Advanced Metal-Free Synthesis of 3-Quinolinyl-5-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
Advanced Metal-Free Synthesis of 3-Quinolinyl-5-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable pathways to bioactive scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a robust preparation method for 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These nitrogen-containing heterocycles are pivotal structures found in numerous drug intermediates and functional materials, including ligands for organic light-emitting diodes (OLEDs). The patent introduces a transformative oxidative cyclization strategy that bypasses the limitations of classical synthesis, utilizing a catalytic system composed of tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP). This innovation not only streamlines the synthetic route but also aligns with modern green chemistry principles by eliminating the need for toxic heavy metal catalysts and严苛 reaction conditions.
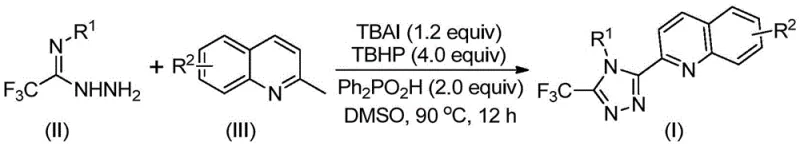
For R&D directors and process chemists, the structural versatility offered by this methodology is particularly compelling. The general formula (I) allows for extensive diversification at the R1 and R2 positions, enabling the rapid generation of libraries for structure-activity relationship (SAR) studies. The ability to introduce electron-withdrawing groups like trifluoromethyl and halogens, alongside electron-donating groups like methoxy and alkyl chains, provides a powerful toolkit for medicinal chemists aiming to optimize pharmacokinetic properties. Furthermore, the compatibility of this reaction with various substituted 2-methylquinolines ensures that a wide array of complex molecular architectures can be accessed from simple, commercially available starting materials, thereby accelerating the drug discovery timeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiency and operational complexity. Traditional protocols typically rely on quinoline-2-formic acid as the primary building block, necessitating a tedious five-step reaction sequence to construct the desired triazole ring. This multi-step approach suffers from a dismal overall yield of approximately 17%, representing a massive loss of valuable material and a significant increase in production costs. Moreover, the reaction conditions required for these classical methods are often severe, demanding strict control over parameters that are difficult to maintain on a large scale. The reliance on expensive or hazardous reagents in these older pathways further exacerbates the economic and environmental burden, making them unsuitable for the cost-sensitive and sustainability-focused demands of modern pharmaceutical manufacturing.
The Novel Approach
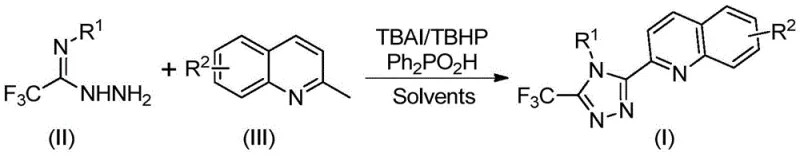
In stark contrast, the novel method disclosed in the patent leverages a direct oxidative coupling between trifluoroethylimine hydrazide and 2-methylquinoline derivatives. This one-pot transformation is catalyzed by the TBAI/TBHP system in the presence of diphenylphosphoric acid, operating effectively at moderate temperatures between 80°C and 100°C. By shifting the starting material from quinoline-2-formic acid to the much cheaper and more accessible 2-methylquinoline, the process achieves a dramatic reduction in raw material costs. The reaction proceeds through an elegant cascade involving oxidation, condensation, and cyclization, delivering the target triazole compounds in high yields, with some examples reaching up to 97%. This streamlined approach not only simplifies the operational workflow but also significantly enhances the atom economy of the synthesis.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The success of this transformation hinges on the synergistic interaction between the iodide salt and the peroxide oxidant. Mechanistically, the tetrabutylammonium iodide (TBAI) acts as a catalyst that facilitates the oxidation of the methyl group on the 2-methylquinoline substrate. In the presence of tert-butyl peroxide (TBHP), the methyl group is converted into a reactive 2-quinoline carbaldehyde intermediate in situ. This aldehyde then undergoes a condensation reaction with the trifluoroethylimine hydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination and intramolecular electrophilic substitution trigger the cyclization event, followed by aromatization to yield the stable 1,2,4-triazole core. This radical-mediated or cationic pathway avoids the need for pre-functionalized substrates, allowing for the direct use of simple methyl-arenes.
Furthermore, the inclusion of diphenylphosphoric acid plays a crucial role in modulating the reaction environment and potentially stabilizing key intermediates. The use of dimethyl sulfoxide (DMSO) as the solvent is critical, as it effectively dissolves both the organic substrates and the ionic catalyst while promoting the oxidative process. The tolerance of the system to water, evidenced by the use of aqueous TBHP solution, is a notable feature that relaxes the requirement for strictly anhydrous conditions. This mechanistic robustness ensures that the reaction can proceed smoothly even with slight variations in reagent quality, providing a reliable platform for the synthesis of diverse triazole derivatives without the formation of significant impurities that would complicate downstream purification.
