Advanced Synthetic Route for Bempedoic Acid Intermediates Enhancing Commercial Scalability
Introduction to Next-Generation Bempedoic Acid Synthesis
The pharmaceutical landscape for cardiovascular therapeutics has been significantly reshaped by the approval of Bempedoic Acid (ETC-1002), a potent non-statin oral medication designed to lower LDL-C levels in patients with heterozygous familial hypercholesterolemia and atherosclerotic cardiovascular disease. As global demand for this critical therapy escalates, the chemical industry faces an urgent imperative to establish robust, scalable, and cost-efficient manufacturing pathways. Patent CN112110828A discloses a groundbreaking synthetic methodology that addresses the longstanding bottlenecks associated with traditional production routes. By introducing novel intermediate compounds featuring amide or cyano electron-withdrawing groups, this technology fundamentally restructures the synthesis logic, offering a viable solution for reliable pharmaceutical intermediate suppliers aiming to secure long-term supply chains. The strategic shift from unstable ester-based precursors to robust amide architectures not only streamlines the reaction sequence but also dramatically enhances the physicochemical properties of key intermediates, facilitating easier purification and higher overall yields.
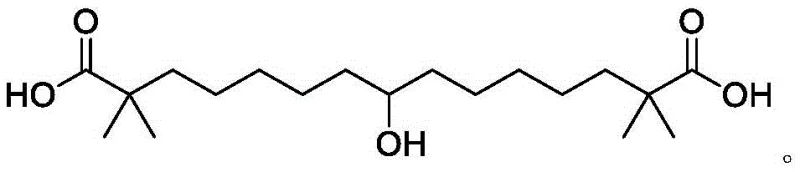
This technical insight report delves deep into the mechanistic advantages and commercial implications of this patented process. For R&D directors and procurement leaders, understanding these nuances is paramount for evaluating potential technology partners. The disclosed method eliminates the reliance on hazardous reagents like sodium hydride and expensive coupling agents such as p-toluenesulfonylmethyl isonitrile, which have historically plagued the economic viability of Bempedoic Acid production. Instead, it leverages widely available starting materials and milder reaction conditions, paving the way for substantial cost reduction in API manufacturing. Furthermore, the integration of enzymatic reduction steps highlights a commitment to green chemistry principles, aligning with increasingly stringent environmental regulations governing modern chemical production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Bempedoic Acid has been hindered by complex multi-step sequences that suffer from poor atom economy and significant safety hazards. Prior art, such as the route described in WO2004067489, relies on the alkylation of ethyl isobutyrate with 1,5-dibromopentane using LDA at cryogenic temperatures, followed by a problematic 2+1 condensation with p-toluenesulfonylmethyl isonitrile. This approach is fraught with difficulties, including the high cost of the isonitrile reagent, the extreme danger associated with scaling up reactions involving sodium hydride, and the generation of substantial toxic waste during the acidic hydrolysis of sulfone intermediates. Moreover, the final reduction step typically requires large excesses of sodium borohydride, leading to cumbersome post-processing and low recovery rates. Similarly, alternative routes reported by other entities, such as WO2020141419, attempt to simplify the process via direct alkylation of diethyl acetone dicarboxylate but encounter severe issues with side reactions. The ester groups in these precursors are prone to unwanted acylation, resulting in difficult-to-remove viscous by-products that compromise the purity of the final drug substance and necessitate expensive chromatographic purification.
The Novel Approach
In stark contrast, the methodology outlined in CN112110828A introduces a paradigm shift by utilizing Compound 1, which bears stable electron-withdrawing groups (EWG) such as N,N-dimethylamide, cyclohexylamide, morpholinamide, or cyano groups. This structural modification is the cornerstone of the invention's success. By replacing the reactive ester functionality with these robust groups during the initial alkylation and subsequent condensation with the dicarbonyl Compound 4, the process effectively suppresses competitive acylation side reactions. The result is a much cleaner reaction profile and the formation of the bis-alkylated intermediate Compound 5 with superior selectivity. As illustrated in the comprehensive reaction scheme below, this route offers multiple divergent pathways to the final product, providing flexibility for process optimization. Whether proceeding through the ketone intermediate Compound 7 via direct hydrolysis or through the amide intermediate Compound 6 followed by reduction, the new method ensures high crystallinity of intermediates, which is a critical factor for industrial scalability.
