Advanced Mo-Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
Advanced Mo-Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
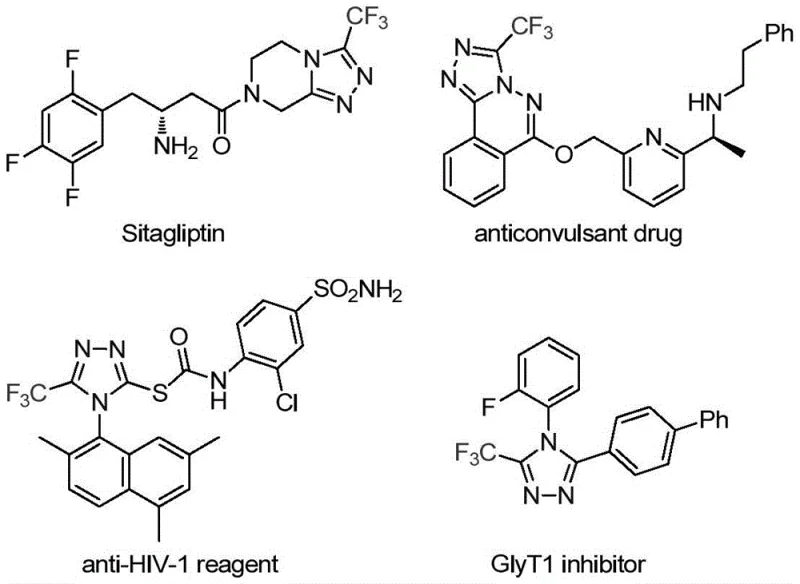
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in Chinese Patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology addresses the growing demand for fluorinated heterocycles, which are ubiquitous in modern medicinal chemistry due to their ability to enhance metabolic stability, lipophilicity, and bioavailability. As illustrated in the structural diversity of known drugs like Sitagliptin, the incorporation of the 1,2,4-triazole motif is a proven strategy for developing potent therapeutics. The disclosed method utilizes a synergistic molybdenum and copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride and functionalized isonitriles, offering a streamlined alternative to legacy synthetic pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that present significant challenges for large-scale manufacturing and process safety. Traditional literature reports predominantly describe the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These routes often suffer from the requirement of harsh reaction conditions, limited substrate scope, and the use of unstable or hazardous reagents. Furthermore, copper-catalyzed multi-component reactions involving diazonium salts and trifluorodiazoethane, while effective in academic settings, introduce severe safety risks associated with the handling of explosive diazo species. The reliance on such dangerous intermediates complicates regulatory compliance and increases the cost of goods due to the need for specialized containment and safety protocols, making these conventional methods less attractive for reliable pharmaceutical intermediate suppliers aiming for cost-effective production.
The Novel Approach
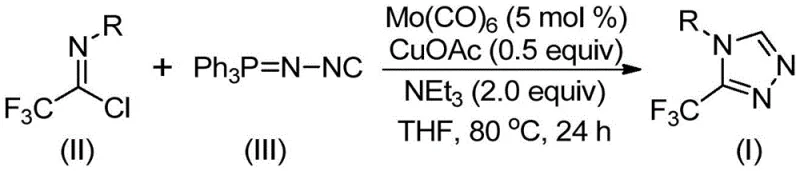
In stark contrast to these legacy techniques, the invention described in CN113307778A introduces a remarkably mild and operationally simple protocol. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as starting materials, the process avoids the generation of hazardous diazonium intermediates entirely. The reaction proceeds efficiently in common organic solvents such as tetrahydrofuran (THF) at moderate temperatures ranging from 70°C to 90°C. This shift in reactivity profile allows for the direct construction of the 3,4-disubstituted 1,2,4-triazole core through a [3+2] cycloaddition mechanism promoted by the dual metal system. The general reaction scheme highlights the elegance of this transformation, where complex molecular architectures are assembled from simple precursors with high atom economy and minimal waste generation.
Mechanistic Insights into Mo-Cu Co-Catalyzed Cycloaddition
The success of this synthetic methodology lies in the intricate interplay between the molybdenum and copper catalysts, which orchestrate the formation of the triazole ring through a sophisticated mechanistic pathway. According to the patent disclosure, molybdenum hexacarbonyl acts as a crucial metal activator that coordinates with the functionalized isonitrile to form a reactive metal complex. This activation lowers the energy barrier for the subsequent nucleophilic attack. Simultaneously, the cuprous acetate catalyst promotes the [3+2] cycloaddition of the activated isonitrile species to the trifluoroethylimidoyl chloride. This cooperative catalysis facilitates the formation of a five-membered ring intermediate, which is the precursor to the final triazole product. The presence of water in the system, potentially generated in situ or added, plays a vital role in the final step by facilitating the removal of the triphenylphosphine oxide byproduct, thereby driving the equilibrium towards the desired 3-trifluoromethyl-substituted 1,2,4-triazole compound.
