Advanced Synthesis of S-(4-tolyl)benzenesulfonate: Scaling High-Purity Intermediates for Global Pharma
The chemical landscape for producing critical sulfonate intermediates has evolved significantly with the disclosure of patent CN102558004A, which details a robust and efficient synthesis method for S-(4-tolyl)benzenesulfonate. This compound serves as a pivotal building block in the manufacture of biologically active compounds and polymeric materials, necessitating a production route that balances high purity with operational safety. The patented methodology replaces hazardous traditional reagents with stable disulfides and sodium benzenesulfinate, activated by N-halosuccinimide in common organic solvents. This strategic shift not only mitigates the risks associated with volatile thiols but also streamlines the downstream processing, offering a compelling value proposition for manufacturers seeking to optimize their supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of S-(4-tolyl)benzenesulfonate relied heavily on the condensation of benzenesulfonyl acid with p-methylthiophenol, a process fraught with significant industrial challenges. The primary reagent, p-methylthiophenol, is characterized by its extreme toxicity, high volatility, and intensely foul odor, which imposes stringent requirements on reactor sealing and ventilation systems. Furthermore, the use of dichloromethane as a solvent in conjunction with cyanuric chloride creates a hazardous working environment that increases the risk of occupational exposure and complicates waste disposal protocols. These factors collectively drive up the capital expenditure for safety infrastructure and elevate the operational costs, making the conventional route less attractive for large-scale commercial production where margin compression is a constant concern.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes bis(4-tolyl) disulfide and sodium benzenesulfinate as the foundational starting materials, fundamentally altering the safety and efficiency profile of the synthesis. By employing N-halosuccinimide as a promoter in solvents like acetonitrile or tetrahydrofuran, the reaction proceeds under remarkably mild conditions, typically between 20°C and 50°C, eliminating the need for energy-intensive heating or cryogenic cooling. This transition from volatile thiols to stable disulfides drastically reduces the hazard potential of the raw material inventory, allowing for simpler storage and handling procedures. The result is a streamlined process that delivers high yields, often exceeding 86%, while simultaneously lowering the barrier to entry for facilities aiming to produce this valuable intermediate without investing in specialized containment technology.
Mechanistic Insights into N-Halosuccinimide Mediated Coupling
The core of this synthetic breakthrough lies in the oxidative coupling mechanism facilitated by N-halosuccinimide, which activates the disulfide bond for nucleophilic attack by the sulfinate anion. In this transformation, the N-halosuccinimide, particularly N-bromosuccinimide, likely generates an electrophilic sulfur species or a transient sulfonyl halide intermediate that is highly susceptible to substitution. The sodium benzenesulfinate acts as a potent nucleophile, attacking the activated sulfur center to form the new sulfur-sulfur bond characteristic of the sulfonate ester. This mechanism avoids the formation of free thiol intermediates, thereby preventing the reversion to starting materials or the formation of symmetric disulfide byproducts that often plague thiol-based chemistries. The precise stoichiometry, typically maintaining a molar ratio of disulfide to sulfinate around 1:4.0, ensures that the equilibrium is driven strongly towards the desired product, minimizing side reactions.
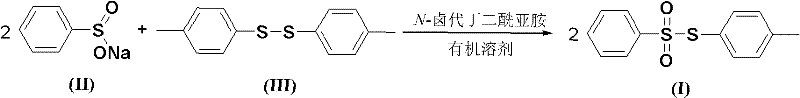
Impurity control in this system is inherently superior due to the stability of the reactants and the specificity of the N-halosuccinide activation. Unlike methods involving strong acids or bases that can lead to hydrolysis or rearrangement of the sulfonate group, this neutral to mildly acidic environment preserves the integrity of the sensitive functional groups. The use of acetonitrile as a preferred solvent further enhances purity by providing excellent solubility for the ionic sulfinate salt while allowing for easy separation of the succinimide byproduct during the aqueous workup. Consequently, the crude product often achieves purity levels above 98% prior to rigorous purification, significantly reducing the load on final crystallization steps and ensuring a consistent impurity profile that meets the stringent specifications required for pharmaceutical applications.
How to Synthesize S-(4-tolyl)benzenesulfonate Efficiently
Implementing this synthesis route requires careful attention to the addition order and temperature control to maximize the efficiency of the N-halosuccinimide promoter. The process begins by dissolving the sodium benzenesulfinate and the N-halosuccinimide in the chosen organic solvent, creating a homogeneous suspension ready for activation. A separate solution of the bis(4-tolyl) disulfide is then added dropwise to this mixture, a critical step that prevents local overheating and ensures uniform reaction kinetics throughout the vessel. Maintaining the reaction temperature within the optimal window of 20°C to 50°C for a duration of 5 to 10 hours allows the coupling to proceed to completion without degrading the product. For detailed standardized operating procedures and specific equipment configurations, please refer to the technical guide below.