How to Synthesize 3-Quinolinyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and thermal management, although the procedure itself is remarkably straightforward. The protocol involves charging a reaction vessel with the requisite amounts of tetrabutylammonium iodide, aqueous tert-butyl peroxide, diphenylphosphoric acid, the specific trifluoroethylimine hydrazide derivative, and the 2-methylquinoline substrate. These components are suspended or dissolved in an appropriate volume of DMSO, typically ranging from 5 to 10 mL per mmol of the quinoline starting material. The mixture is then heated to a temperature of 90°C and stirred for a duration of 12 hours to ensure full conversion. Upon completion, the workup involves a simple filtration step followed by silica gel treatment and column chromatography, yielding the pure product ready for characterization.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP) aqueous solution, diphenylphosphoric acid, trifluoroethylimine hydrazide, and 2-methylquinoline derivative in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete oxidative cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic benefits that extend beyond mere chemical efficiency. The elimination of heavy metal catalysts is a primary driver for cost reduction, as it removes the need for expensive scavenging resins and complex purification steps required to meet stringent residual metal limits in pharmaceutical ingredients. Additionally, the starting materials, specifically 2-methylquinoline and various aromatic amines used to prepare the hydrazides, are commodity chemicals available from multiple global suppliers, ensuring a resilient and competitive supply base. The simplicity of the reaction conditions, which do not require inert atmosphere techniques or cryogenic cooling, further lowers the barrier to entry for contract manufacturing organizations (CMOs), allowing for broader sourcing options and reduced manufacturing lead times.
- Cost Reduction in Manufacturing: The transition from a five-step sequence to a single-pot reaction fundamentally alters the cost structure of producing these intermediates. By consolidating multiple synthetic operations into one, manufacturers save significantly on labor, energy, and solvent consumption. The avoidance of precious metal catalysts eliminates a major variable cost component, while the high yields observed across a broad substrate scope minimize waste disposal costs. This holistic reduction in operational complexity translates directly into a lower cost of goods sold (COGS), providing a distinct competitive advantage in the pricing of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-proprietary reagents mitigates the risk of supply disruptions often associated with specialized or custom-synthesized catalysts. Since the process tolerates aqueous conditions and does not demand ultra-dry solvents, the logistics of raw material storage and handling are simplified, reducing the potential for spoilage or degradation prior to use. This robustness ensures consistent production output, which is critical for maintaining uninterrupted supply lines to downstream API manufacturers. The ability to source key inputs from standard chemical distributors enhances the overall agility of the supply chain, allowing for rapid scaling in response to market demand fluctuations.
- Scalability and Environmental Compliance: The inherent safety profile of this metal-free oxidation supports easier scale-up from gram to kilogram and ton scales without the need for specialized reactor configurations. The absence of toxic heavy metals simplifies wastewater treatment and waste stream management, facilitating compliance with increasingly rigorous environmental regulations. The use of DMSO, a solvent with well-established recovery and recycling protocols, further enhances the sustainability credentials of the process. These factors collectively make the technology highly attractive for long-term commercial production, ensuring that the manufacturing process remains viable and compliant as production volumes increase to meet global pharmaceutical needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These insights are derived directly from the experimental data and scope analysis presented in the patent documentation, providing clarity on substrate compatibility and process robustness. Understanding these nuances is essential for project managers evaluating the feasibility of adopting this route for their specific pipeline candidates.
Q: What are the key advantages of this metal-free synthesis over traditional methods?
A: Unlike traditional methods requiring quinoline-2-formic acid and five reaction steps with low yields, this novel approach utilizes cheap 2-methylquinoline and operates under mild, metal-free conditions, drastically simplifying purification and reducing raw material costs.
Q: Can this process tolerate diverse functional groups on the quinoline ring?
A: Yes, the method demonstrates excellent substrate tolerance, successfully accommodating substituents such as halogens (Br, Cl), alkyl groups (methyl), alkoxy groups (methoxy), and nitro groups at various positions on the quinoline scaffold without compromising yield.
Q: Is this synthesis suitable for large-scale industrial production?
A: The process is highly scalable as it avoids strict anhydrous or oxygen-free conditions and utilizes commercially available, inexpensive reagents like TBAI and TBHP, making it ideal for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolinyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the potential of the metal-free oxidative cyclization described in CN113307790B and is well-positioned to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-quinolinyl-5-trifluoromethyl-1,2,4-triazole intermediate meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to explore how this advanced synthesis can optimize your supply chain and reduce your overall project costs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your novel analogues. Let us be your partner in turning complex chemical challenges into commercial successes.
