Scalable Manufacturing of Prasugrel Intermediates via Safe Catalytic Oxidation Technology
Introduction to Advanced Prasugrel Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust and safe pathways for synthesizing critical antiplatelet agents like Prasugrel. Patent CN102241689A introduces a groundbreaking methodology for preparing the key intermediate, 2,4,5,6,7,7alpha-hexahydro thieno[3,2-c]pyridine-2-ketone, which serves as the structural core for this vital medication. Unlike legacy processes that rely on hazardous reagents, this innovation leverages a metal salt-catalyzed oxidation strategy under high-pressure oxygen atmospheres to achieve superior safety profiles and yields. The target molecule, depicted below, represents a pivotal junction in the synthetic pathway where efficiency gains translate directly to commercial viability. By shifting away from cryogenic organometallic chemistry, this process addresses the fundamental pain points of industrial scalability and operator safety.
![Chemical structure of 2,4,5,6,7,7alpha-hexahydro thieno[3,2-c]pyridine-2-ketone](/insights/img/prasugrel-intermediate-synthesis-catalysis-pharma-supplier-20260307024311-01.png)
This technological advancement is particularly relevant for reliable pharmaceutical intermediate supplier networks aiming to secure the supply chain for cardiovascular therapeutics. The method encompasses a three-stage sequence: protection of the nitrogen center, catalytic oxidation of the thiophene ring system, and subsequent deprotection. Each step has been optimized to minimize waste and maximize throughput, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates. As global demand for generic versions of Efient rises, the ability to produce this core scaffold efficiently becomes a strategic asset for procurement teams focused on cost reduction in pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thienopyridine derivatives has been plagued by significant safety hazards and operational complexities. Prior art, such as the route described in US470510, relies heavily on the use of n-Butyl Lithium, a highly pyrophoric reagent that demands stringent handling protocols and specialized infrastructure. This reagent must be utilized at cryogenic temperatures, typically around -40°C, to control reactivity and prevent thermal runaway or explosion. Such extreme conditions not only inflate energy consumption but also limit the batch size due to heat transfer constraints in large reactors. Furthermore, the sensitivity of organolithium species to moisture and oxygen necessitates an inert atmosphere throughout the process, increasing the capital expenditure for equipment and maintenance. These factors collectively create a bottleneck for reducing lead time for high-purity pharmaceutical intermediates, as any deviation in temperature or atmosphere can lead to catastrophic batch failures.
The Novel Approach
In stark contrast, the methodology outlined in CN102241689A replaces these dangerous reagents with a benign catalytic system driven by molecular oxygen. The novel approach utilizes inexpensive metal salts, such as iron(III) chloride or copper sulfate, to mediate the oxidation of the protected tetrahydro-thienopyridine precursor. This reaction proceeds in a mixed solvent system of acetic acid and water at ambient temperature, eliminating the need for energy-intensive cooling systems. The use of oxygen pressure (0.3 to 2 MPa) in an autoclave is a standard unit operation in the fine chemical industry, allowing for seamless translation from laboratory to multi-ton production scales. By removing the dependency on exotic, hazardous reagents, this route significantly simplifies the process workflow and enhances the inherent safety of the manufacturing plant. This shift represents a paradigm change towards greener chemistry, aligning with modern environmental regulations while simultaneously driving down production costs through simplified engineering controls.
Mechanistic Insights into Metal Salt-Catalyzed Oxidation
The core innovation of this patent lies in the efficient transformation of the sulfur-containing heterocycle into the corresponding ketone functionality. The mechanism involves the activation of molecular oxygen by the transition metal catalyst, likely forming a high-valent metal-oxo species or facilitating a radical chain reaction that targets the alpha-position of the sulfur atom. In the presence of acetic acid and water, the metal salt acts as a Lewis acid and redox mediator, promoting the insertion of oxygen into the carbon-sulfur framework without degrading the sensitive piperidine ring. The solvent mixture plays a crucial role in stabilizing the transition states and solubilizing both the organic substrate and the inorganic catalyst, ensuring homogeneous reaction kinetics. This precise control over the oxidation state prevents over-oxidation to sulfones or sulfonic acids, which are common impurities in less selective oxidative processes.
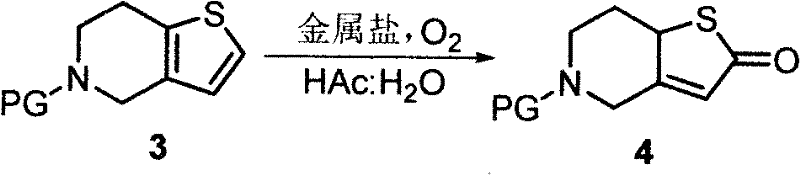
Impurity control is further enhanced by the mild reaction conditions employed in this protocol. Traditional strong oxidants often lead to non-selective attack on the electron-rich aromatic rings or the amine functionality, generating complex byproduct profiles that are difficult to purge. However, the metal-catalyzed aerobic oxidation described here exhibits high chemoselectivity, primarily targeting the specific methylene group adjacent to the sulfur. The subsequent workup involves simple pH adjustment and extraction, which effectively removes metal residues and acidic byproducts. This results in a crude product of high purity, reducing the burden on downstream purification steps such as chromatography or recrystallization. For R&D directors, this implies a more robust process window where minor fluctuations in parameters do not compromise the quality of the high-purity pharmaceutical intermediate, ensuring consistent batch-to-batch reproducibility essential for regulatory compliance.
