Revolutionizing Glufosinate Production: A Technical Deep Dive into Weak-Acid Catalyzed Synthesis
The landscape of agrochemical intermediate manufacturing is constantly evolving, driven by the relentless pursuit of higher purity and reduced operational expenditures. A pivotal advancement in this domain is documented in patent CN100503624C, which discloses a sophisticated three-step process for the preparation of glufosinate and its derivatives. This technology represents a significant departure from traditional methodologies by employing a suitable weak acid as a condensing agent during the critical Michael addition step. Unlike conventional routes that rely on harsh acid anhydrides and alcohol auxiliaries, this innovative approach leverages the subtle catalytic properties of weak acids to facilitate the reaction between methyl phosphonite and acrolein. The result is a streamlined synthetic pathway that not only enhances the theoretical yield but also drastically improves the purity profile of the final herbicide active ingredient. For R&D directors and process chemists, this patent offers a compelling blueprint for optimizing the production of phosphinothricin precursors, addressing long-standing challenges related to impurity management and reaction efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of glufosinate has been plagued by inefficiencies inherent in older synthetic strategies, such as the route developed by Hoechst which utilizes acid anhydrides as condensing agents. In these traditional processes, the presence of acid anhydrides necessitates the use of alcohols as auxiliary solvents or reagents to manage the reaction environment effectively. This dependency introduces a cascade of complications, primarily the generation of substantial quantities of reaction by-products that are chemically similar to the desired intermediate. These impurities are notoriously difficult to separate, often requiring energy-intensive purification steps like repeated crystallization or complex chromatographic techniques, which erode profit margins. Furthermore, the use of anhydrides and alcohols increases the raw material costs and introduces safety hazards associated with handling volatile and corrosive substances. The cumulative effect is a process with a lower overall yield and a higher environmental footprint, making it less attractive for modern, sustainability-focused manufacturing facilities.
The Novel Approach
The methodology outlined in the patent data introduces a paradigm shift by replacing the aggressive acid anhydride system with a molar equivalent of a suitable weak acid. This strategic substitution fundamentally alters the reaction dynamics of the Michael addition between methyl phosphonite and acrolein. By eliminating the need for alcohol auxiliaries, the new process inherently reduces the complexity of the reaction mixture, thereby minimizing the formation of ether or ester-based side products. The weak acid acts as a precise proton donor, activating the acrolein for nucleophilic attack without promoting the extensive degradation or polymerization often seen with stronger acidic conditions. This results in a cleaner reaction profile where the effective methyl phosphonate intermediate is generated with high selectivity. Consequently, the downstream processing becomes significantly more straightforward, allowing for the isolation of high-purity glufosinate with reduced solvent consumption and waste generation, directly translating to operational excellence.
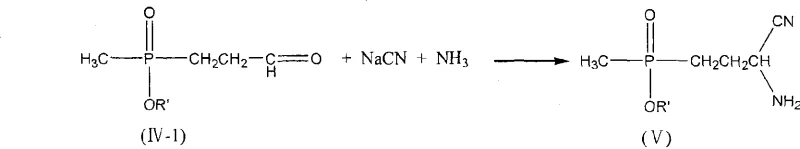
Mechanistic Insights into Weak-Acid Catalyzed Michael Addition and Strecker Reaction
To fully appreciate the technical merit of this synthesis, one must delve into the mechanistic nuances of the weak-acid catalyzed Michael addition. In the first step, the weak acid, which can range from aliphatic carboxylic acids like acetic acid to beta-dicarbonyl compounds, interacts with the carbonyl group of acrolein. This interaction increases the electrophilicity of the beta-carbon, making it more susceptible to nucleophilic attack by the phosphorus center of the methyl phosphonite. Unlike strong acids that might protonate the phosphonite excessively and deactivate it, weak acids maintain a delicate equilibrium that favors the formation of the carbon-phosphorus bond. The resulting intermediate, typically represented as structure IV-1, retains the integrity of the phosphorus oxidation state while establishing the necessary carbon backbone. This precision is crucial because it sets the stage for the subsequent Strecker reaction, where the aldehyde functionality of the intermediate reacts with ammonia and cyanide sources.
The versatility of this mechanism is further evidenced by the potential formation of various intermediate species depending on the specific reaction conditions and substituents. As illustrated in the structural variations provided in the patent documentation, the intermediate can exist in different tautomeric or adduct forms, such as structures IV-2, IV-3, or IV-4, depending on the nature of the R and R' groups.  Regardless of the specific form, these intermediates converge effectively during the Strecker reaction phase. The patent details that the intermediate can react via an iminium ion pathway (structure VI) or a cyanohydrin pathway (structure VII) before stabilizing into the alpha-aminonitrile (structure V). This mechanistic flexibility ensures that even if minor variations occur in the initial Michael addition, the process remains robust enough to drive the reaction toward the desired aminonitrile product, thereby ensuring consistent quality and yield across different batches.
