Advanced Catalytic Route for 6-Aminocapronitrile: Technical Upgrade and Commercial Scalability
The chemical industry continuously seeks robust methodologies for producing high-value nitrile intermediates, and patent CN1100752C presents a significant technological advancement in the manufacturing process for 6-aminocapronitrile. This specific intellectual property outlines a sophisticated two-stage catalytic sequence that begins with the hydroformylation of pentenenitrile isomers and concludes with a selective reductive amination step. For R&D Directors and technical decision-makers, the core innovation lies in the precise manipulation of ligand environments around Group VIII metal centers, which dramatically alters the regioselectivity of the formylation reaction. By leveraging specific phosphite ligands, the process achieves a superior ratio of linear to branched aldehyde intermediates, a critical parameter that directly influences the purity profile of the final 6-aminocapronitrile product. This technical breakthrough addresses long-standing challenges in nitrile chemistry where traditional methods often suffered from poor conversion rates or required excessively harsh conditions that compromised product integrity. The patent data suggests that by optimizing the catalyst composition and reaction parameters, manufacturers can secure a more reliable supply of this key intermediate, which serves as a crucial precursor for caprolactam and nylon 6 production. Understanding the nuances of this patented route is essential for any organization aiming to optimize their supply chain for high-purity polymer additives and pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amino-nitriles from unsaturated nitriles has been plagued by significant inefficiencies that hinder commercial viability and cost-effectiveness. Prior art, such as the methods described in EP-A11401, often relied on cobalt-based catalysts which, while active, demonstrated limited selectivity and required extreme operating pressures that escalated energy consumption and safety risks. In many conventional processes, the reductive amination of aldehyde intermediates resulted in low conversion rates, sometimes as low as 25 percent based on the delta compound, necessitating complex recycling loops that increased capital expenditure. Furthermore, the inability to effectively distinguish between linear and branched isomers during the hydroformylation stage led to contaminated effluent streams, requiring energy-intensive distillation sequences to isolate the desired 5-formylvaleronitrile. These technical bottlenecks not only inflated the cost reduction in fine chemical manufacturing but also introduced variability in the impurity profile, which is unacceptable for sensitive pharmaceutical applications. The degradation of catalyst activity over time in these older systems further compounded operational downtime, creating supply chain vulnerabilities for downstream users relying on consistent quality. Consequently, the industry has long required a more refined catalytic system that could operate under milder conditions while delivering superior selectivity and yield.
The Novel Approach
The patented process introduces a paradigm shift by employing rhodium complexes modified with specific polyphosphite ligands to drive the hydroformylation of pentenenitriles with exceptional precision. This novel approach utilizes catalysts where the molar ratio of cocatalyst to rhodium is carefully tuned, often ranging from 1:1 to 300:1, to maximize the formation of the linear 5-formylvaleronitrile isomer. Experimental data from the patent indicates that using ligands such as the diphosphite of Formula III can push conversion rates to greater than 99.9 percent while maintaining a favorable isomer ratio, a stark contrast to the poor performance of earlier cobalt systems. 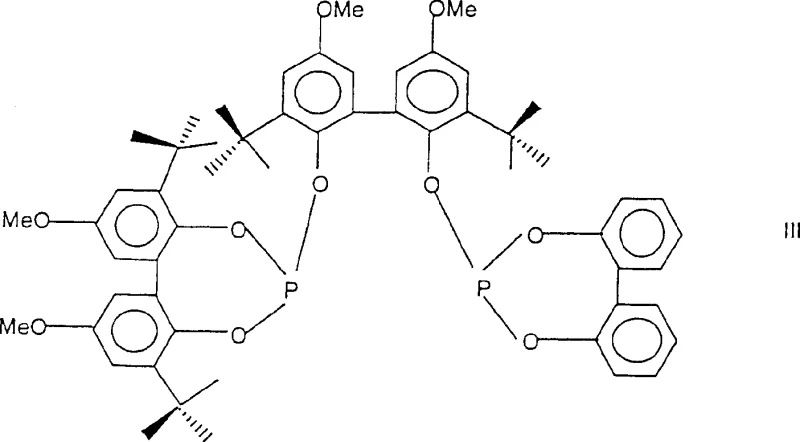
This high level of control allows for the efficient production of 6-aminocapronitrile or mixtures with hexamethylenediamine by simply adjusting temperature and space velocity in the subsequent hydrogenation step. The ability to recycle unconverted olefins and high-boiling catalyst residues back into the reactor further enhances the economic feasibility of the route, aligning with modern green chemistry principles. By mitigating the formation of unwanted byproducts at the source, this method significantly simplifies the downstream purification workflow, thereby reducing the overall manufacturing footprint. For procurement managers, this translates to a more stable cost structure and reduced dependency on complex waste treatment protocols. The integration of these advanced catalytic systems represents a tangible step forward in the commercial scale-up of complex nitriles, offering a competitive edge in the global market for specialty chemicals.
