Scaling Linagliptin Production: A Breakthrough in Mixed Anhydride Coupling Technology
The pharmaceutical industry is constantly seeking robust synthetic pathways that balance high purity with industrial scalability, particularly for complex antidiabetic agents like DPP-IV inhibitors. Patent CN114057751B introduces a transformative preparation method for the key intermediate of Linagliptin, specifically targeting the synthesis of tert-butyl (R)-4-((R)-8-methyl-3-(trifluoromethyl)-5,6-dihydroimidazo[1,5-a]pyrazine-7(8H)-yl)-4-oxo-1-(2,4,5-trifluorophenyl)butan-2-ylcarbamate. This innovation addresses critical bottlenecks in the existing manufacturing landscape by replacing hazardous and expensive coupling reagents with a cost-effective mixed anhydride activation strategy. The technical breakthrough lies in the utilization of trimethylacetyl chloride and N-methylmorpholine to activate the carboxylic acid precursor, thereby facilitating a highly efficient amide bond formation with the imidazopyrazine core. This approach not only resolves longstanding issues regarding genotoxic impurity profiles but also dramatically enhances the overall process yield, making it an ideal candidate for reliable API intermediate supplier networks aiming for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical chiral intermediate has relied heavily on carbodiimide-mediated coupling protocols, such as those utilizing EDCI (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide) and HOBt (1-hydroxybenzotriazole). As illustrated in the prior art, these conventional methods suffer from severe drawbacks that hinder large-scale production efficiency and product safety. The use of HOBt is particularly problematic as it can react with solvents like dichloromethane to generate disubstituted coupling impurities, which are notoriously difficult to remove and significantly degrade the final product quality. Furthermore, the degradation products of these condensing agents introduce a substantial nitrogen load into the waste stream, creating significant environmental compliance burdens. Most critically, the presence of potential genotoxic impurities associated with EDCI and HOBt residues necessitates rigorous and costly purification steps to meet stringent regulatory limits, often resulting in a dismal overall yield of approximately 26% due to the loss of material during extensive cleanup and chiral resolution processes.
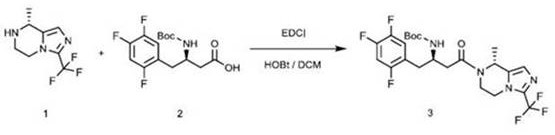
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent employs a mixed anhydride activation technique that fundamentally redesigns the coupling step for superior performance. By activating the Boc-protected amino acid with pivaloyl chloride in the presence of an organic base like N-methylmorpholine, the process generates a highly reactive intermediate that couples efficiently with the amine component without the need for hazardous additives. This strategic shift eliminates the risk of genotoxic impurities at the source, ensuring a cleaner reaction profile that simplifies downstream processing. The new route achieves exceptional purity levels exceeding 99% and boosts yields to over 90%, representing a massive leap forward in manufacturing efficiency. This method not only utilizes cheap and environmentally friendly raw materials but also solves various problems of product quality, safety, and cost, making it completely suitable for industrialization and market requirements for high-purity pharmaceutical intermediates.
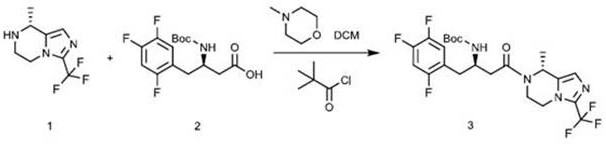
Mechanistic Insights into Mixed Anhydride Activation
The core of this technological advancement lies in the precise mechanistic control of the amide bond formation through mixed anhydride intermediates. In this process, the carboxylic acid group of the starting material is first deprotonated by the organic base to form a carboxylate salt, which then undergoes nucleophilic attack on the carbonyl carbon of trimethylacetyl chloride. This reaction generates a sterically hindered mixed anhydride that is sufficiently electrophilic to react with the amine nucleophile but stable enough to minimize racemization of the chiral center. The use of N-methylmorpholine is crucial here, as it acts as a non-nucleophilic base that scavenges the generated HCl without interfering with the activation step. The subsequent addition of the imidazopyrazine amine leads to a rapid acylation, driven by the release of the stable pivalate leaving group. This mechanism avoids the formation of stable urea byproducts typical of carbodiimide chemistry, thereby preventing the accumulation of hard-to-remove impurities that plague conventional syntheses.
Furthermore, the impurity control mechanism is inherently built into the choice of reagents and reaction conditions. By avoiding HOBt, the process prevents the formation of benzotriazole-derived side products that often co-elute with the desired compound during chromatography. The reaction conditions, specifically maintaining temperatures between -5°C and 15°C during activation and allowing a natural warm-up to 25°C for coupling, ensure kinetic control over the reaction pathway. This thermal profile minimizes epimerization at the alpha-carbon, preserving the stereochemical integrity essential for the biological activity of the final DPP-IV inhibitor. The workup procedure, involving sequential washes with sodium bicarbonate and ammonium chloride, effectively removes unreacted acids and amine salts, while the final slurry in n-heptane and ethyl acetate serves as a powerful crystallization-induced purification step that locks in the high purity specifications required for pharmaceutical applications.