From an impurity control perspective, this mechanism offers distinct advantages over acid-mediated or high-temperature thermal cyclizations. The mild reaction temperature of 70-90°C minimizes the risk of thermal decomposition of sensitive functional groups on the aromatic rings, such as nitro or halo substituents. Furthermore, the specificity of the metal-ligand interactions ensures high regioselectivity, predominantly yielding the 3,4-disubstituted isomer rather than a mixture of regioisomers which would complicate downstream purification. The use of triethylamine as a base further aids in scavenging acidic byproducts, maintaining a neutral to slightly basic environment that preserves the integrity of the trifluoromethyl group and prevents defluorination side reactions, ensuring a clean impurity profile suitable for pharmaceutical applications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this technology involves a straightforward procedure that is highly amenable to standard laboratory and pilot plant equipment. The process begins with the precise weighing of molybdenum hexacarbonyl, cuprous acetate, triethylamine, and molecular sieves, which are suspended in an anhydrous organic solvent. To this mixture, the key electrophile, trifluoroethylimidoyl chloride, and the nucleophile, functionalized isonitrile, are added. The reaction is typically conducted under an inert atmosphere to prevent oxidation of the metal catalysts. Following the reaction period, the workup involves simple filtration to remove solid residues and metal salts, followed by standard chromatographic purification. For a comprehensive understanding of the specific stoichiometric ratios and optimization parameters required for different substrates, please refer to the detailed standardized synthesis steps provided below.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Mo-Cu co-catalyzed synthesis represents a strategic opportunity to optimize the sourcing of high-value heterocyclic intermediates. The transition from hazardous diazonium-based chemistries to this stable imidoyl chloride route fundamentally alters the risk profile of the supply chain, reducing the need for expensive safety infrastructure and specialized handling procedures. This shift not only enhances operational safety but also streamlines the logistics of raw material transportation and storage, as the starting materials are commercially available and stable solids or liquids that do not require cryogenic shipping or explosive hazard classifications.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and abundant catalysts such as cuprous acetate and molybdenum hexacarbonyl, which are used in low catalytic loadings. Unlike precious metal catalysts like palladium or rhodium that often require complex recovery systems to meet residual metal specifications, copper and molybdenum residues are easier to manage and remove, leading to substantial cost savings in downstream processing. Additionally, the high reaction efficiency and yield reported across a broad range of substrates minimize the loss of valuable starting materials, directly contributing to a lower cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining supply continuity in the pharmaceutical sector. The tolerance of the method towards various functional groups means that a single platform technology can be applied to synthesize a diverse library of triazole derivatives, reducing the need for multiple dedicated production lines. This flexibility allows suppliers to respond rapidly to changing market demands for different analogues without significant retooling or process redevelopment, thereby securing a more resilient supply chain for customers.
- Scalability and Environmental Compliance: The patent explicitly notes that the reaction can be expanded to gram-level scales with ease, indicating strong potential for kilogram and tonne-scale manufacturing. The use of THF, a common and recyclable solvent, aligns with green chemistry principles and simplifies solvent recovery operations. Furthermore, the avoidance of toxic hydrazines and explosive diazo compounds significantly reduces the environmental footprint of the manufacturing process, easing the burden on waste treatment facilities and ensuring compliance with increasingly stringent environmental regulations regarding hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this technology for commercial production. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of the Mo-Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods requiring hazardous diazonium salts or harsh hydrazinolysis conditions, this patent describes a mild protocol (70-90°C) using readily available trifluoroethylimidoyl chloride and functionalized isonitriles, significantly improving safety and operational simplicity.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly demonstrates that the reaction can be expanded to gram-level scales with high efficiency. The use of common solvents like THF and standard purification techniques like column chromatography supports feasible commercial scale-up.
Q: What is the substrate scope for the R group in this triazole synthesis?
A: The method exhibits broad substrate tolerance, successfully accommodating phenethyl groups as well as substituted or unsubstituted aryl groups with various functionalities including methyl, methoxy, fluoro, chloro, and nitro substituents at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the Mo-Cu co-catalyzed pathway described in CN113307778A and possesses the expertise to translate this laboratory-scale innovation into robust commercial processes. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from discovery to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data for our available triazole derivatives and to discuss route feasibility assessments that can accelerate your timeline to clinical trials and commercial launch.
