Advanced Stereospecific Synthesis of Piperidine Derivatives for Commercial Scale-up
Advanced Stereospecific Synthesis of Piperidine Derivatives for Commercial Scale-up
The pharmaceutical industry continuously demands high-purity chiral intermediates to ensure the efficacy and safety of final drug products. Patent CN102093260A introduces a groundbreaking methodology for the stereospecific synthesis of piperidine derivatives, addressing critical challenges in stereochemical control and process efficiency. This technology leverages readily available amino acid precursors, specifically L-glutamic acid, to construct complex nitrogen-containing heterocycles with exceptional optical purity. By utilizing novel dialdehyde and dinitrile intermediates, the disclosed methods enable the production of key structural units used in the synthesis of potent therapeutic agents, including DPP-4 inhibitors and kinase inhibitors. For R&D directors and procurement specialists, understanding this proprietary route offers a strategic advantage in securing reliable supply chains for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for piperidine derivatives often suffer from significant drawbacks that hinder commercial viability and cost-effectiveness. Conventional methods frequently rely on multi-step sequences involving the isolation of unstable intermediates, such as dialdehydes, which are prone to decomposition and polymerization under standard reaction conditions. These instability issues lead to inconsistent yields and complicate purification processes, resulting in higher production costs and extended lead times. Furthermore, achieving high stereochemical purity typically requires expensive chiral catalysts or cumbersome resolution steps that generate substantial waste. The reliance on harsh reaction conditions and transition metal catalysts also raises concerns regarding residual metal impurities, necessitating additional downstream processing to meet stringent regulatory standards for active pharmaceutical ingredients.
The Novel Approach
The innovative approach detailed in the patent overcomes these limitations through the development of robust one-pot synthesis strategies and the utilization of stable dinitrile precursors. By converting diester compounds directly into cyclic nitrogen-containing compounds without isolating sensitive dialdehyde intermediates, the process significantly reduces operational complexity and material loss. The method employs stereospecific alkylation controlled by the inherent chirality of the starting amino acid, ensuring high diastereomeric excess without the need for external chiral auxiliaries. Additionally, the option to utilize dinitrile intermediates provides a stable alternative that facilitates easier handling and storage, enhancing the overall reliability of the manufacturing process. This streamlined workflow not only improves yield consistency but also aligns with green chemistry principles by minimizing solvent usage and waste generation.
Mechanistic Insights into Stereospecific Alkylation and Cyclization
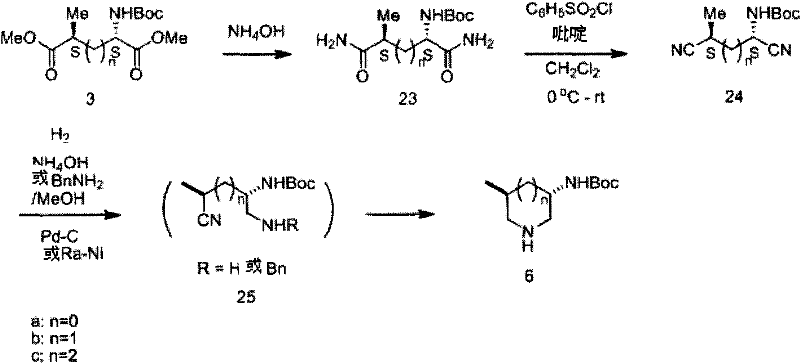
The core of this synthetic strategy lies in the precise control of stereochemistry during the alkylation of the protected amino acid derivative. The process begins with the protection of L-glutamic acid, followed by deprotonation at the gamma-position using a strong base such as lithium hexamethyldisilazide (LiHMDS) at low temperatures. The existing chiral center at the alpha-position exerts a powerful directing effect, guiding the incoming alkylating agent to attack from the less hindered face. This substrate-controlled diastereoselection ensures the predominant formation of the desired 4S isomer, establishing the critical stereocenters required for biological activity. Subsequent reduction of the diester to a dialdehyde or dinitrile preserves this stereochemical integrity, setting the stage for the final ring-closing step.
The cyclization mechanism involves a reductive amination or catalytic hydrogenation sequence that efficiently closes the six-membered piperidine ring. In the one-pot dialdehyde route, the intermediate is treated with ammonia or a primary amine in the presence of a reducing agent like sodium borohydride. This tandem reaction forms the imine intermediate in situ, which is immediately reduced to the amine, driving the equilibrium towards the cyclic product. Alternatively, the dinitrile pathway utilizes catalytic hydrogenation with Raney nickel or palladium on carbon to reduce the nitrile groups and facilitate cyclization. Both pathways are designed to minimize racemization, ensuring that the final piperidine derivatives maintain high optical purity, often exceeding 98% ee as verified by chiral HPLC analysis.