Regardless of the specific form, these intermediates converge effectively during the Strecker reaction phase. The patent details that the intermediate can react via an iminium ion pathway (structure VI) or a cyanohydrin pathway (structure VII) before stabilizing into the alpha-aminonitrile (structure V). This mechanistic flexibility ensures that even if minor variations occur in the initial Michael addition, the process remains robust enough to drive the reaction toward the desired aminonitrile product, thereby ensuring consistent quality and yield across different batches.
How to Synthesize Glufosinate Efficiently
The execution of this synthesis requires careful control of reaction parameters to maximize the benefits of the weak acid catalysis. The process begins with the preparation of the Michael adduct at controlled low temperatures, typically between -10°C and 20°C, to prevent polymerization of the acrolein. Following the formation of the intermediate, the reaction mixture is directly subjected to Strecker conditions without the need for intermediate isolation, which is a key factor in improving overall throughput. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are critical for reproducibility and are outlined in the technical guide below.
- Perform Michael addition of methyl phosphonite and acrolein using a suitable weak acid condensing agent at -10 to 20°C.
- Conduct Strecker reaction on the resulting intermediate using ammonia and sodium cyanide to form the alpha-aminonitrile.
- Hydrolyze the aminonitrile under acidic or alkaline conditions at 90-110°C to yield the final glufosinate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this weak-acid catalyzed process offers tangible strategic advantages that extend beyond mere chemical elegance. The primary value proposition lies in the substantial simplification of the raw material portfolio. By removing the requirement for acid anhydrides and alcohol auxiliaries, the supply chain becomes less vulnerable to price volatility associated with these petrochemical derivatives. Furthermore, the reduction in by-product formation means that less waste needs to be treated and disposed of, leading to significant cost reductions in environmental compliance and waste management. The process operates under moderate conditions that do not require specialized high-pressure equipment, allowing for utilization of existing reactor infrastructure with minimal retrofitting. This ease of integration accelerates the time-to-market for generic versions of the herbicide, providing a competitive edge in the fast-moving agrochemical sector.
- Cost Reduction in Manufacturing: The elimination of expensive acid anhydrides and the associated alcohol solvents directly lowers the bill of materials for every kilogram of product produced. Additionally, the simplified workup procedure reduces the consumption of energy and solvents during the purification stages. Since the crude product contains fewer difficult-to-separate impurities, the yield loss typically associated with aggressive purification steps is minimized, effectively increasing the mass balance of the process. This cumulative effect results in a drastically lower cost of goods sold (COGS), enabling manufacturers to offer more competitive pricing in the global market while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reagents required for this process, such as acetic acid, acrolein, and methyl phosphonite, are commodity chemicals with well-established global supply chains. Unlike specialized catalysts or exotic reagents that might suffer from supply disruptions, these materials are readily available from multiple vendors. This diversity in sourcing options mitigates the risk of production stoppages due to raw material shortages. Moreover, the stability of the weak acid reagents allows for easier storage and handling compared to moisture-sensitive anhydrides, reducing the logistical burden and safety risks associated with inbound logistics and warehouse management.
- Scalability and Environmental Compliance: From a scale-up perspective, the exothermic nature of the Michael addition is better managed under weak acid conditions, reducing the risk of thermal runaways in large-scale reactors. The absence of volatile alcohols also lowers the fire hazard rating of the process, potentially lowering insurance premiums and safety compliance costs. Environmentally, the reduction in organic waste and the avoidance of halogenated or toxic by-products align with increasingly stringent global regulations on chemical manufacturing. This 'green chemistry' aspect not only ensures regulatory compliance but also enhances the brand reputation of the manufacturer as a sustainable partner in the agrochemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a reliable foundation for decision-making. Understanding these details is essential for evaluating the feasibility of adopting this technology for commercial production.
Q: What is the primary advantage of using weak acids over acid anhydrides in glufosinate synthesis?
A: Using weak acids eliminates the need for alcohol auxiliaries, significantly reducing by-product formation and simplifying the purification process, which leads to higher purity end-products.
Q: How does this process impact the impurity profile of the final agrochemical intermediate?
A: The novel route minimizes the formation of difficult-to-separate impurities commonly associated with anhydride-based condensation, resulting in a cleaner crude product that requires less intensive downstream processing.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes common reagents like acetic acid and operates under moderate temperature conditions, making it highly suitable for commercial scale-up without requiring exotic catalysts or extreme pressures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glufosinate Supplier
The technical potential of the weak-acid catalyzed synthesis route for glufosinate is immense, offering a clear path toward more efficient and sustainable herbicide production. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this laboratory innovation to full industrial reality. Our team of expert process chemists is adept at optimizing reaction parameters to ensure stringent purity specifications are met consistently. With our rigorous QC labs and state-of-the-art manufacturing facilities, we are uniquely positioned to deliver high-purity agrochemical intermediates that meet the exacting standards of the global pharmaceutical and crop protection industries.
We invite you to explore how this advanced synthesis route can optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us be your partner in navigating the complexities of fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