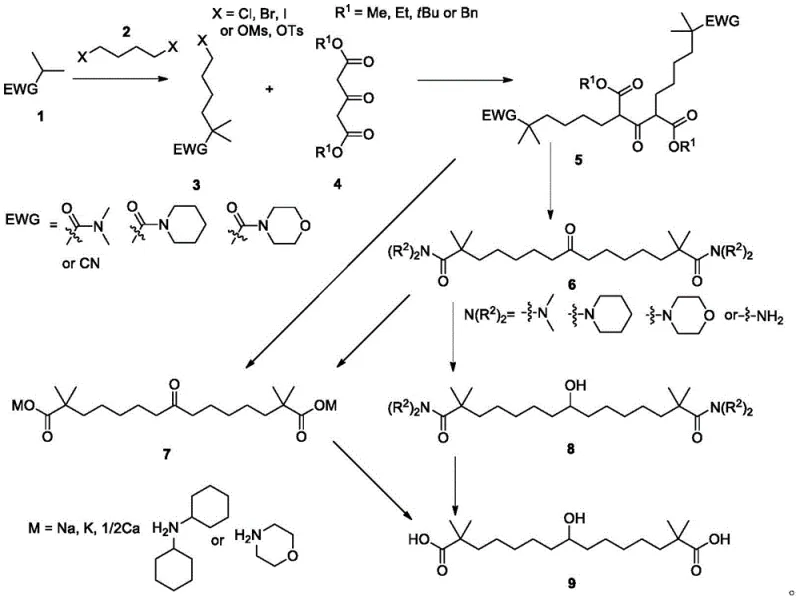
Mechanistic Insights into Amide-Mediated Alkylation and Condensation
The core innovation of this synthesis lies in the electronic and steric properties imparted by the amide or cyano substituents on the alpha-carbon. In conventional ester-based routes, the enolate generated from the ester is highly nucleophilic but also renders the carbonyl carbon susceptible to attack by other nucleophiles present in the reaction mixture, leading to the aforementioned acylation by-products. However, amides and nitriles possess different resonance stabilization characteristics that modulate the reactivity of the adjacent carbon center. When Compound 1 is treated with a base such as n-butyllithium, LDA, or LiHMDS, the resulting anion is sufficiently nucleophilic to displace the halide in Compound 2 efficiently, yet the resulting Compound 3 retains a stability that prevents it from acting as an electrophile in subsequent steps. This orthogonality is crucial when reacting with the tricarbonyl Compound 4. The condensation reaction proceeds cleanly to form the C-C bonds necessary for the fifteen-carbon backbone without the scrambling of functional groups that plagues ester chemistry. Furthermore, the choice of EWG allows for tunable hydrolysis conditions; for instance, the cyano group can be hydrolyzed under acidic conditions to the carboxylic acid, while the amide groups can be selectively cleaved or retained depending on the desired intermediate, offering process chemists valuable control over the synthesis trajectory.
Another critical mechanistic advantage is the enhancement of solid-state properties. The intermediates generated in this pathway, particularly Compounds 5, 6, 7, and 8, exhibit markedly improved crystallization behavior compared to their oily or amorphous ester-based counterparts. From a thermodynamic perspective, the introduction of polar amide or salt functionalities increases the lattice energy of the molecules, promoting orderly packing in the solid state. This phenomenon is not merely academic; it has profound practical implications for impurity control. Viscous liquid by-products, which are notoriously difficult to separate from liquid intermediates, are easily excluded from the growing crystal lattice of the solid product. Consequently, simple filtration and washing steps can achieve purity levels that would otherwise require resource-intensive distillation or column chromatography. This inherent purification capability is a key driver for the high purity specifications achievable with this route, ensuring that the final API meets the rigorous quality standards demanded by regulatory agencies for cardiovascular medications.