How to Synthesize Linagliptin Intermediate Efficiently
The synthesis of this complex chiral building block requires strict adherence to the optimized protocol to ensure maximum yield and stereochemical purity. The process begins with the dissolution of the protected amino acid in dichloromethane, followed by cooling and the controlled addition of base and activator to form the mixed anhydride in situ. Once activated, the amine component is introduced under rigorous temperature monitoring to facilitate the coupling reaction over a prolonged period. The detailed standardized synthesis steps, including specific mass ratios, stirring times, and purification parameters, are outlined below to guide process chemists in replicating this high-efficiency route.
- Dissolve the protected amino acid (Compound B) in dichloromethane, cool to 0°C, and add N-methylmorpholine followed by trimethylacetyl chloride to form the activated mixed anhydride.
- Prepare a solution of the amine component (Compound A) in dichloromethane and add it dropwise to the activated mixture while maintaining temperature control between -5°C and 15°C.
- Allow the reaction to warm naturally to 25°C and stir for 16 hours, followed by aqueous workup and purification via slurry in n-heptane and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound strategic advantages that extend beyond mere technical metrics. The elimination of expensive coupling agents like EDCI and HOBt directly translates into a significant reduction in raw material costs, as pivaloyl chloride and N-methylmorpholine are commodity chemicals with stable pricing and abundant availability. This shift reduces the dependency on specialized reagent suppliers and mitigates the risk of supply chain disruptions caused by the shortage of niche coupling additives. Moreover, the drastic improvement in yield means that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold and enhancing the overall economic viability of the manufacturing campaign.
- Cost Reduction in Manufacturing: The replacement of high-cost condensing agents with inexpensive acid chlorides results in substantial cost savings per kilogram of produced intermediate. By removing the need for complex chromatographic purification steps often required to remove urea byproducts from carbodiimide reactions, the process significantly reduces solvent consumption and labor hours associated with downstream processing. This streamlined workflow allows for a more efficient allocation of production resources, driving down the overall manufacturing overhead and enabling more competitive pricing strategies for the final API.
- Enhanced Supply Chain Reliability: The use of common, commercially available reagents ensures a robust and resilient supply chain that is less susceptible to market volatility. Since the process does not rely on proprietary or hard-to-source catalysts, procurement teams can easily qualify multiple vendors for raw materials, thereby securing a continuous supply of critical inputs. The high yield and purity of the process also mean that production schedules are more predictable, reducing the lead time for high-purity pharmaceutical intermediates and ensuring timely delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The simplified workup procedure, which relies on standard aqueous extractions and crystallization rather than column chromatography, makes this process highly scalable from pilot plant to multi-ton commercial production. The reduction in nitrogenous waste and the avoidance of genotoxic reagents align perfectly with modern green chemistry principles, easing the burden on environmental health and safety departments. This compliance advantage facilitates faster regulatory approvals and reduces the costs associated with waste treatment and disposal, further enhancing the sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this mixed anhydride coupling technology. These insights are derived directly from the patent data and are intended to clarify the practical benefits and procedural nuances for stakeholders evaluating this synthesis route for their own production pipelines.
Q: Why is the mixed anhydride method superior to EDCI/HOBt coupling for Linagliptin intermediates?
A: The mixed anhydride method eliminates the use of expensive carbodiimides and hydroxybenzotriazole, which are known to introduce genotoxic impurities and difficult-to-remove byproducts. This new approach utilizes cheap pivaloyl chloride, resulting in significantly higher purity (>99%) and yields compared to the traditional 26% yield routes.
Q: What are the critical process parameters for the activation step?
A: Temperature control is vital during the formation of the mixed anhydride. The reaction should be initiated at 0°C with N-methylmorpholine and trimethylacetyl chloride, ensuring the temperature does not exceed 15°C during addition to prevent side reactions and racemization before warming to 25°C for the coupling phase.
Q: How does this process impact environmental compliance and waste management?
A: By avoiding nitrogen-rich condensing agents like EDCI and HOBt, the process drastically reduces the nitrogen load in wastewater. Furthermore, the simplified workup involving standard aqueous washes and solvent slurry replaces complex chromatographic separations, minimizing organic solvent consumption and hazardous waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has thoroughly evaluated the mixed anhydride route described in CN114057751B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are fully equipped to implement this technology with stringent purity specifications and rigorous QC labs to ensure that every batch meets the highest international standards for DPP-IV inhibitor intermediates.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through this innovative approach. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering capabilities can support your long-term production goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
