Scalable Palladium-Catalyzed Synthesis of Substituted Pyridine Derivatives for Commercial Production
The pharmaceutical industry continuously seeks robust synthetic routes for complex intermediates, particularly those serving as critical building blocks for therapeutic agents treating chronic conditions like psoriasis. Patent CN1046709C introduces a transformative approach to the preparation of substituted pyridine derivatives, specifically addressing the limitations of prior art methods that struggled with scalability and efficiency. This innovation leverages advanced organometallic chemistry to establish a reliable pathway for generating high-value compounds that were previously difficult to manufacture in bulk quantities. By focusing on palladium-catalyzed vinyl substitution reactions, the technology provides a foundation for producing intermediates with superior purity profiles and consistent quality. For global procurement teams, understanding the technical nuances of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent regulatory demands. The shift towards this catalytic methodology represents a significant leap forward in process chemistry, enabling manufacturers to overcome historical bottlenecks associated with traditional pyridine functionalization strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted pyridine derivatives relied on methodologies that were inherently unsuitable for large-scale production applications due to their complexity and poor yield profiles. Conventional routes often involved multiple protection and deprotection steps, which not only increased the overall processing time but also introduced significant opportunities for impurity generation and material loss. These traditional methods frequently required harsh reaction conditions that compromised the structural integrity of sensitive functional groups, leading to inconsistent batch-to-batch quality that is unacceptable for pharmaceutical grade materials. Furthermore, the reliance on stoichiometric reagents rather than catalytic systems resulted in excessive waste generation and higher raw material costs, creating substantial economic inefficiencies for manufacturers. The inability to effectively scale these processes meant that supply chains remained fragile, with production capacities often unable to meet the growing global demand for psoriasis treatments. Consequently, the industry faced persistent challenges in securing cost reduction in pharmaceutical intermediates manufacturing without compromising on the critical quality attributes required for downstream drug synthesis.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a palladium-catalyzed coupling reaction that streamlines the synthetic route and dramatically enhances operational efficiency for commercial production. By employing organometallic catalysts, the process facilitates the direct substitution of vinyl groups onto the pyridine core under relatively mild conditions, thereby preserving the integrity of other sensitive substituents present on the molecule. This methodology allows for the use of commercially available starting materials, such as halopyridines and acrylates, which simplifies the supply chain and reduces dependency on exotic or hard-to-source reagents. The reaction proceeds effectively in polar solvents like DMF, often with the addition of water, which aids in solubility and heat transfer during the exothermic coupling phases. This flexibility in solvent selection and reaction parameters enables manufacturers to optimize the process for maximum throughput while maintaining rigorous control over impurity levels. Ultimately, this innovative strategy supports the commercial scale-up of complex pharmaceutical intermediates by providing a robust, reproducible, and economically viable manufacturing platform.
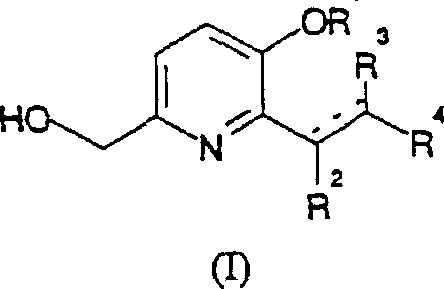
Mechanistic Insights into Palladium-Catalyzed Vinyl Substitution
The core of this technological advancement lies in the intricate mechanistic pathway of the palladium-catalyzed coupling reaction, which dictates the efficiency and selectivity of the final product formation. The catalytic cycle typically initiates with the oxidative addition of the palladium species to the carbon-halogen bond of the pyridine substrate, forming a reactive organopalladium intermediate that is primed for subsequent transformations. This step is critical as it determines the rate of the overall reaction and is influenced heavily by the electronic nature of the ligands surrounding the metal center. Following oxidative addition, the coordination and insertion of the vinyl compound, such as an acrylate ester, occur to extend the carbon framework and establish the desired substitution pattern on the pyridine ring. The cycle concludes with a reductive elimination step that releases the final substituted pyridine product and regenerates the active palladium catalyst for further turnover. Understanding these mechanistic details is vital for R&D directors aiming to optimize reaction conditions, as slight variations in temperature or ligand choice can significantly impact the conversion rates and the profile of byproducts formed during the synthesis.
Controlling the impurity profile is paramount when synthesizing pharmaceutical intermediates, and this process incorporates specific mechanisms to minimize the formation of unwanted side products. The use of specific phosphine ligands, such as triphenylphosphine or tris(2-furyl)phosphine, helps stabilize the palladium center and prevents the aggregation of metal particles that could lead to catalyst deactivation or non-selective reactions. Additionally, the reaction conditions, including the precise control of temperature between 80°C and 160°C, are optimized to favor the desired coupling pathway over competing decomposition or polymerization reactions of the vinyl components. The presence of bases like potassium acetate serves to neutralize acidic byproducts generated during the cycle, maintaining a pH environment that supports catalyst longevity and reaction fidelity. By rigorously managing these parameters, manufacturers can achieve high-purity pharmaceutical intermediates that meet the strict specifications required for clinical and commercial drug production. This level of control ensures that the final material possesses the necessary chemical consistency to support downstream formulation and regulatory approval processes without extensive and costly purification steps.
