Advanced Synthesis of S-(4-tolyl)benzenesulfonate for Commercial Scale-up of Complex Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the dual imperatives of regulatory compliance and economic efficiency. A pivotal advancement in this domain is documented in Chinese Patent CN102558004B, which discloses a robust chemical synthesis method for S-(4-tolyl)benzenesulfonate, a critical reagent often utilized as a benzene sulfuration agent and a blocking agent for thiol groups in protein chemistry. This patent represents a significant departure from legacy methodologies by introducing a novel oxidative coupling strategy that utilizes sodium benzenesulfinate and bis(4-tolyl) disulfide as primary starting materials. Unlike conventional routes that rely on hazardous thiols, this innovative approach leverages N-halosuccinimide as a mild oxidant in an organic solvent system, operating effectively within a broad temperature range of -40 to 120°C. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this patent is crucial, as it offers a pathway to high-purity products while drastically mitigating the safety risks associated with malodorous and toxic sulfur-containing precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the technology described in CN102558004B, the industrial synthesis of S-(4-tolyl)benzenesulfonate was fraught with significant operational and environmental challenges. Traditional literature, such as methods cited in the Journal of Sulfur Chemistry, typically employed benzenesulfonyl acid and p-methylthiophenol as reactants, utilizing dichloromethane as a solvent in the presence of cyanuric chloride and N-methylmorpholine. This legacy approach presents severe drawbacks for modern manufacturing facilities; specifically, p-methylthiophenol is characterized by its extreme toxicity, high volatility, and intensely foul odor, which necessitates expensive, specialized containment infrastructure to protect worker safety and prevent environmental contamination. Furthermore, the use of dichloromethane, a chlorinated solvent with increasing regulatory scrutiny due to its environmental persistence and health hazards, complicates waste disposal and increases the overall cost of compliance. The reliance on cyanuric chloride also introduces handling risks due to its lachrymatory nature, making the entire process less desirable for companies aiming to implement green chemistry principles and reduce their carbon footprint in fine chemical intermediates production.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the novel approach detailed in the patent utilizes a much safer and more atom-economical reaction pathway. By substituting the volatile thiol with bis(4-tolyl) disulfide, a stable and odorless solid, the process eliminates the primary source of occupational exposure and odor complaints in the plant. The reaction proceeds through an oxidative coupling mechanism mediated by N-halosuccinimide, preferably N-bromosuccinimide (NBS), which activates the disulfide bond for nucleophilic attack by the sulfinate anion. This transformation is conducted in versatile organic solvents such as acetonitrile or tetrahydrofuran, which are easier to recover and recycle compared to chlorinated alternatives. The reaction equation illustrates this elegant transformation where two equivalents of sodium benzenesulfinate react with one equivalent of the disulfide to yield the target sulfonate ester efficiently.
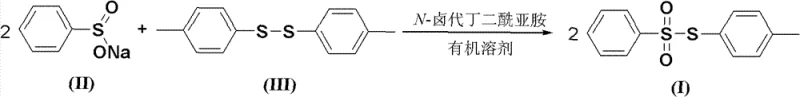
This methodological shift not only enhances safety but also improves the reaction profile, allowing for operation under mild conditions (preferably 20-50°C) that reduce energy consumption and minimize thermal degradation of sensitive functional groups, thereby ensuring a cleaner impurity profile for the final high-purity pharmaceutical intermediate.
