Advanced Manufacturing Of Acesulfame K Intermediates Via Sulfamic Acid Cyclization Technology
Advanced Manufacturing Of Acesulfame K Intermediates Via Sulfamic Acid Cyclization Technology
The global demand for high-intensity sweeteners continues to drive innovation in fine chemical manufacturing, particularly for established compounds like Acesulfame K (6-methyl-3,4-dihydro-1,2,3-oxathiazine-4-one 2,2-dioxide potassium salt). Patent CN1026106C presents a transformative approach to synthesizing this critical food additive by shifting away from traditional hazardous isocyanate chemistries toward a more manageable sulfamic acid derivative pathway. This technical evolution addresses long-standing safety concerns and supply chain bottlenecks associated with the procurement of chlorosulfonyl isocyanate, offering a viable alternative for large-scale industrial production. By leveraging the reactivity of sulfamic acid salts with diketene followed by sulfur trioxide cyclization, manufacturers can achieve superior process control and consistency. This report analyzes the mechanistic advantages and commercial implications of this patented methodology for stakeholders seeking to optimize their sweetener supply chains.
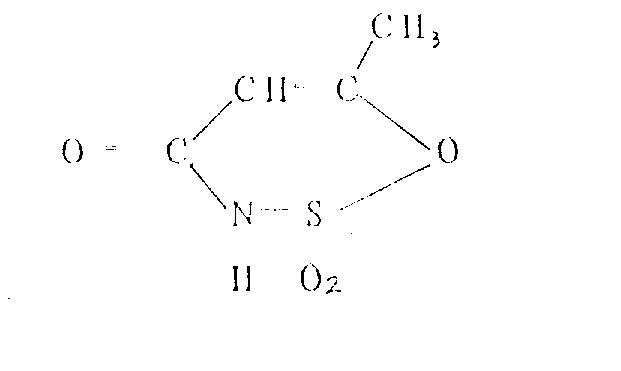
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 6-methyl-3,4-dihydro-1,2,3-oxathiazine-4-one 2,2-dioxide has relied heavily on the use of chlorosulfonyl isocyanate or fluorosulfonyl isocyanate as key starting materials. These reagents are inherently unstable and pose significant handling risks due to their extreme reactivity with moisture and potential for generating toxic byproducts during storage and transport. The synthesis of these isocyanates themselves requires hazardous precursors such as hydrogen cyanide, chlorine, and sulfur trioxide, creating a complex and regulated upstream supply chain that is vulnerable to disruptions. Furthermore, reactions involving these isocyanates often require stringent temperature controls and specialized equipment to manage exothermic events, which increases capital expenditure and operational complexity for manufacturing facilities. The reliance on such dangerous intermediates also complicates waste treatment protocols, as residual isocyanates must be meticulously quenched to prevent environmental contamination.
The Novel Approach
In contrast, the methodology described in CN1026106C utilizes sulfamic acid salts, which are readily available, stable, and significantly safer to handle than their isocyanate counterparts. This novel route initiates with the acetoacetylation of sulfamic acid derivatives using diketene in the presence of an amine or phosphine catalyst within an inert organic solvent system. This shift eliminates the need for generating or transporting hazardous isocyanates, thereby simplifying the raw material logistics and reducing the overall safety footprint of the production facility. The subsequent cyclization step employs sulfur trioxide directly on the acetoacetamide-N-sulfonate intermediate, a reaction that proceeds with high efficiency under controlled conditions. By decoupling the synthesis from the volatile isocyanate chemistry, this approach offers a more robust and scalable platform for producing high-purity sweetener intermediates suitable for sensitive food and pharmaceutical applications.
Mechanistic Insights into Sulfamic Acid Salt Acetoacetylation and Cyclization
The core of this synthetic strategy lies in the precise formation of the acetoacetamide-N-sulfonate intermediate, which serves as the precursor for the heterocyclic ring closure. In the first stage, a sulfamic acid salt, partially soluble in an inert organic solvent such as methylene dichloride or acetone, reacts with at least an equimolar quantity of diketene. The presence of a tertiary amine catalyst, such as triethylamine, facilitates the nucleophilic attack of the sulfamate nitrogen on the ketene double bond, driving the formation of the acetoacetyl linkage with yields ranging from 90% to 100%. This high conversion rate is critical for minimizing downstream purification burdens and ensuring that the subsequent cyclization step proceeds with minimal interference from unreacted starting materials. The choice of solvent is paramount, as it must dissolve the sulfamate salt sufficiently to allow reaction while remaining inert to the aggressive acetoacetylating agent.