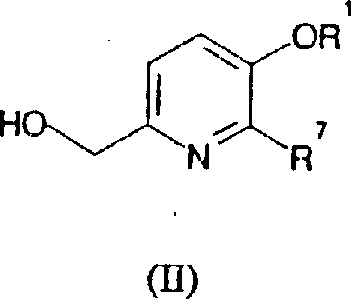
How to Synthesize Substituted Pyridine Derivatives Efficiently
Implementing this synthesis route requires a thorough understanding of the operational parameters defined in the patent to ensure successful replication and scaling of the chemical transformation. The process begins with the careful selection of the halopyridine precursor and the vinyl coupling partner, ensuring that both materials meet the necessary purity standards to prevent catalyst poisoning or side reactions. Operators must maintain an inert atmosphere, typically using nitrogen, to protect the sensitive palladium catalyst from oxidation and moisture ingress that could degrade performance. The reaction mixture is then heated to the specified temperature range, where monitoring via HPLC is essential to determine the endpoint and ensure complete consumption of the starting materials. Detailed standardized synthesis steps are crucial for maintaining consistency across different production batches and facilities.
- Prepare the reaction mixture by combining the halopyridine intermediate with the vinyl compound and palladium catalyst in a polar solvent system.
- Heat the mixture to elevated temperatures between 80°C and 160°C under an inert atmosphere to facilitate the coupling reaction.
- Isolate the final product through aqueous workup, extraction, and purification techniques such as crystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers profound strategic benefits that extend far beyond simple chemical transformation metrics. The streamlined nature of the palladium-catalyzed process significantly reduces the number of unit operations required, which directly translates to lower capital expenditure on equipment and reduced operational overheads for manufacturing facilities. By eliminating the need for complex multi-step sequences found in older methods, the process minimizes the handling of hazardous intermediates and reduces the overall environmental footprint of the production cycle. This efficiency gain allows suppliers to offer more competitive pricing structures while maintaining healthy margins, providing a tangible cost reduction in pharmaceutical intermediates manufacturing for their clients. Furthermore, the robustness of the reaction conditions ensures high reliability in production scheduling, reducing the risk of delays caused by failed batches or difficult purifications. These factors collectively enhance the resilience of the supply chain, ensuring that critical materials are available when needed to support uninterrupted drug development and commercialization timelines.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal steps and the use of catalytic rather than stoichiometric reagents drastically lowers the raw material costs associated with production. By optimizing the catalyst loading and recycling potential, manufacturers can achieve substantial cost savings without sacrificing yield or quality. The simplified workup procedures reduce solvent consumption and waste disposal costs, contributing to a more sustainable and economically efficient operation. Additionally, the higher yields obtained through this method mean that less starting material is required to produce the same amount of final product, further driving down the cost per kilogram. These cumulative efficiencies create a strong value proposition for buyers seeking to optimize their bill of materials for complex drug candidates.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials mitigates the risk of supply disruptions that often plague specialized chemical supply chains. The robustness of the catalytic system allows for flexible manufacturing schedules, enabling producers to respond quickly to fluctuations in market demand without lengthy changeover times. This agility is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug manufacturers can adhere to their own production commitments. The consistency of the process also reduces the need for extensive quality testing and rework, speeding up the release of materials for shipment. Consequently, partners can rely on a steady flow of high-quality intermediates that support their long-term strategic planning and inventory management goals.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, allowing for seamless transition from laboratory benchtop experiments to multi-ton commercial production runs without significant re-engineering. The use of aqueous solvent mixtures and the reduction of hazardous waste streams align with modern environmental regulations and corporate sustainability goals. This compliance reduces the regulatory burden on manufacturers and minimizes the risk of fines or shutdowns due to environmental violations. The ability to scale efficiently ensures that supply can grow in tandem with the commercial success of the final drug product, preventing shortages during peak demand periods. This scalability reinforces the position of the supplier as a long-term strategic partner capable of supporting the entire lifecycle of a pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this palladium-catalyzed synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners. Understanding these details helps stakeholders make informed decisions about integrating this technology into their supply networks.
Q: What are the primary advantages of this palladium-catalyzed method over conventional synthesis?
A: This method eliminates the need for harsh conditions found in older protocols, significantly improving suitability for large-scale commercial production while maintaining high purity standards.
Q: Which catalysts are preferred for optimizing yield in this vinyl substitution reaction?
A: Palladium(II) catalysts such as Pd(OAc)2 combined with phosphine ligands like triphenylphosphine are preferred to ensure efficient coupling and minimize side reactions.
Q: How does this process impact the supply chain stability for psoriasis drug intermediates?
A: By utilizing commercially available starting materials and robust reaction conditions, the process enhances supply continuity and reduces the risk of production bottlenecks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Pyridine Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging deep expertise in organometallic catalysis to deliver exceptional value to our global clientele. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale supply. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of substituted pyridine derivatives meets the highest international standards. Our commitment to quality and consistency makes us a trusted partner for companies requiring high-purity pharmaceutical intermediates for critical therapeutic applications. We understand the complexities of the pharmaceutical supply chain and are dedicated to providing solutions that enhance your operational efficiency and product quality.
We invite you to engage with our technical procurement team to discuss how our capabilities can align with your specific project requirements and timelines. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our manufacturing processes can optimize your budget without compromising on quality. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your unique chemical needs. Let us collaborate to drive innovation and efficiency in your supply chain, ensuring the successful development and commercialization of your next-generation therapies.