Mechanistic Insights into N-Halosuccinimide Mediated Oxidative Coupling
The core of this synthetic breakthrough lies in the precise activation of the sulfur-sulfur bond within the disulfide substrate by the N-halosuccinimide reagent. Mechanistically, the N-halosuccinimide acts as an electrophilic halogen source that interacts with the electron-rich disulfide bond, generating a transient sulfenyl halide species or a hypervalent sulfur intermediate in situ. This activated species is significantly more susceptible to nucleophilic attack by the sulfur atom of the sodium benzenesulfinate anion. The reaction likely proceeds through a concerted or stepwise substitution where the sulfinate sulfur displaces the leaving group, forming the new sulfur-sulfur bond characteristic of the thiosulfonate structure. The stoichiometry is critical here; the patent specifies a molar ratio of disulfide to sulfinate to N-halosuccinimide ranging from 1:2.0-5.0:1.0-4.0, with a preferred embodiment of 1:4.0:2.0. This excess of sulfinate ensures that the activated intermediate is rapidly trapped, preventing side reactions such as the formation of symmetric disulfides or over-oxidation to sulfones, which are common impurities in sulfur chemistry. The choice of N-bromosuccinimide over chloro- or iodo- variants is particularly astute, as the bromine atom offers an optimal balance of electrophilicity and leaving group ability, facilitating rapid conversion at moderate temperatures without the aggressive reactivity that might lead to ring halogenation on the aromatic systems.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed condensation routes. In traditional methods, the presence of strong acids or dehydrating agents like cyanuric chloride can promote polymerization or decomposition of the thiol starting material, leading to complex tarry byproducts that are difficult to separate. In the patented oxidative coupling method, the byproducts are primarily succinimide and sodium halide salts, both of which are water-soluble and easily removed during the aqueous workup phase described in the examples. The use of acetonitrile as a solvent further aids in purity; its polarity stabilizes the ionic intermediates while allowing for easy separation of the organic product from inorganic salts upon addition of saturated brine. This clean reaction profile means that the crude product often requires minimal purification, potentially just a recrystallization or simple distillation, to achieve the reported purity levels of over 98%, which is essential for applications in protein chemistry where trace metal or organic impurities could interfere with biological activity.
How to Synthesize S-(4-tolyl)benzenesulfonate Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters identified in the patent examples to ensure reproducibility and safety. The general procedure involves dissolving the sodium benzenesulfinate and the N-halosuccinimide oxidant in the chosen organic solvent, typically acetonitrile, to form a homogeneous suspension or solution depending on the scale and temperature. A separate solution of the bis(4-tolyl) disulfide is prepared in the same solvent and added dropwise to the reaction vessel under vigorous stirring. This controlled addition rate is vital to manage the exotherm and maintain the concentration of the activated intermediate at a level that favors the desired cross-coupling over homocoupling. The reaction temperature is maintained within the preferred window of 20-50°C, although the patent demonstrates feasibility from -10 to 120°C, allowing flexibility based on available cooling or heating infrastructure. Following the addition, the mixture is stirred for a period of 5 to 10 hours to ensure complete consumption of the starting materials, as monitored by TLC or HPLC. Upon completion, the solvent is removed under reduced pressure, and the residue is partitioned between water and an organic extraction solvent. The organic layer is separated, dried, and concentrated to yield the target S-(4-tolyl)benzenesulfonate. For detailed standard operating procedures and specific troubleshooting tips regarding scale-up, please refer to the technical guide below.
- Dissolve sodium benzenesulfinate and N-halosuccinimide (preferably N-bromosuccinimide) in an organic solvent such as acetonitrile.
- Prepare a solution of bis(4-tolyl) disulfide in the same organic solvent and add it dropwise to the reaction mixture at a controlled temperature between 20-50°C.
- Maintain the reaction temperature until completion, then remove the solvent, extract with saturated brine, and purify the organic layer to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the radical simplification of the raw material supply chain; by eliminating the need for p-methylthiophenol, a specialty chemical that is often subject to volatile pricing and strict transportation regulations due to its toxicity and odor, manufacturers can source more commodity-grade starting materials like sodium benzenesulfinate and bis(4-tolyl) disulfide. These materials are not only more stable for long-term storage but are also generally available from a wider pool of global suppliers, reducing the risk of supply disruptions. Furthermore, the operational simplicity of the process—requiring no exotic catalysts or extreme pressure conditions—means that it can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, avoiding the need for capital-intensive equipment upgrades. This flexibility allows for rapid response to market demand fluctuations, ensuring that lead times for high-purity pharmaceutical intermediates can be minimized even during periods of high industry throughput.
