Advanced Manufacturing of High-Purity Phenylalaninol for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-efficient pathways for producing high-value chiral intermediates. A pivotal development in this domain is detailed in patent CN101250116B, which discloses a novel method for the preparation of phenylalaninol with superior impurity control. Phenylalaninol serves as a critical building block in organic synthesis, most notably as a precursor for Evans reagents (benzyl oxazolidinones), which are indispensable chiral auxiliaries in asymmetric synthesis. Furthermore, it acts as a key intermediate in the production of Aliskiren, a potent renin inhibitor used for treating hypertension. The structural integrity and purity of this molecule are paramount for downstream applications.
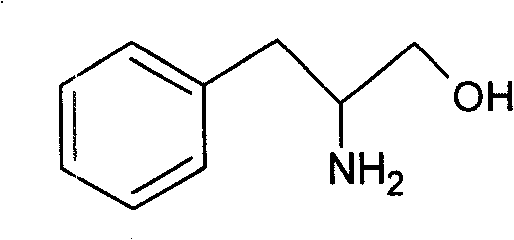
The breakthrough presented in this patent addresses the longstanding economic and technical challenges associated with traditional reduction methods. By shifting from expensive sodium borohydride to the more economical potassium borohydride and optimizing the solvent system to a methanol-water mixture, the inventors have created a process that not only reduces raw material costs but also significantly enhances product quality. This technical advancement represents a substantial opportunity for reliable phenylalaninol suppliers to offer a more competitive product to the global market, ensuring a steady supply of high-purity intermediates for complex drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the standard industrial approach for synthesizing phenylalaninol often relied on methodologies described in literature such as Organic Process Research & Development (2003). These conventional routes typically utilized sodium borohydride (NaBH4) as the reducing agent in tetrahydrofuran (THF) solvent systems. While chemically feasible, this approach presents severe drawbacks for large-scale manufacturing. Firstly, sodium borohydride is considerably more expensive than its potassium counterpart, directly inflating the cost of goods sold (COGS). Secondly, the use of THF poses significant logistical and safety challenges; it is difficult to recover efficiently due to its tendency to form peroxides and its high volatility, leading to increased solvent consumption and waste disposal costs. Moreover, these older methods often struggled with impurity profiles, specifically the formation of condensation byproducts that complicated purification and reduced overall yield, making them less suitable for the rigorous demands of modern GMP pharmaceutical production.
The Novel Approach
The methodology outlined in patent CN101250116B fundamentally re-engineers the reduction step to overcome these deficiencies. The core innovation lies in the substitution of sodium borohydride with potassium borohydride (KBH4), a reductant that offers a much more favorable cost profile without compromising reactivity. Crucially, the process replaces the problematic THF solvent with a mixed system of methanol and water. This change is transformative for cost reduction in pharmaceutical intermediates manufacturing, as methanol and water are inexpensive, easily recyclable, and safer to handle on a multi-ton scale. The patent describes a precise sequence involving esterification followed by reduction under optimized conditions. By carefully controlling the molar ratios of L-phenylalanine to thionyl chloride (1:1.2 to 1:2) and managing the addition rate of the reductant, the process effectively suppresses side reactions. This results in a cleaner reaction profile, simplifying downstream processing and delivering a high-quality final product that meets stringent purity specifications required by top-tier pharmaceutical clients.
Mechanistic Insights into Potassium Borohydride Reduction and Impurity Suppression
To fully appreciate the technical sophistication of this route, one must understand the mechanistic nuances of the reduction and the specific strategies employed to inhibit impurity formation. The synthesis begins with the esterification of L-phenylalanine using thionyl chloride in methanol, converting the amino acid into its methyl ester hydrochloride salt. This activation step is critical for facilitating the subsequent reduction. During the reduction phase, potassium borohydride acts as a hydride donor, attacking the carbonyl carbon of the ester to generate the primary alcohol functionality found in phenylalaninol. However, the reaction environment is delicate; if not strictly controlled, the newly formed amino alcohol can react with trace aldehyde intermediates or unreacted esters to form imines. The patent identifies a specific major impurity, N-(2-phenylpropyl alcohol amino) phenylalaninol, which arises from the condensation of phenylalaninol with phenylpropionaldehyde followed by reduction.
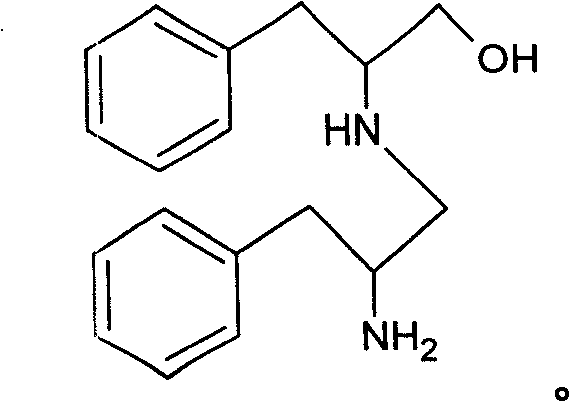
The genius of the patented process lies in its thermal management and solvent engineering to prevent this condensation. By maintaining the reduction temperature at a low range of 0-5°C during the addition of potassium borohydride, the kinetic energy of the molecules is kept low enough to favor the desired reduction over the slower condensation pathway. Furthermore, the use of a methanol-water solvent system (volume ratio 1:2 to 1:5) creates a polarity environment that stabilizes the transition states for reduction while disfavoring the dehydration steps necessary for imine formation. After the reduction is complete, the protocol mandates heating the mixture to 60-80°C. This step serves a dual purpose: it decomposes any excess potassium borohydride to prevent safety hazards during workup, and it helps to hydrolyze any transient imine species that may have formed, reverting them back to the desired amine and aldehyde, which can then be separated. This multi-layered approach to impurity control ensures that the final crystallization from toluene yields phenylalaninol of exceptional purity, minimizing the need for costly chromatographic purification.