How to Synthesize Bempedoic Acid Efficiently
The execution of this synthetic strategy involves a logical progression of bond-forming and functional group transformation steps that are amenable to standard chemical engineering practices. The process begins with the precise alkylation of the EWG-bearing starting material, followed by a controlled double condensation to build the carbon skeleton. Subsequent steps involve the strategic removal of the directing groups and the establishment of the final oxidation state. The beauty of this protocol lies in its modularity; manufacturers can choose to isolate stable intermediates like Compound 7 for quality control checkpoints before proceeding to the final reduction. Detailed operational parameters, including specific solvent systems, temperature ramps, and workup procedures, are critical for maximizing yield and safety. For a comprehensive breakdown of the standardized operating procedures required to implement this technology in a GMP environment, please refer to the technical guide below.
- Perform alkylation of Compound 1 (containing amide or cyano EWG) with dihaloalkane Compound 2 under basic conditions to form Compound 3.
- Condense Compound 3 with dicarbonyl Compound 4 using a base catalyst to generate the bis-alkylated intermediate Compound 5.
- Subject Compound 5 to acid hydrolysis and salt formation to yield Compound 7, followed by enzymatic or chemical reduction to obtain Bempedoic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates directly into enhanced operational resilience and financial efficiency. The primary value proposition is the significant simplification of the supply chain for raw materials. By eliminating the need for specialized and costly reagents like p-toluenesulfonylmethyl isonitrile, manufacturers can source starting materials from a broader base of commodity chemical suppliers, thereby reducing the risk of supply disruptions. Furthermore, the avoidance of cryogenic conditions in certain variations of the process reduces energy consumption and alleviates the burden on specialized cooling infrastructure, leading to lower utility costs per kilogram of product. The improved safety profile, characterized by the removal of pyrophoric sodium hydride and the reduction of hazardous waste streams, also lowers the overhead costs associated with environmental compliance and workplace safety management. These factors collectively contribute to a more sustainable and economically viable manufacturing model.
- Cost Reduction in Manufacturing: The elimination of expensive coupling reagents and the reduction in the number of purification steps drive down the direct cost of goods sold. By avoiding the use of stoichiometric amounts of hazardous reducing agents and minimizing solvent usage through improved crystallization, the process achieves substantial cost savings without compromising quality. The ability to use cheaper bases and solvents in the alkylation steps further optimizes the cost structure, making the final API more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials ensures a consistent and reliable supply of intermediates. The robustness of the amide and cyano intermediates against degradation during storage and transport reduces the risk of material loss and quality deviations. This stability allows for larger batch sizes and longer campaign runs, improving overall equipment effectiveness and ensuring that delivery timelines for downstream pharmaceutical customers are consistently met.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily manageable in large-scale reactors. The generation of less hazardous waste and the potential for enzymatic biocatalysis align with green chemistry initiatives, reducing the environmental footprint of production. This compliance with environmental standards mitigates regulatory risks and enhances the corporate social responsibility profile of the manufacturing entity, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of the process. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this route for commercial production.
Q: Why does the new synthesis route utilize amide or cyano groups instead of esters?
A: Amide and cyano groups offer superior stability compared to ester groups during the alkylation and condensation steps. This stability significantly reduces unwanted acylation side reactions with the dicarbonyl coupling partner, leading to higher crude purity and simplified downstream purification processes.
Q: How does this method improve the crystallization of intermediates?
A: The structural modification of the intermediates, specifically Compounds 5, 6, 7, and 8, enhances their crystallization properties in various solvents. Improved crystallinity allows for more effective removal of viscous by-products and impurities through standard filtration and washing, ensuring consistent high purity for the final API.
Q: Is the enzymatic reduction step suitable for large-scale manufacturing?
A: Yes, the process utilizes water-soluble salt intermediates (Compound 7) which are highly compatible with enzymatic carbonyl reduction. This green chemistry approach operates under mild conditions (20-40°C), minimizes hazardous waste generation, and avoids the safety risks associated with large-scale handling of stoichiometric metal hydride reducing agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bempedoic Acid Supplier
As the global demand for lipid-lowering therapies continues to surge, securing a manufacturing partner with the technical expertise to execute complex synthetic routes is critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of the amide-mediated synthesis described in CN112110828A, ensuring stringent purity specifications and rigorous QC labs validate every batch. We understand that the transition from bench-scale discovery to commercial supply requires not just chemical knowledge but also deep engineering insight, which our team delivers with precision.
We invite pharmaceutical companies and contract research organizations to collaborate with us to leverage this advanced technology for their Bempedoic Acid supply needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and bring life-saving medications to patients faster and more efficiently.