Mechanistic Insights into Rhodium-Catalyzed Hydroformylation and Reductive Amination
The core of this technological advancement rests on the intricate coordination chemistry governing the hydroformylation cycle, where the electronic and steric properties of the ligand play a decisive role. The catalyst system typically comprises a rhodium carbonyl complex modified with bulky polyphosphite ligands, such as those defined by Formula I, which create a specific pocket around the metal center. 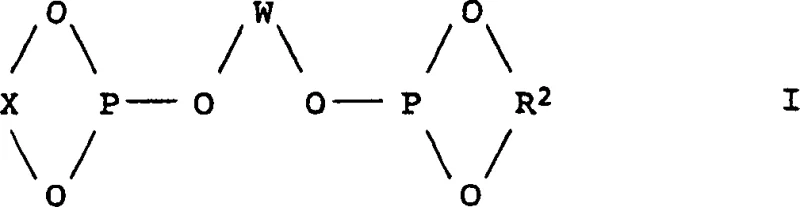
This steric environment favors the coordination of the terminal double bond of the pentenenitrile over internal isomers, directing the insertion of carbon monoxide to occur primarily at the terminal position to yield the linear aldehyde. The presence of electron-withdrawing groups on the phosphite ligands enhances the electrophilicity of the rhodium center, accelerating the oxidative addition of hydrogen and the subsequent migratory insertion steps. In the second stage, the reductive amination of the isolated 5-formylvaleronitrile proceeds via an imine intermediate formed with ammonia, which is then hydrogenated over a heterogeneous catalyst such as ruthenium on alumina. The selection of the hydrogenation catalyst is critical; metals like rhenium, copper, or Group VIII elements supported on porous oxides ensure that the nitrile group remains intact while the aldehyde is converted to the amine. This dual-catalyst strategy ensures that the impurity spectrum is tightly controlled, minimizing the formation of secondary amines or over-reduced diamines unless specifically desired. Such mechanistic precision is vital for R&D teams focusing on the purity and杂质 profile of API intermediates, as it guarantees a consistent quality of the 6-aminocapronitrile output.
Furthermore, the structural nuances of the ligands, such as the bisarylene radicals depicted in Formula IV, contribute to the thermal stability and longevity of the catalyst under reaction conditions. 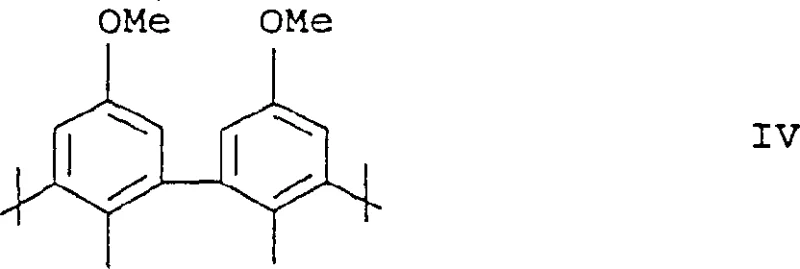
These structural elements prevent the decomposition of the ligand at elevated temperatures, allowing the process to run for extended periods without significant loss of activity. The ability to fine-tune these molecular components allows manufacturers to adapt the process for specific output requirements, whether targeting pure 6-aminocapronitrile for nylon 6 synthesis or a mixture containing hexamethylenediamine for nylon 6,6 applications. This flexibility underscores the robustness of the chemistry, making it a reliable foundation for long-term production strategies. Understanding these mechanistic details empowers technical teams to troubleshoot potential deviations and optimize reactor performance for maximum efficiency.
How to Synthesize 6-Aminocapronitrile Efficiently
Implementing this synthesis route requires a systematic approach to reactor management and separation technology to fully realize the benefits outlined in the patent data. The process begins with the preparation of the hydroformylation catalyst, where rhodium precursors are complexed with the appropriate phosphite ligands in an inert solvent such as toluene or high-boiling esters. Operators must maintain strict control over the partial pressures of carbon monoxide and hydrogen, typically keeping the total pressure between 5 and 20 bar to balance reaction rate with equipment safety. Following the reaction, the effluent undergoes a series of distillation steps where light ends like unreacted pentenenitriles are stripped off and recycled, while the valuable 5-formylvaleronitrile is isolated under vacuum to prevent thermal degradation.