How to Synthesize Phenylalaninol Efficiently
Implementing this synthesis requires strict adherence to the operational parameters defined in the patent to ensure reproducibility and safety. The process is designed to be robust, moving from laboratory scale to industrial production with minimal deviation. The following guide outlines the critical operational phases derived from the patent embodiments, focusing on the precise stoichiometry and thermal controls necessary for success. Operators must pay close attention to the exothermic nature of the esterification and the careful addition of the reductant to maintain the narrow temperature window required for high yield. For a comprehensive understanding of the exact equipment setup and safety protocols, the detailed standardized synthesis steps are provided in the guide below.
- Esterify L-phenylalanine with methanol and thionyl chloride at controlled temperatures to form the methyl ester intermediate.
- Perform reduction using potassium borohydride in a methanol-water solvent system at low temperatures (0-5°C) to minimize side reactions.
- Quench excess reductant by heating to 60-80°C, followed by organic extraction and toluene crystallization to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN101250116B offers tangible strategic benefits beyond mere technical elegance. The shift to a potassium borohydride-based process directly addresses the pressure to reduce manufacturing costs in the highly competitive API intermediate sector. By eliminating the reliance on expensive sodium borohydride and difficult-to-recycle THF, the overall variable cost of production is significantly lowered. This cost efficiency allows suppliers to offer more competitive pricing structures without sacrificing margins, providing a buffer against raw material price volatility. Furthermore, the simplified solvent system enhances the environmental profile of the manufacturing process, aligning with the increasingly strict ESG (Environmental, Social, and Governance) criteria demanded by multinational pharmaceutical corporations. The ability to source high-purity phenylalaninol from a process that is both economically and environmentally sustainable is a key differentiator in the global supply chain.
- Cost Reduction in Manufacturing: The replacement of sodium borohydride with potassium borohydride represents a direct material cost saving, as the latter is a commodity chemical available at a fraction of the price. Additionally, the switch from THF to a methanol-water system drastically reduces solvent costs. Methanol and water are not only cheaper to purchase but are also significantly easier to recover and recycle through standard distillation processes compared to THF, which requires specialized handling to prevent peroxide formation. This optimization leads to substantial cost savings in utility consumption and waste treatment, as the aqueous waste stream is easier to manage than organic solvent waste. Consequently, the total cost of ownership for the manufacturing process is reduced, allowing for more aggressive pricing strategies in the marketplace.
- Enhanced Supply Chain Reliability: The raw materials required for this process—L-phenylalanine, thionyl chloride, methanol, and potassium borohydride—are all widely available commodity chemicals with stable global supply chains. Unlike specialized reagents that might suffer from supply disruptions, these inputs can be sourced from multiple vendors, mitigating the risk of production stoppages due to raw material shortages. The robustness of the process, demonstrated by the successful scaling to 1kmol batches in the patent examples, ensures that suppliers can meet large volume orders consistently. This reliability is crucial for pharmaceutical customers who require uninterrupted supply to maintain their own production schedules for critical drugs like Aliskiren. The simplified workflow also reduces the lead time for high-purity phenylalaninol, as fewer purification steps are needed to remove complex solvent residues or difficult impurities.
- Scalability and Environmental Compliance: The patent explicitly validates the scalability of this method through Embodiment 5, which successfully processed 165kg of starting material to achieve an 87.4% yield. This demonstrates that the chemistry translates effectively from the bench to the plant floor, a common failure point for many novel synthetic routes. From an environmental compliance perspective, the process generates less hazardous waste. The absence of THF reduces the burden of volatile organic compound (VOC) emissions, and the use of water as a co-solvent dilutes the effluent, making wastewater treatment more straightforward and cost-effective. This alignment with green chemistry principles facilitates smoother regulatory approvals and audits, ensuring long-term operational continuity for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of phenylalaninol using this optimized method. These answers are derived directly from the experimental data and technical specifications provided in patent CN101250116B, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your supply chain.
Q: Why is potassium borohydride preferred over sodium borohydride for this synthesis?
A: Potassium borohydride is significantly more cost-effective than sodium borohydride. Furthermore, the optimized process conditions using KBH4 effectively suppress the formation of imine-based impurities, leading to higher purity phenylalaninol suitable for chiral auxiliary synthesis.
Q: What is the critical factor in controlling impurities during the reduction step?
A: Temperature control is paramount. Maintaining the reaction temperature between 0-5°C during the addition of the reductant prevents the condensation of phenylalaninol with aldehyde intermediates, which would otherwise form difficult-to-remove imine impurities.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly demonstrates scalability with a 1kmol (165kg starting material) embodiment achieving an 87.4% yield. The use of common solvents like methanol and toluene facilitates easy recovery and large-scale processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylalaninol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development and manufacturing. Our technical team has thoroughly analyzed the pathway described in CN101250116B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient process to life. We are committed to delivering phenylalaninol that meets stringent purity specifications, utilizing our rigorous QC labs to ensure every batch is free from the impurities that plague conventional methods. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this potassium borohydride reduction, guaranteeing a consistent and reliable supply for your chiral synthesis needs.
We invite you to collaborate with us to optimize your supply chain for phenylalaninol and related chiral building blocks. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this improved synthesis route can impact your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot runs and to discuss route feasibility assessments for your upcoming projects. Let us be your partner in driving efficiency and quality in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
