distinct chemical transformations to yield the final active pharmaceutical ingredient precursor. By optimizing reaction conditions to be milder and more selective, this technology ensures a final product purity exceeding 99%, thereby meeting the rigorous quality standards required for global regulatory compliance. For R&D directors and supply chain managers, this represents a pivotal shift towards more robust and economically viable production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dabigatran etexilate, as reported in earlier patents such as WO9837075, has been plagued by significant technical and economic inefficiencies. The conventional route typically employs palladium-carbon hydrogenation for the reduction of nitro groups, a process that demands specialized high-pressure reaction assemblies and stringent safety protocols. A critical failure point in these legacy methods is the formation of undesirable byproducts; the reduction step often yields an oily mixture containing between 20% to 40% impurities. This high impurity load necessitates complex and costly purification steps, frequently involving column chromatography, which is notoriously difficult to scale for industrial manufacturing. Furthermore, subsequent amidinization reactions in traditional routes exhibit poor selectivity, generating strongly acidic wastewater that complicates environmental compliance. The reliance on chloroformic acid esters containing isomeric impurities further degrades the quality of the final product, often requiring repeated recrystallization to achieve pharmaceutical grade standards, thus driving up both production time and operational costs.
The Novel Approach
In stark contrast, the methodology outlined in CN103275065A offers a streamlined and highly efficient alternative that circumvents these historical challenges. The new process initiates with a controlled esterification of methylamino-m-nitrobenzoate (D1) to form the esterification product (D2), utilizing thionyl chloride and triethylamine under moderate thermal conditions. 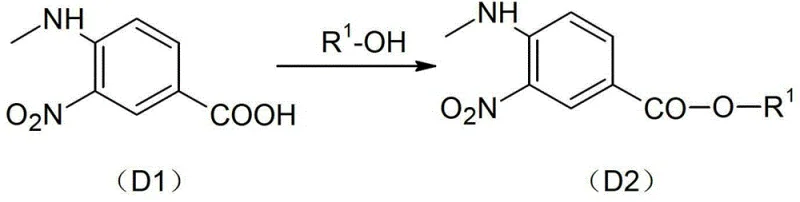 This initial step sets the stage for a series of high-selectivity transformations, including the reduction of the nitro group to an amino group to yield intermediate D3. The subsequent cyclization with compound D10 to form the benzimidazole core (D4) is executed with exceptional precision, avoiding the formation of the oily impurities characteristic of older methods. By maintaining mild reaction temperatures, typically ranging from 50°C to 100°C depending on the specific step, the process eliminates the need for extreme high-pressure equipment in the early stages. The result is a synthesis pathway that is not only simpler to operate but also delivers a total yield reaching 52% with a purity profile superior to 99%, making it ideally suited for large-scale industrial application.
This initial step sets the stage for a series of high-selectivity transformations, including the reduction of the nitro group to an amino group to yield intermediate D3. The subsequent cyclization with compound D10 to form the benzimidazole core (D4) is executed with exceptional precision, avoiding the formation of the oily impurities characteristic of older methods. By maintaining mild reaction temperatures, typically ranging from 50°C to 100°C depending on the specific step, the process eliminates the need for extreme high-pressure equipment in the early stages. The result is a synthesis pathway that is not only simpler to operate but also delivers a total yield reaching 52% with a purity profile superior to 99%, making it ideally suited for large-scale industrial application.
Mechanistic Insights into Optimized Esterification and Coupling
The core of this technological advancement lies in the precise control of reaction kinetics and thermodynamics during the esterification and final coupling stages. In the initial esterification step, the use of thionyl chloride in the presence of a catalytic amount of N,N-dimethylformamide facilitates the rapid conversion of the carboxylic acid to the corresponding acid chloride intermediate, which is then immediately trapped by ethanol. This in-situ generation and consumption of the reactive intermediate minimizes side reactions and ensures high conversion rates. The subsequent reduction of the nitro group is carefully managed using a palladium-carbon catalyst in ethyl acetate at temperatures between 50°C and 65°C. This specific solvent and temperature combination is crucial for preventing over-reduction or the formation of hydroxylamine byproducts, which are common pitfalls in nitro reductions. The mechanism ensures that the amino group is generated cleanly, providing a high-quality substrate for the subsequent cyclization reaction with N,N'-carbonyldiimidazole activated D10.
Furthermore, the final coupling reaction between the carboxylic acid intermediate (D7) and the pyridine-containing amine (D8) demonstrates remarkable chemoselectivity. Activated by thionyl chloride in methylene dichloride, the carboxylic acid forms a reactive acyl chloride species that efficiently couples with the amine nucleophile. 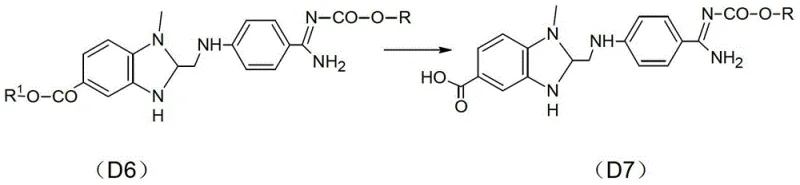 The choice of methylene dichloride as the solvent is strategic, providing excellent solubility for both reactants while maintaining a stable environment for the acid chloride intermediate. The reaction is conducted at a controlled temperature of 40-50°C, which is sufficient to drive the reaction to completion without promoting thermal degradation of the sensitive benzimidazole moiety. This mechanistic precision is what allows the process to achieve such high purity levels without the need for extensive chromatographic purification, effectively solving the impurity burden that has hindered previous manufacturing attempts.