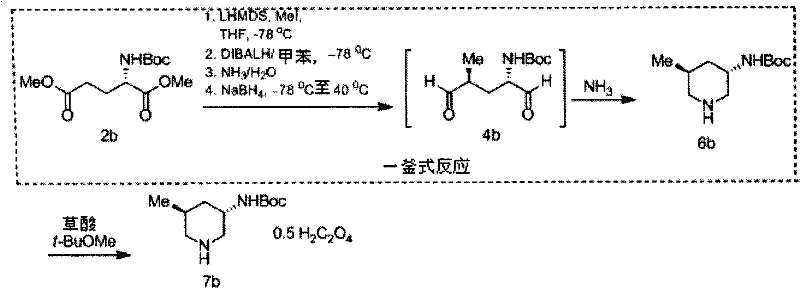
How to Synthesize (3S,5S)-3-(tert-butoxycarbonylamino)-5-methylpiperidine Efficiently
The synthesis of this specific piperidine derivative exemplifies the efficiency of the patented technology, offering a clear pathway from commodity chemicals to high-value intermediates. The procedure integrates protection, alkylation, and cyclization into a cohesive workflow that minimizes unit operations. Detailed standardized synthesis steps are provided in the guide below to assist process chemists in replicating these results.
- Prepare the protected amino acid diester intermediate by reacting L-glutamic acid with thionyl chloride and di-tert-butyl dicarbonate to ensure amino group protection.
- Perform stereospecific alkylation at the gamma-position using a strong base like LiHMDS and an alkylating agent such as methyl iodide to establish the desired chiral center.
- Execute a one-pot reduction and cyclization sequence using DIBALH followed by reductive amination with ammonia or amines to form the final piperidine ring structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route presents tangible benefits in terms of cost reduction and supply security. The reliance on L-glutamic acid, a fermentation-derived commodity chemical, ensures a stable and abundant source of raw materials, mitigating risks associated with supply chain disruptions for exotic starting materials. The elimination of isolation steps for unstable intermediates reduces the requirement for specialized equipment and extensive quality control testing between stages, leading to significant operational cost savings. Furthermore, the high stereochemical fidelity of the process reduces the need for costly chiral separation technologies, streamlining the production timeline and enhancing throughput capacity for commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The one-pot nature of the synthesis drastically reduces solvent consumption and labor hours associated with multiple workup and purification stages. By avoiding the use of expensive transition metal catalysts for asymmetric induction and instead relying on substrate control, the process lowers the cost of goods sold. The ability to purify the final product via simple salt formation and recrystallization, rather than complex chromatography, further contributes to substantial cost savings in the manufacturing of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing robust intermediates like dinitriles enhances the stability of the supply chain by allowing for the stockpiling of key precursors without degradation. The modular nature of the synthesis allows for flexible production scheduling, as different amine components can be introduced in the final cyclization step to generate a library of derivatives. This flexibility ensures that manufacturers can respond rapidly to changing market demands for specific piperidine scaffolds used in various therapeutic areas, reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from gram to hundred-gram levels in the patent examples, indicating strong potential for ton-scale production. The avoidance of hazardous reagents and the minimization of waste streams align with increasingly strict environmental regulations, reducing the burden of waste disposal and compliance monitoring. This environmentally friendly profile makes the technology attractive for sustainable manufacturing initiatives, ensuring long-term viability and regulatory approval for commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereospecific synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners.
Q: How is stereochemical purity controlled during the alkylation step?
A: Stereochemical purity is controlled by the existing chirality at the C-2 position of the protected glutamic acid derivative. The steric environment directs the incoming alkyl group to the C-4 position with high diastereoselectivity, predominantly forming the 4S isomer as confirmed by HPLC analysis.
Q: What are the advantages of using dinitrile intermediates over dialdehydes?
A: Dinitrile intermediates offer superior stability compared to dialdehydes, which can be unstable and prone to polymerization. Using dinitriles allows for easier handling, storage, and purification prior to the final cyclization step, thereby improving overall process robustness and yield consistency.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the patent describes successful preparation on scales up to 100 grams and outlines one-pot procedures that minimize isolation steps. The use of commercially available starting materials like L-glutamic acid and standard reagents supports scalability for industrial manufacturing of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperidine Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like the stereospecific piperidine synthesis route to deliver superior value to our global clients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to industrial reactor. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of piperidine derivatives meets the highest international standards for pharmaceutical applications. Our commitment to quality and technical excellence makes us the preferred partner for companies seeking reliable sources of complex heterocyclic building blocks.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of these efficient synthetic routes. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your drug development timelines and commercial goals. Let us help you engineer success with our proven expertise in fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
