Advanced Cobalt-Catalyzed Synthesis of Tetrahydro-beta-carbolinones for Commercial Scale-up
Advanced Cobalt-Catalyzed Synthesis of Tetrahydro-beta-carbolinones for Commercial Scale-up
The pharmaceutical industry continuously seeks robust, scalable, and cost-efficient synthetic routes for complex heterocyclic scaffolds that serve as the backbone for bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN115260188A, which discloses a novel preparation method for tetrahydro-beta-carbolinone compounds. These nitrogen-containing heterocycles are pivotal structural motifs found in numerous natural products and drug candidates, including the antiviral agent bauerine C and the anxiolytic candidate SL651498. The patent introduces a transition metal cobalt-catalyzed C-H activated carbonylation reaction that utilizes tryptamine derivatives as starting materials. This methodology represents a paradigm shift from traditional noble metal catalysis, offering a streamlined pathway that is not only operationally simple but also highly compatible with diverse functional groups, thereby addressing critical pain points in the manufacturing of high-purity pharmaceutical intermediates.
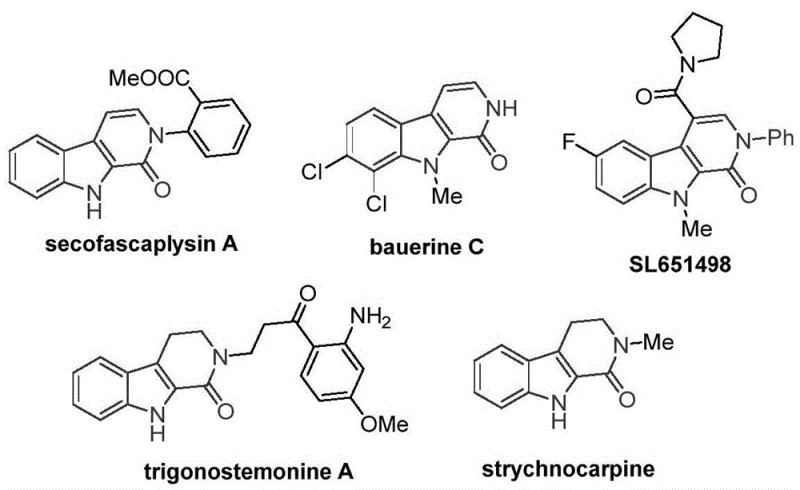
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrahydro-beta-carbolinone skeleton via carbonylation reactions has been heavily reliant on palladium catalysis. While effective, palladium-based methodologies suffer from inherent drawbacks that pose significant challenges for large-scale commercial production. The primary concern is the exorbitant cost of palladium precursors, which directly impacts the cost of goods sold (COGS) for the final active pharmaceutical ingredient (API). Furthermore, palladium is a heavy metal that requires stringent removal processes to meet regulatory limits for residual metals in drug substances, often necessitating additional purification steps such as scavenging or recrystallization. These extra steps reduce overall yield and increase processing time. Additionally, conventional methods often exhibit limited substrate scope, struggling with electron-rich or sterically hindered tryptamine derivatives, which restricts the chemical diversity accessible to medicinal chemists during lead optimization phases.
The Novel Approach
In stark contrast, the method disclosed in CN115260188A leverages a cobalt-catalyzed system that effectively circumvents the limitations of palladium chemistry. By employing cobalt acetate tetrahydrate as the catalyst, the process utilizes an earth-abundant base metal that is orders of magnitude cheaper than palladium. The reaction proceeds through a C-H activation mechanism that is highly efficient, utilizing 1,3,5-tricarboxylic acid phenol ester as a safe and convenient carbon monoxide surrogate, eliminating the need for handling toxic CO gas. This novel approach demonstrates exceptional substrate compatibility, successfully tolerating various substituents including halogens, alkyl, and alkoxy groups at different positions on the indole ring. The operational simplicity, combined with the ability to perform the reaction in common solvents like dioxane at moderate temperatures (120-140°C), makes this technology a superior candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Cobalt-Catalyzed C-H Activation Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific analogs. The reaction initiates with the oxidation of the cobalt(II) catalyst by silver carbonate (Ag2CO3), generating a reactive cobalt(III) species in situ. This high-valent cobalt center then coordinates with the tryptamine derivative, specifically directing the activation of the C-H bond at the 2-position of the indole ring. This C-H activation step is facilitated by the pivalic acid additive, which likely acts as a proton shuttle, lowering the energy barrier for metallation. Following the formation of the cobalt(III)-aryl intermediate, the carbon monoxide released from the thermal decomposition of the phenyl ester surrogate inserts into the cobalt-carbon bond. This insertion generates an acyl-cobalt(III) intermediate, setting the stage for the final ring-closing event.

