Scalable Synthesis of Novel Furan Derivatives for Next-Generation Antibacterial Drug Development
The pharmaceutical industry is constantly seeking novel mechanisms to combat antibiotic resistance, and patent CN102603683A presents a significant breakthrough in the field of anti-AI-2 quorum sensing drugs. This technical insight report analyzes the disclosed furan compounds and their preparation methods, highlighting their potential as next-generation antibacterial agents. The patent details a series of novel furan derivatives designed through computer-aided drug design targeting the LuxPQ receptor protein, offering a strategic alternative to traditional bactericidal approaches. By interfering with bacterial communication rather than killing bacteria directly, these compounds mitigate the risk of resistance development, a critical concern for R&D directors globally. The synthesis route described leverages accessible starting materials and mild conditions, positioning these molecules as viable candidates for both research and commercial production. As a reliable pharmaceutical intermediates supplier, understanding the nuances of this chemistry is essential for integrating such innovative scaffolds into broader drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for complex heterocyclic compounds often suffer from severe limitations that hinder their adoption in large-scale manufacturing environments. Conventional methods frequently rely on harsh reaction conditions, including extreme temperatures and pressures, which necessitate specialized equipment and increase operational risks. Furthermore, many existing routes utilize expensive transition metal catalysts that require rigorous removal steps to meet stringent purity specifications for pharmaceutical applications. The presence of residual metals can complicate regulatory approval processes and significantly inflate production costs due to the need for additional purification stages like chromatography or specialized scavenging resins. Additionally, conventional approaches often involve multiple protection and deprotection steps, leading to lower overall yields and increased waste generation. These inefficiencies create bottlenecks in the supply chain, extending lead times and reducing the economic feasibility of producing high-purity API intermediates. For procurement managers, these factors translate into higher raw material costs and less predictable supply continuity, making such processes less attractive for commercial ventures.
The Novel Approach
In contrast, the novel approach outlined in patent CN102603683A offers a streamlined and efficient pathway that addresses many of the drawbacks associated with traditional synthesis. This method utilizes a diazotization coupling strategy followed by reductive amination or acylation, which proceeds under mild reaction conditions typically around room temperature or slightly elevated heat. The use of readily available raw materials such as substituted anilines and furfural derivatives ensures a stable and cost-effective supply chain foundation. Crucially, the process avoids the use of precious metal catalysts, relying instead on cuprous chloride which is more economical and easier to manage in a production setting. The post-treatment procedures are simplified to standard filtration and recrystallization steps, eliminating the need for complex chromatographic separations in many instances. This simplicity not only reduces the environmental footprint by minimizing solvent usage and waste but also enhances the scalability of the process. For supply chain heads, this translates to cost reduction in antibacterial drug manufacturing and a more robust production capability that can adapt to fluctuating market demands without compromising quality.
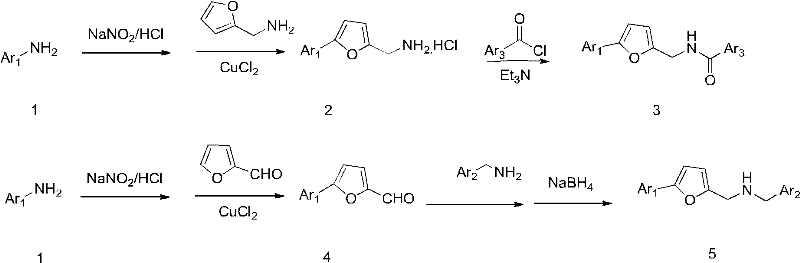
Mechanistic Insights into Diazotization Coupling and Furan Functionalization
The core of this synthetic strategy lies in the efficient construction of the aryl-furan bond through a diazotization-coupling mechanism. The process begins with the conversion of substituted anilines into diazonium salts under acidic conditions using sodium nitrite at controlled low temperatures to ensure stability. This reactive intermediate then undergoes a coupling reaction with furfural or furfurylamine in the presence of cuprous chloride, facilitating the formation of the critical carbon-carbon or carbon-nitrogen linkage. This step is pivotal as it establishes the core scaffold of the target molecule with high regioselectivity. The reaction conditions are optimized to minimize side reactions, ensuring that the resulting phenylfuran intermediates are formed with high purity. Understanding this mechanism is vital for R&D teams aiming to replicate or modify the synthesis for analog development. The robustness of this coupling reaction allows for a wide range of substituents on the aromatic ring, providing flexibility in tuning the physicochemical properties of the final drug candidate. This mechanistic clarity supports the commercial scale-up of complex quorum sensing inhibitors by providing a predictable and reproducible chemical transformation.
Following the formation of the core scaffold, the synthesis diverges into two main pathways to generate the final target compounds of Formula I and Formula II. For Formula I, the intermediate phenylfuran methylamine hydrochloride undergoes acylation with aromatic formyl chlorides in the presence of a base like triethylamine. This amidation reaction is highly efficient and proceeds smoothly at low temperatures to prevent degradation of sensitive functional groups. For Formula II, the phenylfuran carboxaldehyde intermediate reacts with substituted benzylamines to form a Schiff base, which is subsequently reduced using sodium borohydride. This reductive amination step is crucial for introducing the secondary amine functionality while maintaining the integrity of the furan ring. Impurity control is managed through careful stoichiometry and temperature regulation during these steps, ensuring that by-products are minimized. The final products are isolated as hydrochloride salts or free bases, depending on the desired formulation, with purity levels suitable for biological evaluation. This level of control over impurity profiles is essential for meeting the rigorous standards required for high-purity furan compounds in pharmaceutical development.