- Dissolve sodium benzenesulfinate and N-halosuccinimide (preferably N-bromosuccinimide) in an organic solvent such as acetonitrile.
- Prepare a separate solution of bis(4-tolyl) disulfide in the same organic solvent and add it dropwise to the reaction mixture.
- Maintain the reaction temperature between 20°C and 50°C for 5 to 10 hours, then isolate the product via extraction and evaporation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative advantages by decoupling production capability from the availability of hazardous and regulated thiol reagents. The shift to bis(4-tolyl) disulfide and sodium benzenesulfinate leverages commodity chemicals that are widely available in the global market, reducing the risk of supply disruptions caused by environmental regulations on volatile organic compounds. This stability in raw material sourcing translates directly into more predictable lead times and reduced inventory holding costs, as the precursors do not require specialized hazmat storage facilities. Furthermore, the simplified workup procedure, which involves standard brine washing and solvent evaporation, minimizes the consumption of auxiliary chemicals and reduces the volume of hazardous waste generated, aligning perfectly with modern sustainability goals.
- Cost Reduction in Manufacturing: The elimination of p-methylthiophenol removes the substantial costs associated with its safe handling, including specialized ventilation, personal protective equipment, and hazardous waste disposal fees. Additionally, the high reaction yield and mild temperature requirements significantly lower energy consumption compared to processes requiring reflux or cryogenic conditions. The ability to recover and reuse the organic solvent, such as acetonitrile, further drives down the variable cost per kilogram, creating a leaner manufacturing model that enhances overall profit margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: By utilizing stable solid reagents like bis(4-tolyl) disulfide, the supply chain becomes more resilient to logistical challenges that often affect liquid thiols, such as leakage during transport or strict shipping restrictions. The robustness of the reaction conditions means that production can be scaled across multiple geographic locations without the need for highly customized reactor setups, ensuring continuity of supply even if one facility faces operational downtime. This flexibility allows procurement managers to diversify their supplier base and negotiate better terms, knowing that the technical barriers to entry for potential vendors are significantly lower.
- Scalability and Environmental Compliance: The process is inherently scalable due to the absence of exothermic spikes and the use of common industrial solvents, facilitating a smooth transition from pilot plant to multi-ton commercial production. The reduction in three wastes (waste water, gas, and solids) simplifies the permitting process for new production lines and ensures ongoing compliance with increasingly strict environmental regulations. This eco-friendly profile not only mitigates regulatory risk but also enhances the brand reputation of the end-product, appealing to downstream customers who prioritize green chemistry principles in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and experimental data. Understanding these nuances is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of this supply source. The answers provided reflect the specific advantages of the N-halosuccinimide mediated pathway over legacy methods, highlighting the practical benefits observed in pilot studies.
Q: Why is the new disulfide route safer than the traditional thiol method?
A: The traditional method relies on p-methylthiophenol, which is highly toxic, volatile, and possesses a foul odor, requiring specialized containment equipment. The new patent method utilizes bis(4-tolyl) disulfide, a stable solid that eliminates these severe safety hazards and reduces equipment corrosion risks.
Q: What represents the optimal reaction conditions for maximum yield?
A: According to the patent data, using acetonitrile as the solvent with N-bromosuccinimide at a temperature range of 20°C to 50°C yields the best results. Specifically, a molar ratio of 1:4.0:2.0 for disulfide, sulfinate, and N-halosuccinimide achieved yields up to 87% with purity exceeding 98%.
Q: How does this process impact environmental compliance?
A: By avoiding volatile thiols and utilizing mild reaction temperatures, the process significantly reduces the generation of hazardous waste gases. The simplified post-treatment involving standard brine washing and solvent recovery further minimizes the environmental footprint compared to complex purification steps required for thiol-based routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-(4-tolyl)benzenesulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipelines, which is why we have invested heavily in mastering advanced synthesis technologies like the one described in CN102558004A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of S-(4-tolyl)benzenesulfonate meets the exacting standards required for pharmaceutical and fine chemical applications, providing you with a partner you can trust for long-term collaboration.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer, more efficient method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering value through innovation and operational excellence in the global chemical market.
