Advanced Synthesis of 4,5-Diaryl-2H-1,2,3-Triazoles for Commercial API Production
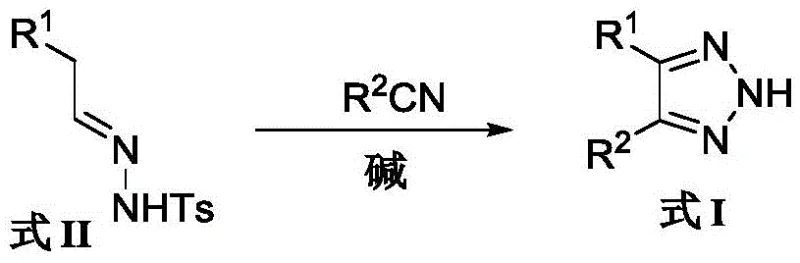
The pharmaceutical industry continuously seeks robust synthetic pathways for heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN108794412B introduces a transformative preparation method for 4,5-diaryl-2H-1,2,3-triazole compounds, addressing long-standing challenges in yield, safety, and purity. These triazole derivatives are not merely academic curiosities; they are potent structural analogs of combretastatin A-4, exhibiting significant antitumor activity against various human cancer cell lines. For R&D directors and procurement specialists, this patent represents a pivotal shift away from hazardous azide chemistry toward a more sustainable, base-promoted cycloaddition strategy. By leveraging aromatic aldehyde sulfonyl hydrazones and aromatic nitriles, this technology enables the efficient construction of the triazole core under mild conditions, ensuring a reliable supply of high-purity pharmaceutical intermediates for oncology drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazoles has been dominated by cycloaddition reactions involving aryl alkynes and sodium azide or its derivatives. While chemically feasible, these traditional routes suffer from severe drawbacks that hinder their application in regulated commercial manufacturing. The primary concern is the inherent toxicity and explosive nature of azide reagents, which necessitates specialized equipment, rigorous safety protocols, and expensive waste disposal procedures, thereby inflating the overall cost of goods. Furthermore, alternative methods reported by researchers like Peter A. Crooks, which utilize functionalized alkenes and azides, still rely on these hazardous materials and require complex multi-step preparations of the alkene precursors. Another significant bottleneck identified in prior art, such as the work by Subhankar Panda using cesium carbonate, is the formation of stubborn self-coupling byproducts. When different aryl aldehyde sulfonyl hydrazones are employed, the reaction indiscriminately generates both cross-coupled and self-coupled triazoles. These impurities possess polarities and molecular sizes nearly identical to the target product, making downstream purification via chromatography extremely difficult and resulting in poor overall yields, often below 50 percent.
The Novel Approach
The methodology disclosed in patent CN108794412B offers a decisive break from these inefficiencies by utilizing a specific combination of strong bases, namely potassium tert-butoxide (t-BuOK) or sodium bis(trimethylsilyl)amide (NaHMDS), to promote the reaction between formula II compounds and nitriles. This strategic selection of reagents fundamentally alters the reaction pathway, effectively suppressing the formation of self-coupling impurities that plague earlier hydrazone-based methods. The result is a clean transformation where the expected 4,5-diaryl-2H-1,2,3-triazole is generated with high conversion rates and minimal side products. Moreover, the reaction conditions are remarkably mild, operating optimally between 60°C and 80°C, which is significantly lower than the harsh temperatures required by previous techniques. This reduction in thermal stress not only enhances energy efficiency but also preserves the integrity of sensitive functional groups on the aromatic rings, allowing for a broader substrate scope that includes halo, alkoxy, and alkyl substituents without degradation.
Mechanistic Insights into Base-Promoted Cyclization
From a mechanistic perspective, the success of this synthesis lies in the precise activation of the sulfonyl hydrazone precursor by the strong non-nucleophilic base. Unlike weaker bases such as cesium carbonate, t-BuOK and NaHMDS facilitate the rapid generation of a reactive diazo or diazene intermediate in situ, which then undergoes a concerted [3+2] cycloaddition with the nitrile dipolarophile. This pathway is kinetically favored over the dimerization of the hydrazone species, which explains the drastic reduction in self-coupling impurities observed in experimental data. For the R&D team, understanding this selectivity is crucial for impurity profiling; the absence of homologous byproducts simplifies the impurity spectrum, ensuring that the final active pharmaceutical ingredient (API) meets stringent regulatory standards for genotoxic impurities. The reaction proceeds through a cyclic transition state that efficiently closes the triazole ring, locking the two aryl groups into the 4 and 5 positions with high regioselectivity. This mechanistic clarity allows process chemists to confidently predict outcomes when scaling up, knowing that the reaction is driven by thermodynamic stability rather than stochastic collisions.