The final step involves a reductive elimination coupled with hydrolysis, which releases the tetrahydro-beta-carbolinone product and regenerates the active cobalt species to continue the catalytic cycle. From an impurity control perspective, this mechanism is advantageous because the oxidative conditions and the specific coordination geometry of the cobalt center minimize side reactions such as homocoupling or over-carbonylation. The use of silver carbonate as both an oxidant and a halide scavenger (if halogenated substrates are used) further ensures a clean reaction profile. The robustness of this catalytic cycle allows for high turnover numbers, evidenced by the high yields reported across a broad range of substrates, ensuring that the final product meets the stringent purity specifications required for downstream pharmaceutical applications.
How to Synthesize Tetrahydro-beta-carbolinone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The procedure involves charging a reaction vessel with the cobalt catalyst, base, additive, oxidant, CO surrogate, and the tryptamine substrate in an organic solvent. The mixture is then heated to facilitate the cyclization. This standardized approach minimizes variability and ensures reproducible results. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with cobalt catalyst (Co(OAc)2·4H2O), base (Et3N), additive (PivOH), oxidant (Ag2CO3), CO surrogate (TFBen), and tryptamine derivative in dioxane.
- Heat the reaction mixture to 120-140°C and stir for 16-24 hours to facilitate C-H activation and carbonylation cyclization.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target tetrahydro-beta-carbolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed technology offers tangible strategic benefits beyond mere technical feasibility. The shift from precious metals to base metals fundamentally alters the cost structure of the synthesis. By eliminating the dependency on volatile palladium markets, manufacturers can achieve significant cost savings in raw material procurement. Moreover, the simplified purification process, resulting from the absence of difficult-to-remove palladium residues, reduces the consumption of specialized scavengers and solvents, further driving down operational expenditures. The use of commercially available and stable reagents, such as cobalt acetate and silver carbonate, ensures a reliable supply chain with minimal risk of disruption, unlike some exotic ligands often required in palladium chemistry.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with inexpensive cobalt salts drastically reduces the direct material cost per kilogram of the intermediate. Additionally, the high reaction efficiency and yield minimize waste generation and maximize throughput, leading to substantial overall cost optimization. The avoidance of toxic carbon monoxide gas in favor of a solid CO surrogate also reduces safety infrastructure costs and regulatory compliance burdens associated with hazardous gas handling.
- Enhanced Supply Chain Reliability: All key reagents, including the cobalt catalyst, oxidants, and additives, are commodity chemicals available from multiple global suppliers. This multi-sourcing capability mitigates the risk of single-source bottlenecks. The robustness of the reaction conditions (120-140°C) allows for the use of standard stainless steel reactors without the need for specialized high-pressure equipment, facilitating easier technology transfer between manufacturing sites and ensuring consistent supply continuity for long-term projects.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable, with the patent noting successful gram-scale synthesis. The simplified workup involving filtration and standard column chromatography is easily adaptable to industrial crystallization or extraction processes. Furthermore, the reduced heavy metal load in the waste stream simplifies effluent treatment, aligning with increasingly strict environmental regulations and supporting sustainable manufacturing practices (Green Chemistry principles) which are becoming a key requirement for major pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit for your specific project needs. Understanding these details is essential for making informed decisions about process adoption and vendor selection.
Q: What is the primary advantage of this cobalt-catalyzed method over traditional palladium methods?
A: The primary advantage is the replacement of expensive and toxic palladium catalysts with earth-abundant, low-cost cobalt salts, significantly reducing raw material costs and heavy metal residue risks in the final API.
Q: What is the substrate scope for this carbonylation reaction?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as methyl, methoxy, halogens (Cl, Br), and different N-substituents (benzyl, allyl, phenyl) on the tryptamine scaffold.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states the method is simple to operate, uses commercially available reagents, and has been validated for gram-scale synthesis, indicating strong potential for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-beta-carbolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the cobalt-catalyzed methodology described in CN115260188A and is well-positioned to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of tetrahydro-beta-carbolinone intermediate we deliver meets the highest quality standards required by global regulatory agencies.
We invite you to collaborate with us to unlock the full potential of this cost-effective synthesis. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your drug development timeline while optimizing your budget. Let us be your partner in delivering high-quality pharmaceutical intermediates with speed and precision.
