Scalable Palladium-Catalyzed Vinyl Substitution for High-Purity Pyridine Intermediates
Scalable Palladium-Catalyzed Vinyl Substitution for High-Purity Pyridine Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes for complex heterocyclic intermediates, and patent CN1126467A presents a significant advancement in this domain. This intellectual property discloses an improved method for preparing substituted pyridine derivatives, specifically through a palladium-catalyzed vinyl substitution of 2-substituted pyridines. The technology addresses critical bottlenecks in the synthesis of key intermediates used for treating various diseases, including psoriasis. By leveraging organo-metallic catalysis, this process enables the efficient construction of carbon-carbon bonds at the pyridine 2-position, a transformation that is historically challenging due to the electronic deactivation of the pyridine ring. The strategic implementation of this patented methodology allows for the production of high-purity compounds that meet the stringent requirements of modern drug development pipelines.
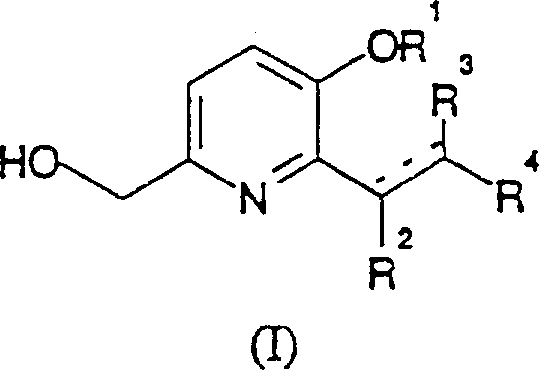
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for functionalizing pyridine rings often suffer from severe limitations that hinder large-scale commercial application. Conventional methods frequently rely on harsh reaction conditions, such as extremely high temperatures or the use of stoichiometric amounts of toxic reagents, which complicate waste management and increase operational costs. Furthermore, older methodologies often exhibit poor regioselectivity, leading to complex mixtures of isomers that require extensive and yield-losing purification steps. The inability to effectively control impurity profiles in these legacy processes poses a significant risk to pharmaceutical manufacturers, as trace impurities can impact drug safety and regulatory approval. Additionally, many conventional routes are not amenable to scale-up, failing to maintain yield and purity when transitioning from laboratory gram-scale to industrial kilogram or ton-scale production.
The Novel Approach
The novel approach detailed in patent CN1126467A overcomes these historical challenges by utilizing a sophisticated palladium-catalyzed coupling mechanism. This method employs specific organo-metallic catalysts, such as palladium acetate combined with phosphine ligands, to facilitate the substitution reaction under relatively milder conditions. The use of a leaving group at the 2-position of the pyridine ring, such as bromine or iodine, activates the substrate for efficient coupling with vinyl compounds like acrylates. This strategy significantly enhances the reaction rate and selectivity, ensuring that the desired vinyl-substituted product is formed with minimal byproduct generation. The process is designed to be robust in polar aprotic solvents like DMF, allowing for better solubility of reagents and improved heat transfer, which are critical factors for safe and efficient large-scale manufacturing operations.
Mechanistic Insights into Palladium-Catalyzed Vinyl Substitution
The core of this technology lies in the intricate catalytic cycle driven by the palladium complex. The mechanism initiates with the oxidative addition of the palladium catalyst into the carbon-halogen bond of the 2-substituted pyridine substrate, forming a reactive organopalladium intermediate. This step is crucial as it activates the otherwise inert pyridine ring towards nucleophilic attack or insertion. Subsequently, the vinyl compound coordinates to the palladium center and undergoes migratory insertion, forming a new carbon-carbon bond. The cycle is completed by a beta-hydride elimination step, which releases the final vinyl-substituted pyridine product and regenerates the active palladium species for the next catalytic turnover. Understanding this cycle is vital for R&D directors, as it highlights the importance of ligand selection in stabilizing the palladium center and preventing catalyst deactivation through the formation of palladium black.
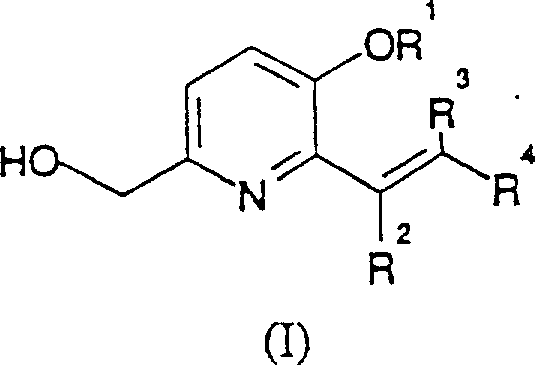
Impurity control is another critical aspect managed by this mechanistic pathway. The specific choice of reaction conditions, including temperature and solvent composition, minimizes side reactions such as homocoupling of the vinyl species or dehalogenation of the pyridine ring. The patent emphasizes the use of aqueous DMF mixtures, which can help solubilize inorganic bases required for the reaction while maintaining the organic solubility of the substrates. This balance prevents the accumulation of insoluble salts that could trap product or catalyze decomposition pathways. Furthermore, the ability to convert the resulting Formula (I) compounds into salts or N-oxides provides an additional purification handle, allowing manufacturers to remove trace metal residues and organic impurities through crystallization. This level of control over the chemical environment ensures that the final intermediate meets the rigorous purity specifications demanded by global regulatory bodies.