Furthermore, the choice of solvent plays a pivotal role in stabilizing the ionic intermediates formed during the base promotion. The patent highlights the efficacy of polar aprotic solvents like DMF, NMP, and dioxane, as well as hydrocarbon solvents like toluene and xylene. These solvents provide the necessary dielectric environment to solvate the cationic counter-ions of the base while keeping the organic substrates in solution, ensuring homogeneous reaction kinetics. This solubility profile is essential for maintaining consistent reaction rates across large batches, preventing localized hot spots that could lead to decomposition. The compatibility with a wide range of solvents also offers flexibility for process optimization; for instance, switching to toluene might be preferred for easier solvent recovery and lower environmental impact during commercial scale-up. The robustness of this mechanism against varying electronic effects on the aryl rings—whether electron-withdrawing trifluoromethyl groups or electron-donating methoxy groups—demonstrates the versatility of the catalytic system, making it a universal platform for synthesizing diverse triazole libraries.
How to Synthesize 4,5-Diaryl-2H-1,2,3-Triazole Efficiently
Implementing this synthesis route requires strict adherence to the optimized molar ratios and temperature profiles established in the patent to maximize yield and minimize waste. The process begins with the dissolution of the aromatic aldehyde sulfonyl hydrazone and the aromatic nitrile in a selected organic solvent, followed by the controlled addition of the base under inert atmosphere to prevent moisture interference. The reaction mixture is then heated to the specified range, typically between 60°C and 100°C, and maintained for a duration of 3 to 4 hours to ensure complete consumption of the starting materials. Monitoring via TLC or HPLC is recommended to confirm the disappearance of the hydrazone precursor before proceeding to the quenching step. Upon completion, the reaction is carefully quenched with saturated ammonium chloride solution to neutralize excess base, followed by extraction and standard purification techniques. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Dissolve the aromatic aldehyde sulfonyl hydrazone and aromatic nitrile in an organic solvent such as toluene, DMF, or dioxane.
- Add a strong base like t-BuOK or NaHMDS under stirring and heat the mixture to 60-100°C for 3-4 hours.
- Quench the reaction with saturated ammonium chloride, extract with ethyl acetate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into tangible operational efficiencies and risk mitigation. The elimination of sodium azide from the synthetic route removes a major regulatory and safety hurdle, significantly reducing the costs associated with hazardous material handling, storage, and disposal. This safety improvement also streamlines the approval process for manufacturing sites, as facilities do not require specialized explosion-proof infrastructure dedicated to azide chemistry. Additionally, the high yields reported in the patent examples, often exceeding 85 percent and reaching up to 96 percent in optimized cases, imply a substantial reduction in raw material consumption per kilogram of finished product. This material efficiency is a key driver for cost reduction in pharmaceutical intermediate manufacturing, allowing for more competitive pricing in a crowded market. The simplified purification process, resulting from the lack of difficult-to-remove self-coupling impurities, further lowers production costs by reducing solvent usage and chromatography resin requirements.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and hazardous azide reagents, replacing them with commercially abundant nitriles and hydrazones. By avoiding the complex purification steps required to remove self-coupling byproducts found in older methods, the overall processing time and solvent consumption are drastically reduced. This streamlined workflow leads to significant cost savings in both raw materials and downstream processing operations, enhancing the overall profit margin for the final API.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted benzaldehydes and benzonitriles, are commodity chemicals available from multiple global suppliers, ensuring a stable and continuous supply chain. The robustness of the reaction conditions means that production is less susceptible to delays caused by sensitive reagent shortages or strict transportation regulations associated with explosives. This reliability allows for better inventory planning and reduces the risk of production stoppages due to supply chain disruptions.
- Scalability and Environmental Compliance: Operating at moderate temperatures of 60°C to 80°C makes this process highly energy-efficient and easily scalable from laboratory to pilot and commercial plants. The use of common organic solvents like toluene and DMF facilitates straightforward solvent recovery and recycling systems, aligning with modern green chemistry principles. Furthermore, the high atom economy and reduced waste generation simplify wastewater treatment processes, ensuring compliance with increasingly stringent environmental regulations without requiring costly additional remediation technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing workflows. The responses cover aspects of safety, purity, and scalability to assist in decision-making processes.
Q: Why is this new triazole synthesis method safer than traditional azide routes?
A: Traditional methods often rely on sodium azide, which is highly toxic and explosive. This patent utilizes aromatic nitriles and sulfonyl hydrazones with a base, eliminating the safety risks associated with handling hazardous azide reagents.
Q: How does this process improve product purity compared to previous hydrazone coupling methods?
A: Previous methods using cesium carbonate often resulted in significant self-coupling impurities that were difficult to separate. This novel approach using t-BuOK or NaHMDS suppresses self-coupling, leading to higher conversion rates and easier purification.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the reaction operates at moderate temperatures (60-100°C) and completes within 3-4 hours using readily available solvents and reagents, making it highly scalable for industrial manufacturing without requiring extreme conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Diaryl-2H-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of oncology therapeutics. Our team of expert process chemists has extensively evaluated the technology disclosed in CN108794412B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering these complex triazole compounds with stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the exacting standards required for clinical and commercial applications. Our state-of-the-art facilities are equipped to handle the specific solvent and base requirements of this synthesis, guaranteeing consistent quality and supply continuity for your drug development programs.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline to market while optimizing your production budget.