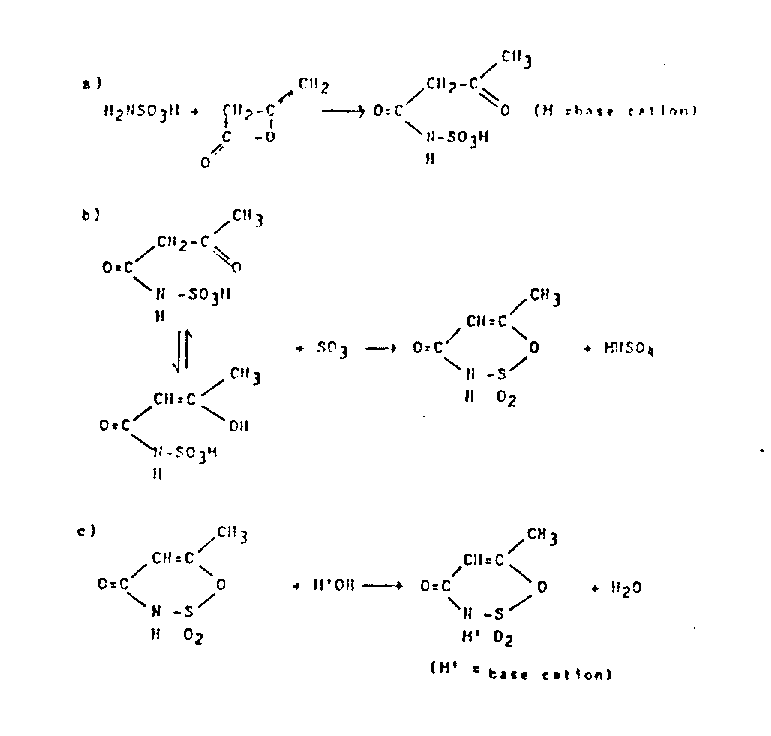
Following the isolation or in-situ generation of the acetoacetamide-N-sulfonate, the process advances to the critical cyclization step mediated by sulfur trioxide. This transformation involves the electrophilic attack of SO3 on the enol form of the acetoacetyl group, promoting intramolecular ring closure to form the oxathiazine dioxide structure. Unlike traditional dehydration methods using phosphorus pentoxide or acetic anhydride which may fail to induce cyclization effectively, the use of SO3 ensures a clean and high-yielding conversion, typically achieving 70% to 95% efficiency based on the sulfonate intermediate. The reaction mechanism avoids the formation of difficult-to-remove alkali metal salts during the ring-closing phase, allowing for a more straightforward workup procedure. Finally, the resulting free acid is neutralized with a base such as potassium hydroxide to yield the stable potassium salt, completing the synthesis with an overall process yield that significantly outperforms legacy isocyanate-based routes.
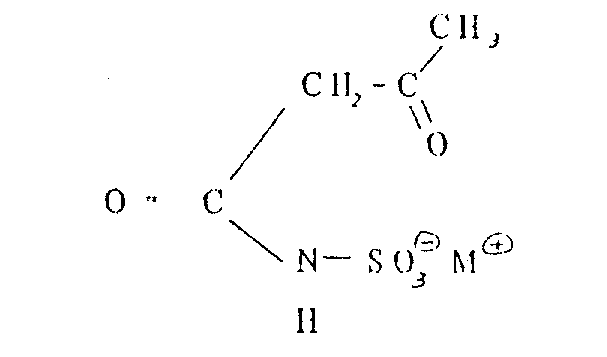
How to Synthesize Acesulfame K Efficiently
The implementation of this synthesis route requires careful attention to solvent selection, temperature control, and stoichiometric balance to maximize yield and purity. The process begins with the preparation of the sulfamic acid salt in a solvent like methylene dichloride, followed by the controlled addition of diketene at temperatures between 0°C and 25°C to manage the exotherm. Detailed standard operating procedures regarding the specific molar ratios of sulfur trioxide and the quenching protocols are essential for safe scale-up. For the complete standardized synthesis steps and specific reaction parameters, please refer to the technical guide below.