How to Synthesize 2,4,5,6,7,7alpha-hexahydro thieno[3,2-c]pyridine-2-ketone Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for executing this transformation with industrial rigor. The process begins with the protection of the secondary amine in the starting material, typically using acetic anhydride in the presence of a base like diisopropylethylamine, which sets the stage for the subsequent oxidation by preventing N-oxide formation. Following isolation of the protected intermediate, the critical oxidation step is performed in a pressurized vessel, leveraging the catalytic power of iron or copper salts to drive the reaction to completion. Finally, the protecting group is cleaved under basic conditions to reveal the free amine of the target ketone.
- Protect the amido group of 4,5,6,7-tetrahydro thieno[3,2-c]pyridine hydrochloride using acetic anhydride and a base like diisopropylethylamine in acetonitrile to form the protected intermediate.
- Perform catalytic oxidation on the protected intermediate using a metal salt catalyst such as iron(III) chloride or copper sulfate in an acetic acid and water solvent system under oxygen pressure.
- Remove the protecting group from the oxidized product using a base such as sodium methoxide in methanol to yield the final target ketone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The elimination of cryogenic operations and pyrophoric reagents fundamentally alters the cost structure of manufacturing, removing the need for specialized low-temperature reactors and extensive safety containment systems. This simplification allows for the utilization of standard stainless steel equipment found in most multipurpose chemical plants, thereby lowering the barrier to entry for contract manufacturing organizations. Moreover, the reliance on commodity chemicals like oxygen, acetic acid, and iron salts insulates the supply chain from the volatility associated with specialized organometallic reagents, ensuring greater continuity of supply even during market disruptions.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalytic aerobic oxidation is profound, primarily driven by the removal of expensive and hazardous reagents like n-Butyl Lithium. By operating at room temperature rather than -40°C, the process eliminates the substantial energy costs associated with industrial refrigeration and cryogenic cooling loops. Additionally, the use of earth-abundant metal catalysts instead of precious metals or stoichiometric oxidants significantly lowers the raw material bill of goods. The high yields reported in the patent examples suggest that less starting material is wasted, further enhancing the overall atom economy and reducing the cost per kilogram of the final active pharmaceutical ingredient. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain or reinvested in further process optimization.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain reliability, as accidents can lead to prolonged plant shutdowns and regulatory scrutiny. By replacing a high-risk organolithium step with a controlled oxidation in an autoclave, the operational risk profile is drastically reduced, minimizing the likelihood of unplanned production stoppages. The reagents required for this process, including metal salts and common solvents, are widely available from multiple global suppliers, reducing the risk of single-source dependency. This diversification of the supply base ensures that production schedules can be maintained consistently, providing pharmaceutical customers with the confidence of on-time delivery for their critical cardiovascular drug portfolios.
- Scalability and Environmental Compliance: The transition from batch to continuous or large-scale batch processing is facilitated by the simplicity of the reaction conditions, which do not require complex mixing or heat exchange setups typical of cryogenic reactions. The use of oxygen as the terminal oxidant generates water as the primary byproduct, aligning with green chemistry principles and simplifying waste stream management. This reduces the environmental footprint of the manufacturing process, helping companies meet increasingly stringent sustainability goals and regulatory requirements regarding hazardous waste disposal. The scalability of the autoclave oxidation step ensures that production volumes can be ramped up quickly to meet surging market demand without the need for extensive process re-engineering or new facility construction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing frameworks. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What are the safety advantages of this new synthesis method compared to traditional routes?
A: Traditional methods often rely on n-Butyl Lithium, which is pyrophoric and requires cryogenic conditions (-40°C), posing significant explosion risks. This patented method utilizes stable metal salt catalysts under controlled oxygen pressure at room temperature, drastically improving operational safety for large-scale production.
Q: Which metal salts are preferred for the catalytic oxidation step?
A: The patent specifies that iron(III) chloride and copper sulfate are the preferred catalysts for the oxidation step. These inexpensive and readily available metal salts facilitate the conversion of the tetrahydro-thienopyridine derivative to the corresponding ketone with high efficiency.
Q: How does this process impact the overall yield of Prasugrel synthesis?
A: By avoiding the harsh conditions and side reactions associated with organolithium reagents, this method achieves higher yields in the intermediate steps. The patent reports yields of approximately 89% for protection, 73% for oxidation, and 90% for deprotection, contributing to a superior total yield for the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5,6,7,7alpha-hexahydro thieno[3,2-c]pyridine-2-ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key intermediates for the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of multinational drug developers. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2,4,5,6,7,7alpha-hexahydro thieno[3,2-c]pyridine-2-ketone meets the highest international standards. Our facility is equipped to handle the specific requirements of metal-catalyzed oxidations safely and efficiently, leveraging the insights from patents like CN102241689A to optimize our internal processes.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this safer, more efficient methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of our high-performance intermediates into your supply chain.