How to Synthesize N-(5-aryl-2-furylmethyl) Derivatives Efficiently
The practical execution of this synthesis requires precise adherence to the reaction parameters defined in the patent to achieve optimal yields and purity. The process is designed to be operationally simple, making it accessible for laboratories equipped with standard glassware and cooling capabilities. Key to the success of this route is the management of the diazonium salt formation, which must be kept at low temperatures to prevent decomposition. Subsequent coupling steps benefit from the use of aqueous cuprous chloride solutions, which facilitate the reaction without requiring anhydrous conditions initially. The workup procedures involve neutralization and extraction, followed by recrystallization from solvents like methanol or ethanol to obtain the final solid products. Detailed standardized synthesis steps are critical for ensuring batch-to-batch consistency, especially when transitioning from gram-scale laboratory experiments to kilogram-scale production. This structured approach ensures that technical teams can reliably reproduce the results reported in the patent literature.
- Prepare substituted aniline diazonium salts under acidic conditions using sodium nitrite at low temperatures.
- Perform cuprous chloride catalyzed coupling with furfural or furfurylamine to generate key phenylfuran intermediates.
- Complete the synthesis via nucleophilic addition followed by sodium borohydride reduction or acylation to yield target furan compounds.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement and supply chain management teams looking to optimize their sourcing strategies. The reliance on commodity chemicals such as anilines and furfural means that raw material availability is high, reducing the risk of supply disruptions caused by scarce reagents. The elimination of expensive catalysts and complex purification steps directly contributes to cost reduction in antibacterial drug manufacturing, allowing for more competitive pricing models. Furthermore, the mild reaction conditions reduce energy consumption and equipment wear, leading to lower operational expenditures over the lifecycle of the product. These factors combined create a compelling economic case for integrating these furan derivatives into existing product portfolios. For organizations focused on efficiency, this process represents a significant opportunity to enhance margin performance while maintaining high quality standards.
- Cost Reduction in Manufacturing: The process eliminates the need for precious metal catalysts and complex chromatographic purification, which are major cost drivers in traditional pharmaceutical synthesis. By utilizing inexpensive reagents like cuprous chloride and standard solvents, the overall material cost is significantly lowered. Additionally, the high yields reported in specific examples demonstrate efficient atom economy, reducing waste disposal costs. This qualitative improvement in process efficiency translates to substantial cost savings without the need for capital-intensive equipment upgrades. Procurement teams can leverage these efficiencies to negotiate better terms with suppliers or reinvest savings into further R&D initiatives.
- Enhanced Supply Chain Reliability: The use of widely available starting materials ensures that the supply chain remains resilient against market fluctuations. Unlike specialized reagents that may have long lead times or single-source dependencies, the inputs for this synthesis are produced by multiple global manufacturers. This diversity in sourcing options reduces the risk of bottlenecks and ensures continuous production capability. Moreover, the simplicity of the synthesis allows for flexible manufacturing arrangements, enabling rapid scaling in response to demand spikes. Reducing lead time for high-purity API intermediates becomes achievable when the underlying chemistry is robust and原料 supply is secure, providing a strategic advantage in fast-moving therapeutic areas.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from laboratory benchtops to industrial reactors without fundamental changes to the chemistry. The absence of hazardous reagents and the generation of manageable waste streams align with modern environmental compliance standards. Simplified workup procedures mean less solvent consumption and lower energy requirements for distillation or drying. This environmental friendliness not only reduces regulatory burdens but also enhances the corporate sustainability profile. For supply chain heads, this means easier permitting processes and fewer interruptions due to environmental audits, ensuring a steady flow of materials to downstream customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these furan compounds. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this technology into their operations. The information covers aspects ranging from reaction mechanics to potential applications in drug development. This section serves as a quick reference guide for technical procurement teams evaluating the feasibility of this synthesis route.
Q: What is the primary advantage of this furan synthesis route over conventional methods?
A: The patented route utilizes mild reaction conditions and readily available raw materials like substituted anilines and furfural, significantly simplifying post-treatment and reducing the need for harsh reagents compared to traditional multi-step heterocycle constructions.
Q: Are these furan compounds suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process avoids expensive transition metal catalysts and uses standard unit operations like crystallization and filtration, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates without specialized equipment.
Q: What is the biological application of these synthesized furan derivatives?
A: These compounds function as AI-2 quorum sensing inhibitors, targeting the LuxPQ receptor protein to disrupt bacterial communication and biofilm formation, offering a novel mechanism for developing broad-spectrum antibacterial drugs with reduced resistance risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furan Compounds Supplier
NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of experts specializes in translating complex laboratory syntheses into robust industrial processes, ensuring that the potential of innovations like patent CN102603683A is fully realized. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards. Our commitment to quality and reliability makes us a trusted partner for pharmaceutical companies seeking to bring novel antibacterial agents to market. By leveraging our infrastructure, you can accelerate your timeline from discovery to commercialization with confidence.
We invite you to contact our technical procurement team to discuss how we can assist with your specific requirements. Request a Customized Cost-Saving Analysis to understand how optimizing this synthesis route can benefit your bottom line. Our team is prepared to provide specific COA data and route feasibility assessments tailored to your project needs. Let us help you navigate the complexities of chemical manufacturing and secure a reliable supply of high-quality intermediates for your next breakthrough therapy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
