Scalable Electrocatalytic Reduction of Carbonyls: A Green Route for High-Purity Pharmaceutical Intermediates
Scalable Electrocatalytic Reduction of Carbonyls: A Green Route for High-Purity Pharmaceutical Intermediates
The landscape of organic synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, driven by the urgent need to reduce environmental footprints and operational costs in the chemical industry. Patent CN114182272B introduces a groundbreaking preparation method for alcohol and pinacol derivatives that leverages electrocatalysis to overcome the limitations of traditional thermal and metal-catalyzed processes. This technology enables the efficient reduction of carbonyl compounds under remarkably mild conditions—specifically at room temperature and atmospheric pressure—without the reliance on hazardous transition metal catalysts or harsh reducing agents. By utilizing a simple electrochemical cell with platinum electrodes and DABCO as a sacrificial anode agent, this method achieves yields ranging from 52% to 95%, demonstrating exceptional efficiency and broad substrate tolerance. For R&D directors and process chemists, this represents a significant advancement in constructing diverse compound frameworks essential for biomedical molecules and functional materials.
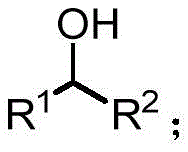
The core innovation lies in the ability to generate high-purity alcohol and pinacol derivatives through a direct electron transfer mechanism, bypassing the complex waste streams associated with stoichiometric metal hydrides. The general structure of the resulting alcohol derivatives, as depicted in the patent, highlights the versatility of this approach in accommodating various aryl, heteroaryl, and alkyl substituents. This structural flexibility is critical for the pharmaceutical industry, where minor modifications to a molecular scaffold can drastically alter biological activity. Furthermore, the method's compatibility with air atmospheres eliminates the need for inert gas protection, simplifying the operational setup and reducing the capital expenditure required for specialized gloveboxes or Schlenk lines. As a reliable pharmaceutical intermediate supplier, understanding such transformative technologies is key to maintaining a competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of alcohols and diols from carbonyl precursors has relied heavily on methods that pose significant safety and environmental challenges. Conventional metal-catalyzed hydrogenation often necessitates high temperatures and high pressures, requiring robust and expensive reactor infrastructure to ensure safety. Moreover, the use of stoichiometric reducing agents like lithium aluminum hydride or sodium borohydride generates substantial amounts of inorganic waste, complicating downstream processing and increasing the burden on wastewater treatment facilities. Another critical drawback is the potential for heavy metal contamination; residual catalysts such as palladium, platinum, or nickel can be notoriously difficult to remove to the stringent parts-per-million levels required for active pharmaceutical ingredients (APIs). These factors collectively contribute to higher production costs, longer lead times, and a larger environmental footprint, making traditional routes less attractive for modern, sustainability-focused manufacturing.
The Novel Approach
In stark contrast, the electrocatalytic method disclosed in CN114182272B offers a streamlined, metal-free alternative that operates under ambient conditions. By replacing thermal energy with electrical energy, the reaction avoids the safety risks associated with high-pressure hydrogen gas and exothermic runaway reactions. The use of DABCO (1,4-diazabicyclo[2.2.2]octane) as a tertiary amine additive serves a dual purpose: it acts as a base to facilitate proton transfer and functions as a sacrificial agent at the anode, preventing electrode passivation and ensuring stable current flow. This approach not only simplifies the reaction setup but also significantly enhances the functional group tolerance, allowing sensitive moieties to remain intact during the reduction process. The result is a cleaner reaction profile with fewer by-products, which translates directly to simplified purification steps and higher overall process efficiency for the production of complex organic intermediates.
Mechanistic Insights into Electrocatalytic Carbonyl Reduction
The mechanistic pathway of this electrochemical transformation involves the generation of reactive radical anion intermediates at the cathode surface. When a constant current is applied across the platinum electrodes, electrons are transferred to the carbonyl substrate, forming a ketyl radical anion. In the presence of the proton source (which can be the solvent or added water/deuterium oxide), this radical species undergoes further reduction and protonation to yield the corresponding alcohol. Alternatively, under specific conditions, two ketyl radicals can couple to form a carbon-carbon bond, resulting in the formation of pinacol derivatives (1,2-diols). This divergence in product selectivity—between simple reduction to alcohols and dimerization to pinacols—is governed by the reaction parameters, such as current density, substrate concentration, and the nature of the electrolyte. The ability to toggle between these pathways using the same fundamental setup provides chemists with a powerful tool for divergent synthesis.
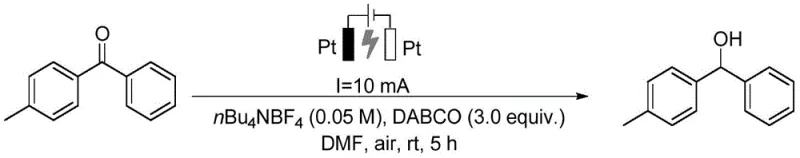
Impurity control is inherently superior in this electrochemical system due to the absence of external chemical oxidants or reductants that often introduce side reactions. The primary impurities typically arise from over-reduction or incomplete conversion, both of which can be finely tuned by monitoring the charge passed (coulometry) during the electrolysis. The patent data indicates that purification is straightforward, often requiring only a single silica gel column chromatography step to achieve purities exceeding 99%. This high level of purity is paramount for pharmaceutical applications, where impurity profiles must be rigorously characterized and controlled. Furthermore, the method's compatibility with deuterium oxide allows for the facile incorporation of deuterium atoms into the molecular scaffold, a technique increasingly used to improve the metabolic stability and half-life of drug candidates without altering their pharmacological profile.
