Advanced One-Step Synthesis of Alpha Beta Diamino Acid Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex chiral scaffolds, particularly those found in bioactive natural products. Patent CN102153488A introduces a groundbreaking methodology for the synthesis of alpha,beta-diamino acid derivatives, a critical structural motif present in potent antibiotics like bleomycins and viomycin, as well as emerging anticancer agents. This technology represents a paradigm shift from traditional multi-step linear syntheses to a highly convergent, one-pot multicomponent reaction strategy. By leveraging the unique reactivity of metal carbenes generated in situ, this process enables the simultaneous formation of two adjacent chiral centers with exceptional stereocontrol. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition: it drastically simplifies the supply chain for high-value pharmaceutical intermediates while ensuring the high purity required for downstream drug development. The ability to access these complex molecules in a single operational step under mild conditions underscores the potential for significant cost reduction in API manufacturing and accelerates the timeline from laboratory discovery to commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of alpha,beta-diamino acid skeletons has been plagued by synthetic inefficiencies that hinder large-scale production. Traditional routes often involve lengthy sequences of protection and deprotection steps, requiring harsh reaction conditions that can compromise sensitive functional groups. These legacy methods typically suffer from poor atom economy, generating substantial chemical waste and driving up the cost of goods sold. Furthermore, achieving high diastereoselectivity in the formation of adjacent chiral centers has remained a formidable challenge, often necessitating expensive chiral auxiliaries or resolution techniques that cut overall yields by half. The cumulative effect of these drawbacks is a manufacturing process that is not only economically burdensome but also environmentally unsustainable, making it difficult for suppliers to guarantee consistent quality and delivery timelines for complex intermediates needed in oncology and infectious disease therapeutics.
The Novel Approach
In stark contrast, the methodology disclosed in CN102153488A utilizes a sophisticated three-component coupling reaction that merges an imine, an amide, and a diazo compound in a single vessel. This approach capitalizes on the high reactivity of metal-stabilized carbenes to insert directly into the N-H bond of the amide, followed by a stereoselective addition to the imine. The result is a streamlined process that operates at room temperature, eliminating the need for energy-intensive heating or cooling cycles. As illustrated in the reaction scheme below, this one-pot transformation constructs the core carbon-nitrogen framework with remarkable precision, delivering products with yields reaching up to 92% and diastereomeric ratios exceeding 20:1. This leap in efficiency transforms the economic landscape of producing these valuable building blocks, offering a reliable pharmaceutical intermediate supplier a distinct competitive advantage in terms of both speed and cost.
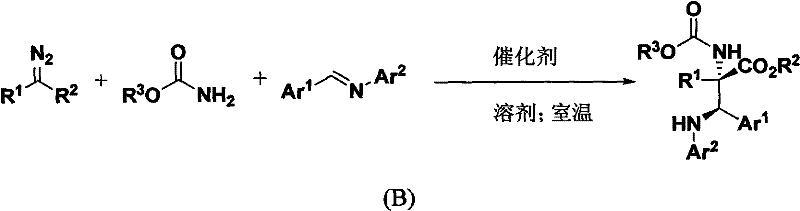
Mechanistic Insights into Rhodium-Catalyzed Multicomponent Coupling
The success of this synthesis hinges on the precise orchestration of a metal-catalyzed catalytic cycle, primarily driven by dirhodium or copper catalysts. The mechanism initiates with the decomposition of the diazo compound by the metal catalyst to form a highly electrophilic metal-carbene species. This reactive intermediate then undergoes nucleophilic attack by the amide nitrogen, generating an ammonium ylide. Crucially, this ylide does not decompose but is instead trapped by the imine substrate in a highly organized transition state. This cascade sequence ensures that the new carbon-carbon and carbon-nitrogen bonds are formed simultaneously with strict stereochemical control, dictated by the steric environment of the catalyst and the substrates. Understanding this mechanistic pathway is vital for process chemists aiming to optimize reaction parameters, as it highlights the importance of maintaining anhydrous conditions and controlled addition rates to prevent carbene dimerization or other side reactions that could lower the overall purity of the final product.
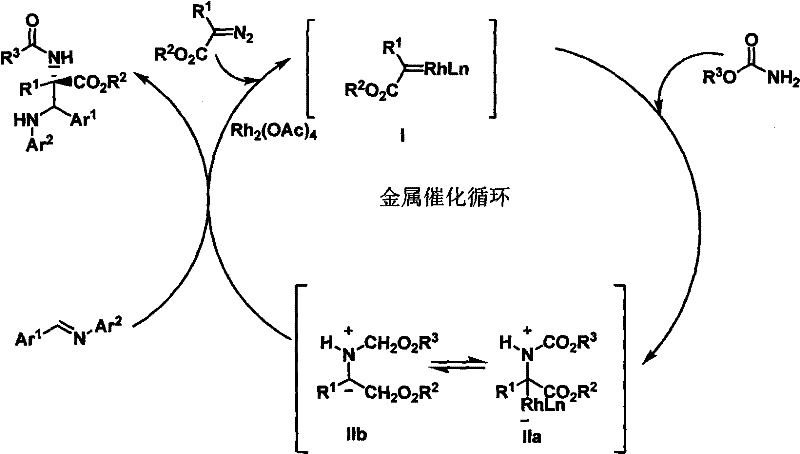
Furthermore, the robustness of this catalytic system allows for a broad substrate scope, accommodating various electron-withdrawing and electron-donating groups on the aromatic rings of the imine and diazo components. This flexibility is essential for medicinal chemists who need to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies. The use of molecular sieves in the reaction mixture plays a pivotal role in scavenging trace water, which could otherwise hydrolyze the sensitive imine or deactivate the metal catalyst. By strictly controlling these mechanistic variables, the process achieves a level of impurity control that minimizes the burden on downstream purification. This results in a cleaner crude profile, reducing the load on chromatographic columns and facilitating a more straightforward isolation of the target alpha,beta-diamino acid derivative, thereby enhancing the overall throughput of the manufacturing operation.