How to Synthesize Substituted Pyridine Derivatives Efficiently
Executing this synthesis requires precise adherence to the patented parameters to ensure optimal yield and safety. The process begins with the preparation of the 2-halo-pyridine substrate, which must be of high purity to prevent catalyst poisoning. The reaction is typically conducted under an inert nitrogen atmosphere to protect the sensitive palladium catalyst from oxidation. Reagents are mixed in a specific order, often adding the base and vinyl compound to the substrate solution before heating.
- Prepare the 2-substituted pyridine substrate (Formula II) with a suitable leaving group such as bromine or iodine at the 2-position.
- Mix the substrate with a vinyl compound (Formula III) such as an acrylate in a polar aprotic solvent like DMF with water.
- Add a palladium catalyst system, such as Pd(OAc)2 with triphenylphosphine, and heat the mixture to 120°C under nitrogen to facilitate coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers substantial strategic advantages beyond mere technical feasibility. The streamlined nature of the palladium-catalyzed route eliminates multiple synthetic steps that were previously required to install the vinyl functionality, directly translating to reduced raw material consumption and lower overall processing time. By simplifying the synthetic sequence, manufacturers can significantly reduce the inventory of hazardous intermediates that need to be stored and handled, thereby lowering insurance and compliance costs. The robustness of the reaction in standard industrial solvents like DMF means that existing manufacturing infrastructure can often be utilized without requiring expensive retrofits or specialized equipment, facilitating a faster time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The elimination of stoichiometric heavy metal reagents in favor of a catalytic system drastically reduces the cost of goods sold. Since the palladium catalyst is used in sub-stoichiometric amounts and can potentially be recovered or recycled, the financial burden of precious metal consumption is minimized. Furthermore, the high selectivity of the reaction reduces the need for costly chromatographic purification steps, allowing for more economical crystallization-based isolation methods. This efficiency gain ensures that the production of these complex pharmaceutical intermediates remains economically viable even when raw material prices fluctuate, providing a stable cost structure for long-term supply agreements.
- Enhanced Supply Chain Reliability: The scalability of this process ensures a consistent and reliable supply of critical intermediates, mitigating the risk of production bottlenecks. The use of commercially available starting materials, such as substituted pyridones and acrylates, means that the supply chain is not dependent on obscure or single-source reagents. This accessibility allows for the qualification of multiple suppliers for raw materials, enhancing supply security. Additionally, the reaction's tolerance to slight variations in conditions makes it robust against minor operational deviations, ensuring that batch-to-batch consistency is maintained, which is crucial for maintaining regulatory compliance and avoiding costly production delays.
- Scalability and Environmental Compliance: From an environmental perspective, this method offers a greener alternative to traditional synthesis routes by reducing waste generation. The high atom economy of the coupling reaction means that a larger proportion of the starting materials end up in the final product, minimizing the volume of chemical waste that requires treatment. The ability to perform the reaction in aqueous solvent mixtures reduces the reliance on purely organic solvents, simplifying solvent recovery and distillation processes. This alignment with green chemistry principles not only reduces environmental impact but also helps manufacturers meet increasingly stringent global environmental regulations, avoiding potential fines and reputational damage associated with non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What catalysts are preferred for this vinyl substitution reaction?
A: The patent specifies palladium (II) catalysts such as Pd(OAc)2, often used in combination with phosphine ligands like triphenylphosphine or tris(2-furyl)phosphine to enhance activity and stability.
Q: What are the optimal reaction conditions for scalability?
A: The reaction is preferably carried out in DMF or aqueous DMF at elevated temperatures ranging from 80°C to 160°C, with 120°C being the preferred setpoint for balancing rate and selectivity.
Q: Can this method produce pharmaceutically acceptable salts?
A: Yes, the resulting Formula (I) compounds can be converted into pharmaceutically useful salts, such as hydrochlorides or fumarates, using standard acid addition techniques described in the examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Pyridine Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent technologies like CN1126467A into commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to plant. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications for every batch of substituted pyridine derivatives we produce. We understand that the consistency of pharmaceutical intermediates is non-negotiable, and our quality management systems are designed to guarantee that every molecule meets the exacting standards required for global drug registration.
We invite you to collaborate with us to optimize your supply chain for these critical materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower your R&D and procurement teams to make informed decisions. Contact us today to discuss how we can support your development of psoriasis treatments and other therapeutic areas requiring high-quality pyridine intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