How to Synthesize Alcohol/Pinacol Derivatives Efficiently
The practical implementation of this electrocatalytic protocol is designed for ease of adoption in both laboratory and pilot-scale settings. The procedure begins with the preparation of a homogeneous mixture containing the carbonyl substrate, the tertiary amine promoter, the supporting electrolyte, and the organic solvent. This mixture is then subjected to constant current electrolysis using platinum plate electrodes. The simplicity of the reagent list—avoiding exotic ligands or sensitive catalysts—makes this method highly robust. Following the reaction, a standard aqueous workup involving extraction and drying is sufficient to isolate the crude product, which is then purified to obtain the target alcohol or pinacol derivative. For detailed operational parameters and specific molar ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Prepare the electrolyte solution by mixing the carbonyl substrate, tertiary amine (DABCO), electrolyte (nBu4NBF4), and organic solvent (DMF) in a reaction vessel.
- Insert two platinum plate electrodes into the mixture and apply a constant current (e.g., 10 mA) under room temperature and air atmosphere with stirring.
- After electrolysis, perform standard workup including extraction with ethyl acetate, drying, and purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrocatalytic technology offers profound benefits for procurement managers and supply chain leaders seeking to optimize cost structures and mitigate risk. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of precious metals and the specialized scavenging resins required for their removal. This simplification of the bill of materials directly contributes to cost reduction in fine chemical manufacturing. Additionally, the operation at room temperature and atmospheric pressure drastically reduces energy consumption compared to thermal processes that require sustained heating or pressurization. The use of electricity as the primary reagent also decouples production from the volatility of chemical reagent markets, providing a more stable and predictable cost base for long-term manufacturing contracts.
- Cost Reduction in Manufacturing: The economic advantage of this method is driven by the removal of expensive catalytic systems and the reduction of waste disposal costs. Traditional methods often generate large volumes of heavy metal-contaminated waste, which requires specialized and costly treatment protocols. By shifting to a metal-free electrochemical process, manufacturers can significantly lower their environmental compliance costs and avoid the logistical complexities of handling hazardous waste. Furthermore, the high atom economy and selectivity of the reaction minimize the loss of valuable starting materials, ensuring that a greater proportion of input costs are converted into sellable product. This efficiency gain is critical for maintaining healthy margins in the competitive landscape of API intermediate production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals such as DABCO and common electrolytes, which are readily available from multiple global suppliers. Unlike specialized chiral ligands or air-sensitive catalysts that may have long lead times or single-source dependencies, the reagents for this electrochemical process are stable and easy to store. This availability reduces the risk of production stoppages due to raw material shortages. Moreover, the mild reaction conditions allow for the use of simpler, less specialized reactor vessels, expanding the pool of potential contract manufacturing organizations (CMOs) capable of executing the process. This flexibility is invaluable for securing supply continuity in the face of geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: Scaling electrochemical processes is increasingly feasible with modern flow chemistry technologies, which offer superior mass and heat transfer compared to batch reactors. This method is well-suited for translation from gram-scale discovery to multi-ton commercial production, facilitating the commercial scale-up of complex organic intermediates. From an environmental standpoint, the process aligns perfectly with green chemistry principles by minimizing waste generation and energy usage. The absence of toxic metal residues simplifies the regulatory approval process for new drug filings, as the burden of proving low metal limits is removed. This environmental stewardship not only meets current regulatory standards but also future-proofs the manufacturing process against tightening global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrocatalytic technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this electrochemical method require transition metal catalysts?
A: No, the method described in patent CN114182272B operates without transition metal catalysts. It utilizes a tertiary amine (DABCO) as an anode sacrificial agent and platinum electrodes, effectively eliminating the risk of heavy metal contamination in the final product.
Q: What are the reaction conditions for this synthesis?
A: The reaction proceeds under mild conditions, specifically at room temperature and atmospheric pressure (air atmosphere). It uses a constant current electrolysis setup, avoiding the need for high-temperature or high-pressure equipment typically required in traditional hydrogenation.
Q: Can this method be used for deuterated compound synthesis?
A: Yes, the patent demonstrates that by introducing deuterium oxide (D2O) into the reaction system, deuterated alcohol derivatives can be successfully synthesized with high deuteration rates, offering a valuable pathway for metabolic stability studies in drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alcohol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrocatalytic synthesis in delivering high-value chemical building blocks. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114182272B can be successfully translated into robust industrial processes. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of alcohol or pinacol derivative meets the exacting standards required by the global pharmaceutical industry. Our infrastructure is designed to support both small-scale custom synthesis for drug discovery and large-scale GMP manufacturing for commercial launch.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our electrocatalytic capabilities can enhance your supply chain efficiency and product quality. Let us be your partner in navigating the complexities of modern chemical synthesis.