How to Synthesize Alpha,Beta-Diamino Acid Derivatives Efficiently
Implementing this synthesis requires careful attention to the stoichiometry and addition protocols to maximize the formation of the desired diastereomer. The standard procedure involves dissolving the imine and amide substrates along with the catalyst and molecular sieves in a dry organic solvent such as dichloromethane or toluene. The diazo compound, which serves as the carbene precursor, is then added slowly via syringe pump or dropping funnel to maintain a low steady-state concentration, preventing uncontrolled exotherms. Following the completion of the addition, the reaction is allowed to stir briefly to ensure full conversion before the solvent is removed under reduced pressure. The detailed standardized synthesis steps are provided in the guide below for technical reference.
- Dissolve imine, amide, metal Lewis acid catalyst (e.g., Rh2(OAc)4), and molecular sieves in an organic solvent such as dichloromethane or toluene at room temperature.
- Slowly add dropwise a solution of the diazo compound (e.g., phenyl diazoacetate) dissolved in the same solvent over a period of 1 hour to control exotherm and selectivity.
- Stir the reaction mixture for an additional 0.5 hours, remove solvent via rotary evaporation, and purify the crude product using column chromatography with ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that extend beyond simple yield improvements. The consolidation of multiple synthetic steps into a single operation fundamentally alters the cost structure of producing these complex intermediates. By eliminating the need for intermediate isolation, drying, and purification between steps, the process significantly reduces solvent consumption, labor hours, and equipment occupancy time. This streamlining of the workflow leads to substantial cost savings in pharmaceutical intermediate manufacturing, allowing buyers to negotiate more favorable pricing without compromising on quality. Additionally, the mild reaction conditions reduce the safety risks associated with handling hazardous reagents at extreme temperatures, lowering insurance and compliance costs for the manufacturing facility.
- Cost Reduction in Manufacturing: The elimination of transition metal removal steps often required in other catalytic processes, combined with the high atom economy of this multicomponent reaction, drives down the raw material costs per kilogram. The high selectivity means less material is wasted on unwanted isomers, maximizing the utility of every gram of starting material purchased. This efficiency creates a buffer against volatility in raw material prices, ensuring stable long-term supply contracts for key clients.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including aromatic aldehydes, amines, and commercially available diazo esters, are widely sourced from the global chemical market. This abundance of feedstock reduces the risk of supply bottlenecks that often plague specialized synthetic routes. Furthermore, the robustness of the reaction allows for flexible scheduling and rapid scale-up from gram to kilogram quantities, ensuring that urgent orders for clinical trial materials can be fulfilled with short lead times.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving primarily solvent evaporation and standard chromatography, makes this process highly amenable to scale-up in existing multipurpose reactors. The reduction in waste generation aligns with green chemistry principles, helping pharmaceutical companies meet their sustainability goals. This environmental compatibility simplifies regulatory filings and reduces the burden of waste disposal, making the supply chain more resilient to changing environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these alpha,beta-diamino acid derivatives. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this technology into their existing drug development pipelines.
Q: What are the key advantages of this synthesis method compared to traditional routes?
A: This method utilizes a one-pot multicomponent reaction that significantly reduces step count, operates under mild room temperature conditions, and achieves high diastereoselectivity (dr > 20:1) and yields up to 92%, overcoming the low yield and繁琐 post-treatment issues of conventional methods.
Q: What types of catalysts are suitable for this transformation?
A: The process employs metal Lewis acid catalysts, specifically rhodium complexes like rhodium acetate, rhodium propionate, or copper complexes such as tetraacetonitrile cuprous hexafluorophosphate, which effectively generate the reactive metal carbene intermediates.
Q: Does the resulting derivative have biological activity?
A: Yes, the synthesized alpha,beta-diamino acid derivatives exhibit significant anticancer activity, specifically demonstrating inhibitory effects against MDA-MB-468 breast cancer cell lines with IC50 values in the micromolar range.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Diamino Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN102153488A can be successfully translated into industrial reality. We operate stringent purity specifications and utilize rigorous QC labs to verify the identity and stereochemical integrity of every batch, guaranteeing that our alpha,beta-diamino acid derivatives meet the exacting standards required for GMP manufacturing. Our commitment to technical excellence ensures that you receive a product that is ready for immediate use in your synthesis campaigns.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall development costs.
