Breakthrough Purification Technology for 6-Aminocapronitrile in Polyamide Manufacturing
The global demand for high-performance polyamides, specifically Nylon 66, continues to drive innovation in the synthesis of its critical precursors. A recent technological advancement disclosed in patent CN113072463A introduces a sophisticated method for recovering 6-aminocapronitrile from the dehydration material of caprolactam ammoniation liquid. This innovation addresses a longstanding bottleneck in the industry where achieving simultaneous high purity and high yield has been technically challenging. By integrating negative pressure rectification with a precise sweating crystallization protocol, this novel approach elevates the purity of 6-aminocapronitrile to exceed 99.999%, a benchmark essential for downstream applications in hexamethylene diamine production. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding this shift from thermal-only separation to hybrid physical-chemical processing is vital for securing supply chains that meet the rigorous specifications of modern polymer manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for isolating 6-aminocapronitrile typically rely heavily on multi-stage rectification processes conducted at elevated temperatures. While effective for bulk separation, these thermal-intensive approaches suffer from significant thermodynamic limitations when attempting to separate 6-aminocapronitrile from structurally similar impurities such as 5-hexenenitrile and unreacted caprolactam. The coexistence of caprolactam and aminocapronitrile at high temperatures during prolonged distillation often triggers undesirable side reactions, including ring-opening and polymerization, which not only degrade the product quality but also foul equipment and reduce overall throughput. Furthermore, standard distillation struggles to break azeotropes or separate components with close boiling points without sacrificing yield, frequently resulting in product purity that stagnates below the critical 99.9% threshold required for high-end nylon synthesis. These inefficiencies create substantial waste streams and increase the cost burden associated with downstream purification steps, making conventional routes less viable for cost-sensitive large-scale operations.
The Novel Approach
In stark contrast, the patented methodology leverages a synergistic combination of vacuum rectification and low-temperature crystallization to overcome these thermodynamic barriers. The process initiates with a gentle negative pressure rectification step, operating between 0.1 to 10 kPa, which concentrates the 6-aminocapronitrile to a mass content of at least 80% without exposing the sensitive nitrile group to excessive thermal stress. Following this concentration phase, the material undergoes a controlled cooling crystallization down to temperatures between -70°C and -40°C. This phase transition exploits the differential solubility and melting points of the target compound versus its impurities, effectively trapping the 6-aminocapronitrile in a solid lattice while excluding liquid impurities. The subsequent sweating process, where the temperature is raised at a precise rate of 0.5 to 10°C/h, allows for the sequential extraction of oil phases rich in 5-hexenenitrile, leaving behind a highly purified solid residue. This strategic decoupling of separation mechanisms ensures that the final product achieves ultra-high purity while maintaining a robust yield profile that traditional thermal methods cannot match.
Mechanistic Insights into Hybrid Rectification and Sweating Crystallization
The core mechanistic advantage of this process lies in the precise manipulation of phase equilibria to isolate 6-aminocapronitrile from the complex reaction matrix. During the initial rectification stage, the reduced pressure lowers the boiling points of the components, allowing for the separation of light ends and heavy residues like caprolactam while collecting a middle fraction enriched with the target nitrile. However, the true purification power is unleashed during the crystallization phase. By cooling the concentrated fraction to sub-zero temperatures, the system enters a metastable state where 6-aminocapronitrile nucleates and grows into crystals. The critical control parameter here is the cooling rate, optimized between 0.5 to 10°C/h, which prevents the occlusion of mother liquor containing impurities within the crystal lattice. Fast cooling would trap impurities, while excessively slow cooling reduces efficiency; the patented range strikes the perfect balance for forming pure, defect-free crystals.
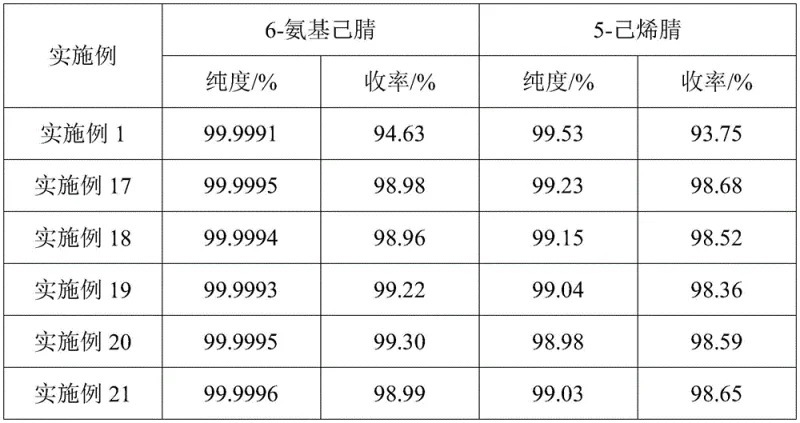
Following crystallization, the sweating mechanism acts as a secondary purification barrier. As the temperature is carefully ramped up, the eutectic mixture containing the lower-melting impurity, 5-hexenenitrile, melts first and is drained off as the first oil product. This is followed by the melting of a second oil phase containing residual 6-aminocapronitrile, which is recycled to the next batch, ensuring zero loss of valuable material. The final residual solid, now stripped of the majority of organic impurities, is melted to yield the final product. This multi-stage physical separation avoids the chemical degradation associated with high-heat distillation, explaining how the process consistently delivers purity levels above 99.999% and yields exceeding 94% under optimized conditions. The ability to recover 5-hexenenitrile as a distinct byproduct with over 98% purity further demonstrates the selectivity of this crystallization-driven mechanism.