- Cost Reduction in Manufacturing: The economic impact of switching to this method is driven by several factors inherent to the process design. Firstly, the replacement of expensive and hazardous reagents like cyanuric chloride and N-methylmorpholine with cost-effective N-halosuccinimides reduces the direct material cost per kilogram of product. Secondly, the elimination of chlorinated solvents like dichloromethane in favor of recoverable solvents like acetonitrile significantly lowers waste disposal costs, which constitute a major portion of operating expenses in fine chemical manufacturing. The high selectivity of the reaction minimizes the formation of hard-to-remove byproducts, thereby reducing the consumption of purification media such as silica gel or activated carbon and shortening the overall cycle time per batch. Additionally, the mild reaction conditions result in lower energy consumption for heating and cooling, contributing to a leaner manufacturing cost structure that enhances competitiveness in the global market for agrochemical and pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the stability and ease of handling of the new raw materials. Bis(4-tolyl) disulfide is a solid with a high melting point and low vapor pressure, making it far easier to transport and store than liquid thiols which require nitrogen blanketing and specialized drumming to prevent oxidation and odor leakage. This stability reduces the incidence of shipping delays caused by hazardous material compliance issues and lowers the insurance premiums associated with raw material inventory. Moreover, the robustness of the reaction conditions, which tolerate a wide range of temperatures and solvent qualities, means that production is less susceptible to minor variations in utility supply or raw material grades. This reliability ensures consistent output quality and volume, allowing downstream customers to plan their own production schedules with greater confidence and reducing the need for excessive safety stock holdings.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions continue to tighten globally, this synthesis method offers a clear pathway to sustainable manufacturing. The process generates significantly less hazardous waste, primarily consisting of aqueous salt solutions and succinimide, which are biodegradable or easily treatable compared to the heavy metal sludge or chlorinated organic waste generated by older methods. The absence of malodorous thiols eliminates the need for complex scrubbing systems to control air emissions, simplifying facility permitting and community relations. From a scalability perspective, the reaction kinetics are well-suited for large-scale batch or continuous flow processing; the exotherm is manageable, and the viscosity of the reaction mixture remains low enough for efficient agitation even at high concentrations. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to increase capacity from pilot scale to multi-ton production without encountering the mass transfer or heat removal limitations that often plague sulfur chemistry processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses cover critical aspects ranging from raw material substitution to process optimization parameters, ensuring that stakeholders have a comprehensive view of the technology's capabilities and limitations before committing to development resources.
Q: Why is the new disulfide-based method safer than traditional thiol routes?
A: Traditional methods utilize p-methylthiophenol, which is highly toxic, volatile, and possesses a foul odor, requiring specialized containment equipment. The patented method replaces this hazardous thiol with bis(4-tolyl) disulfide, a stable solid that significantly reduces occupational health risks and environmental emissions during production.
Q: What are the optimal reaction conditions for maximizing yield?
A: According to patent data, the optimal molar ratio of disulfide to sulfinate to N-halosuccinimide is approximately 1:4.0:2.0. Using acetonitrile as the solvent at temperatures between 20-50°C for 5-10 hours typically yields the highest purity and conversion rates, avoiding the degradation seen at extreme temperatures.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the process is designed for scalability. It operates under mild conditions (-10 to 50°C preferred) without requiring cryogenic cooling or high-pressure vessels. The use of common solvents like acetonitrile and the elimination of difficult-to-remove heavy metal catalysts simplify the downstream processing and waste treatment, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-(4-tolyl)benzenesulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is not just a regulatory requirement but a strategic imperative for maintaining competitiveness in the global fine chemicals market. Our technical team has extensively analyzed the methodology disclosed in CN102558004B and possesses the expertise to adapt this oxidative coupling protocol for diverse production scales. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and compliant with international quality standards. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities at the ppm level, guaranteeing that every batch of S-(4-tolyl)benzenesulfonate meets the exacting demands of protein chemistry and pharmaceutical synthesis applications. We are committed to delivering high-purity pharmaceutical intermediates that empower our clients' research and development pipelines.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our engineering team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this disulfide-based route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments tailored to your target timelines. By partnering with us, you gain access to a supply chain that prioritizes safety, sustainability, and cost-efficiency, positioning your organization for long-term success in the evolving landscape of chemical manufacturing.