The choice of methylene dichloride as the solvent is strategic, providing excellent solubility for both reactants while maintaining a stable environment for the acid chloride intermediate. The reaction is conducted at a controlled temperature of 40-50°C, which is sufficient to drive the reaction to completion without promoting thermal degradation of the sensitive benzimidazole moiety. This mechanistic precision is what allows the process to achieve such high purity levels without the need for extensive chromatographic purification, effectively solving the impurity burden that has hindered previous manufacturing attempts.
How to Synthesize Dabigatran Etexilate Efficiently
The synthesis of dabigatran etexilate via this novel route involves a sequence of seven well-defined chemical steps, each optimized for maximum yield and minimal waste generation. The process begins with the esterification of the starting material, followed by reduction, cyclization, hydrogenation, protection, deprotection, and finally, the coupling reaction. Each stage has been rigorously tested to ensure reproducibility and scalability, with specific attention paid to stoichiometry and temperature control. For instance, the molar ratio of thionyl chloride to the starting acid is maintained between 1:1.7 and 1:2.4 to ensure complete activation without excessive reagent waste. Similarly, the hydrogenation steps utilize standard 10% palladium-carbon catalysts, which are easily filtered and recycled, contributing to the overall cost-effectiveness of the method.
- Perform esterification of methylamino-m-nitrobenzoate (D1) using thionyl chloride and ethanol to obtain ester (D2).
- Reduce the nitro group in D2 to an amino group using a palladium-carbon catalyst to yield intermediate D3.
- React D3 with compound D10 in the presence of carbonyldiimidazole to form the benzimidazole intermediate D4.
- Hydrogenate intermediate D4 to reduce the nitro-imidazole moiety, yielding the amino-benzimidazole D5.
- React D5 with chloroformate under alkaline conditions to protect the amine, forming intermediate D6.
- Hydrolyze the ester group in D6 using alkali followed by acidification to obtain the carboxylic acid intermediate D7.
- Couple intermediate D7 with 3-(pyridin-2-ylamino)propionate (D8) using thionyl chloride to finalize dabigatran etexilate (D9).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The primary advantage stems from the drastic simplification of the purification process; by eliminating the need for column chromatography and reducing the formation of oily impurities, the manufacturing cycle time is significantly shortened. This efficiency translates into lower labor costs and reduced consumption of expensive chromatographic media and solvents. Additionally, the raw materials required for this process, such as methylamino-m-nitrobenzoate and ethanol, are commodity chemicals that are cheap and easily available on the global market. This availability mitigates supply chain risks associated with sourcing specialized or exotic reagents, ensuring a more stable and continuous production flow. The ability to source materials locally or from multiple vendors enhances supply security, a critical factor for maintaining uninterrupted API production schedules.
- Cost Reduction in Manufacturing: The elimination of high-pressure hydrogenation equipment in the initial stages and the removal of column chromatography purification steps result in substantial capital and operational expenditure savings. The process operates under mild conditions, reducing energy consumption for heating and cooling, while the high selectivity minimizes the loss of valuable intermediates to waste streams. Furthermore, the use of recyclable catalysts and common solvents like ethyl acetate and toluene lowers the overall material cost per kilogram of finished product. These cumulative efficiencies allow for a more competitive pricing structure for the final dabigatran etexilate intermediate, providing a distinct market advantage.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is essential for maintaining regulatory compliance and avoiding costly production delays. The high purity of the intermediates (>98.5% at each stage) reduces the risk of batch rejection and the need for re-processing, which can disrupt supply timelines. Moreover, the process generates significantly less hazardous waste compared to traditional methods, simplifying waste disposal logistics and reducing the environmental footprint. This alignment with green chemistry principles not only lowers disposal costs but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies.
- Scalability and Environmental Compliance: Designed with industrial scale-up in mind, this method avoids the technical barriers that often prevent laboratory successes from translating to commercial production. The reactions are exothermic but manageable, allowing for safe scaling from pilot plants to multi-ton reactors without significant re-engineering. The reduction in three wastes (wastewater, waste gas, and solid waste) discharge makes the process environmentally friendly and easier to permit in regions with strict environmental regulations. This scalability ensures that manufacturers can rapidly respond to surges in market demand for anticoagulants without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel dabigatran etexilate synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing operations or for procurement teams assessing supplier capabilities.
Q: How does the purity of this novel synthesis method compare to conventional routes?
A: Conventional methods often result in oily products containing 20-40% impurities that require difficult column chromatography. In contrast, this novel method described in CN103275065A achieves a final purity of over 99% without the need for complex chromatographic separation, significantly simplifying downstream processing.
Q: What are the safety advantages of this production process?
A: Traditional routes often require high-pressure hydrogenation equipment which poses significant safety risks and technical barriers. This optimized process utilizes mild reaction conditions, such as esterification at 85-100°C and reduction at 50-65°C, eliminating the need for extreme high-pressure setups in the initial stages and reducing operational hazards.
Q: What is the overall yield efficiency of this synthetic pathway?
A: The patent data indicates that the total yield of this seven-step process can reach 52%. This is a substantial improvement over older methods where individual step yields were lower and purification losses were high due to the formation of isomeric impurities and oily byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Etexilate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and high-quality synthesis routes for life-saving medications like dabigatran etexilate. Our team of expert chemists has extensively analyzed the pathway described in CN103275065A and possesses the technical expertise to implement this advanced methodology at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-purity intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications. We are committed to delivering not just a product, but a comprehensive solution that enhances your supply chain resilience.
We invite you to collaborate with us to optimize your anticoagulant supply chain and leverage the cost-saving potential of this novel synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about your sourcing strategy. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable dabigatran etexilate intermediate supplier dedicated to innovation, quality, and long-term partnership success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