How to Synthesize 6-Aminocapronitrile Efficiently
Implementing this recovery process requires strict adherence to the thermal profiles and pressure conditions outlined in the patent to ensure reproducibility at scale. The operation begins with the preparation of the dehydrated ammoniation liquid, which serves as the feedstock for the purification train. Operators must monitor the rectification column closely to ensure the cut points for the fractions align with the specified temperature ranges, particularly collecting the 6-aminocapronitrile-rich fraction between 60°C and 130°C under vacuum. The subsequent crystallization step demands precise temperature control equipment capable of reaching -70°C, followed by a programmed heating cycle for the sweating phase. Detailed standard operating procedures regarding the specific heating rates and incubation times are critical for maximizing the exclusion of 5-hexenenitrile. For a comprehensive guide on the exact parameters and equipment setup required for this synthesis, please refer to the standardized protocol below.
- Perform negative pressure rectification on the dehydrated material at 0.1-10 kPa to collect a fraction containing at least 80% 6-aminocapronitrile.
- Cool the collected fraction to between -70°C and -40°C at a controlled rate to induce crystallization, followed by a heat preservation period.
- Heat the crystallized material gradually to sequentially extract oil products containing 5-hexenenitrile and residual 6-aminocapronitrile.
- Melt the final residual material to obtain the purified 6-aminocapronitrile product with purity exceeding 99.999%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hybrid purification technology translates into tangible strategic advantages beyond mere technical specifications. The primary value driver is the drastic simplification of the purification train, which replaces energy-intensive multi-column distillation setups with a more efficient rectification-crystallization loop. This reduction in thermal load directly correlates to lower utility consumption and reduced maintenance costs associated with reboiler fouling and column packing replacement. Furthermore, the process transforms a waste management challenge into a revenue opportunity by isolating 5-hexenenitrile as a saleable byproduct. Instead of incinerating or disposing of this impurity, manufacturers can capture it with high purity, effectively offsetting the raw material costs of the caprolactam feedstock. This circular economy approach significantly enhances the overall economic viability of the production line.
- Cost Reduction in Manufacturing: The elimination of aggressive high-temperature distillation steps reduces the thermal degradation of equipment and the formation of heavy ends that require costly disposal. By recovering valuable byproducts like 5-hexenenitrile with yields exceeding 92%, the net consumption of raw materials per ton of finished product is substantially lowered. Additionally, the high purity of the final 6-aminocapronitrile reduces the burden on downstream hydrogenation catalysts used to produce hexamethylene diamine, extending their lifecycle and reducing the frequency of expensive catalyst change-outs. These cumulative efficiencies result in a leaner cost structure that provides a competitive edge in the volatile polyamide market.
- Enhanced Supply Chain Reliability: The robustness of the crystallization-based purification ensures consistent product quality regardless of minor fluctuations in the upstream ammoniation reaction feed. This stability is crucial for maintaining long-term contracts with nylon producers who require strict adherence to purity specifications to prevent fiber defects. The ability to recycle the second oil product back into the process minimizes material loss, ensuring that production targets are met with higher certainty. Moreover, the modular nature of the crystallization units allows for easier capacity expansion compared to complex distillation towers, enabling suppliers to scale up production rapidly in response to market demand spikes without compromising quality.
- Scalability and Environmental Compliance: Operating under negative pressure significantly reduces the risk of volatile organic compound (VOC) emissions, aligning the process with increasingly stringent environmental regulations. The lower operating temperatures also diminish the formation of hazardous thermal decomposition byproducts, simplifying wastewater treatment and waste stream management. From a scalability perspective, the process parameters, such as cooling rates and vacuum levels, are easily transferable from pilot scale to commercial multi-ton reactors. This ease of scale-up reduces the time-to-market for new production lines and ensures that the supply of high-purity intermediates remains uninterrupted even as global demand for engineering plastics and fibers continues to grow.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this advanced recovery method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this technology resolves specific pain points in 6-aminocapronitrile manufacturing. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing facilities.
Q: Why is conventional distillation insufficient for producing ultra-high purity 6-aminocapronitrile?
A: Conventional distillation often fails to remove trace impurities like 5-hexenenitrile effectively without causing thermal degradation. High temperatures required for separation can lead to caprolactam ring-opening or polymerization side reactions, limiting purity to below 99.9%.
Q: How does the hybrid rectification and crystallization method improve yield?
A: By combining negative pressure rectification to concentrate the target compound followed by low-temperature sweating crystallization, the process minimizes thermal stress. This allows for the recovery of over 94% yield while achieving purity levels greater than 99.999%.
Q: Can valuable byproducts be recovered during this purification process?
A: Yes, the process specifically enriches and recovers 5-hexenenitrile as a valuable byproduct with purity exceeding 98% and yield over 92%, thereby reducing overall material consumption and improving economic efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Aminocapronitrile Supplier
The technological pathway described in patent CN113072463A represents a significant leap forward in the production of high-value nitrile intermediates, yet translating such complex crystallization protocols into consistent commercial output requires deep process engineering expertise. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this hybrid purification method are fully realized in practice. Our facility is equipped with state-of-the-art low-temperature crystallization units and rigorous QC labs capable of verifying purity specifications down to the ppm level. We understand that for applications in Nylon 66 and HDI synthesis, even trace impurities can be detrimental, which is why our stringent purity specifications and comprehensive analytical testing guarantee a product that meets the most demanding global standards.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized recovery processes, we can help you secure a stable supply of high-purity 6-aminocapronitrile while minimizing total landed costs. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Let us demonstrate how our commitment to advanced purification technologies can strengthen your supply chain resilience and drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