- React pentenenitrile isomers with carbon monoxide and hydrogen using a Group VIII catalyst with phosphite ligands to form 5-formylvaleronitrile.
- Separate 5-formylvaleronitrile from the hydroformylation effluent via fractional distillation under vacuum to remove isomers and high boilers.
- Perform reductive amination of the purified aldehyde with ammonia and hydrogen over a supported metal catalyst to yield 6-aminocapronitrile.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers substantial strategic benefits for procurement managers and supply chain heads looking to optimize their sourcing of critical chemical intermediates. The primary advantage lies in the significant cost reduction in fine chemical manufacturing achieved through improved catalyst selectivity and longevity. By minimizing the formation of branched isomers and byproducts, the process reduces the load on purification units, leading to lower energy consumption and reduced solvent usage per kilogram of product. This efficiency gain directly impacts the bottom line, allowing for more competitive pricing structures without compromising on quality standards. Additionally, the ability to recycle catalyst residues and unreacted feedstocks creates a closed-loop system that mitigates raw material volatility, ensuring enhanced supply chain reliability even during market fluctuations. The robustness of the rhodium-phosphite system means fewer catalyst change-outs and less unplanned downtime, which is crucial for maintaining continuous production schedules. For supply chain planners, this translates to predictable lead times and a steady flow of high-purity 6-aminocapronitrile to meet downstream demand.
- Cost Reduction in Manufacturing: The elimination of inefficient separation steps and the high conversion rates achieved by the novel catalyst system lead to substantial cost savings in operational expenditures. By avoiding the need for extensive purification to remove branched isomers, manufacturers can reduce utility costs and waste disposal fees significantly. The qualitative improvement in yield means that less raw material is required to produce the same amount of finished goods, optimizing the overall material balance. This economic efficiency makes the process highly attractive for large-scale production where marginal gains translate into significant financial value.
- Enhanced Supply Chain Reliability: The stability of the catalyst system and the use of readily available feedstocks like pentenenitriles ensure a consistent production output that safeguards against supply disruptions. The process design allows for the recycling of key components, reducing dependency on external raw material supplies and insulating the production line from market shortages. This reliability is essential for maintaining long-term contracts with pharmaceutical and polymer clients who require uninterrupted delivery schedules. The robust nature of the technology ensures that production targets can be met consistently, fostering trust and stability in business relationships.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard high-pressure reactor technology that can be easily expanded from pilot to commercial scale without fundamental changes to the chemistry. The reduced generation of heavy byproducts and the ability to recycle catalysts align with stringent environmental regulations, minimizing the ecological footprint of the manufacturing facility. This compliance reduces the risk of regulatory penalties and enhances the corporate sustainability profile, which is increasingly important for global supply chains. The ease of scale-up ensures that demand surges can be accommodated quickly, providing a competitive advantage in dynamic markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced manufacturing process. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production frameworks.
Q: What is the primary advantage of the rhodium-phosphite catalyst system in this process?
A: The rhodium-phosphite catalyst system significantly enhances selectivity towards the linear 5-formylvaleronitrile isomer compared to traditional cobalt catalysts, reducing downstream separation costs and improving overall yield efficiency.
Q: How is the purity of 6-aminocapronitrile controlled during manufacturing?
A: Purity is controlled through precise fractional distillation of the hydroformylation effluent to isolate 5-formylvaleronitrile from branched isomers, followed by optimized hydrogenation conditions that minimize over-reduction to hexamethylenediamine.
Q: Can this process be scaled for industrial production of nylon precursors?
A: Yes, the process utilizes standard high-pressure reactor technology and distillation columns compatible with large-scale chemical manufacturing, ensuring reliable supply continuity for polymer and pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Aminocapronitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the synthesis of advanced polymers and pharmaceutical compounds. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic routes described in patent CN1100752C can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 6-aminocapronitrile meets the exacting standards required by our global clientele. Our commitment to technical excellence allows us to navigate the complexities of hydroformylation and reductive amination with precision, delivering products that facilitate your downstream innovation. By partnering with us, you gain access to a supply chain that is both resilient and responsive to your specific technical requirements.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs with tailored solutions. Request a Customized Cost-Saving Analysis to understand how our optimized manufacturing processes can enhance your project economics. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality 6-aminocapronitrile consistently. Let us collaborate to secure your supply chain and drive your product development forward with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