- React sulfamic acid salts with diketene in an inert organic solvent with amine catalyst to form acetoacetamide-N-sulfonate.
- Cyclize the intermediate using sulfur trioxide (SO3) in an inert solvent to form the oxathiazine dioxide ring structure.
- Neutralize the resulting acid product with potassium hydroxide to obtain the final Acesulfame K salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this sulfamic acid-based synthesis route offers substantial strategic benefits for procurement managers and supply chain directors looking to optimize their sweetener ingredient sourcing. The primary advantage stems from the replacement of hazardous isocyanates with stable sulfamic acid salts, which drastically simplifies the logistics of raw material transportation and storage. This reduction in hazard classification lowers insurance costs and removes the need for specialized containment infrastructure, leading to significant operational cost savings over the lifecycle of the production facility. Furthermore, the availability of sulfamic acid and diketene from multiple global suppliers reduces dependency on single-source vendors, thereby enhancing supply chain resilience against market fluctuations.
- Cost Reduction in Manufacturing: The elimination of chlorosulfonyl isocyanate from the process flow removes the need for expensive safety measures and waste treatment protocols associated with halogenated isocyanates. By utilizing simpler reagents and achieving higher yields in the initial acetoacetylation step, the overall consumption of raw materials per kilogram of final product is optimized. Additionally, the ability to recycle solvents like methylene dichloride between the acetoacetylation and cyclization steps further drives down variable production costs. These efficiencies translate into a more competitive cost structure for the final sweetener without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sulfamic acid is a commodity chemical with a robust global supply network, unlike specialized isocyanates which may face production constraints due to environmental regulations. This shift ensures a more consistent availability of key starting materials, reducing the risk of production stoppages caused by raw material shortages. The stability of the intermediate acetoacetamide-N-sulfonate salts also allows for potential inventory buffering, providing greater flexibility in production scheduling. Consequently, manufacturers can offer more reliable lead times to their customers, strengthening long-term contractual relationships.
- Scalability and Environmental Compliance: The process is designed to be scalable from laboratory benchtop to multi-ton commercial production without significant changes in reaction kinetics or safety profiles. The use of sulfur trioxide, while requiring care, is a well-understood industrial reagent with established handling protocols that are easier to manage than isocyanate chemistry. Moreover, the reduction in halogenated waste streams and toxic byproducts aligns with increasingly stringent environmental regulations, facilitating smoother permitting processes for new manufacturing lines. This environmental compatibility future-proofs the production asset against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and process descriptions found within the patent documentation to ensure accuracy. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing supplier capabilities.
Q: Why is the sulfamic acid route preferred over chlorosulfonyl isocyanate methods?
A: The sulfamic acid route eliminates the need for hazardous chlorosulfonyl isocyanate (CSI), significantly improving operational safety and reducing raw material procurement risks associated with highly reactive isocyanates.
Q: What yields can be expected from this cyclization process?
A: The patent data indicates that the acetoacetylation step achieves yields of approximately 90% to 100%, while the subsequent cyclization step reaches 70% to 95%, resulting in a robust overall process efficiency.
Q: How does this method impact impurity profiles in the final sweetener?
A: By utilizing specific inert organic solvents like methylene dichloride and controlling the SO3 addition rate, the process minimizes side reactions and decomposition, leading to a cleaner impurity profile suitable for food-grade applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acesulfame K Supplier
The technical potential of the sulfamic acid cyclization route represents a significant advancement in the manufacturing of high-intensity sweeteners, offering a safer and more efficient pathway to market. NINGBO INNO PHARMCHEM stands ready to leverage this chemistry as part of our comprehensive CDMO service portfolio, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications to ensure that every batch meets the exacting standards required for food and pharmaceutical applications. We understand the critical nature of supply continuity and are committed to delivering consistent quality through robust process validation.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific product pipeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer manufacturing protocol. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to enhance your supply chain efficiency and product quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
