Advanced Cefazolin Manufacturing: Enzymatic Deacylation and High-Yield Synthesis
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antibiotics like Cefazolin, a first-generation cephalosporin widely utilized for its efficacy against Gram-positive bacteria. Patent CN1084750C discloses a sophisticated four-step synthesis method that significantly enhances the purity and yield of the final active pharmaceutical ingredient. This novel approach leverages a combination of chemical nucleophilic substitution and biocatalytic deacylation, addressing the longstanding challenges of impurity control and process efficiency. By starting with a 7-protected amino-3-halogenomethyl-cephem carboxylic acid ester, the method avoids the cumbersome purification steps typically required in conventional routes. The integration of immobilized penicillin G acylase represents a pivotal technological advancement, ensuring high regioselectivity and minimizing environmental impact. For procurement and supply chain leaders, this patent data signals a viable route for securing a reliable Cefazolin supplier with enhanced production capabilities. The structural integrity of the final product, as depicted below, is maintained through careful control of reaction conditions at each stage. 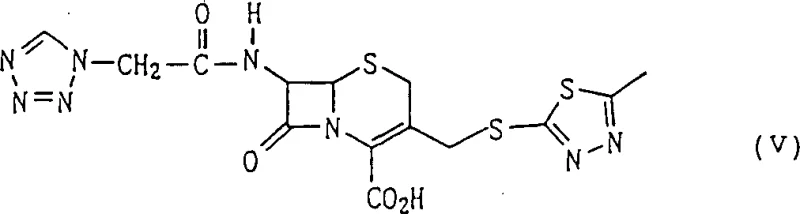
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing processes for Cefazolin often rely on 7-amino-3-acetoxy-methyl-3-cephem-4-carboxylic acid (7-ACA) as the primary starting raw material, which presents inherent inefficiencies. The conventional route requires the replacement of the 3-position acetoxy group with a 2-methyl-1,3,4-thiadiazole-5-thiol moiety, a reaction known for its low selectivity and poor yield. This chemical transformation frequently generates significant amounts of by-products, complicating the downstream purification process and negatively influencing the required degree of production purity. Furthermore, the synthetic intermediates produced via these routine methods often exhibit low stability, necessitating rigorous and costly purification protocols before proceeding to the final acylation step. The reliance on harsh chemical reagents for deacylation in older methods can also lead to the degradation of the sensitive beta-lactam ring, further reducing overall process efficiency. These technical bottlenecks result in higher production costs and inconsistent supply quality, posing risks for pharmaceutical manufacturers seeking cost reduction in API manufacturing. Consequently, the industry has long demanded a more streamlined approach that mitigates these selectivity issues.
The Novel Approach
The method disclosed in the patent introduces a paradigm shift by utilizing 7-phenylacetylamino-3-halogenomethyl-cephem carboxylic acid ester, commonly known as GCLE, as the foundational building block. This starting material allows for a direct and highly efficient nucleophilic substitution reaction at the 3-position, bypassing the problematic acetoxy replacement step entirely. As shown in the structure of the starting material, the halogen atom serves as an excellent leaving group, facilitating the introduction of the thiadiazole side chain with superior conversion rates. 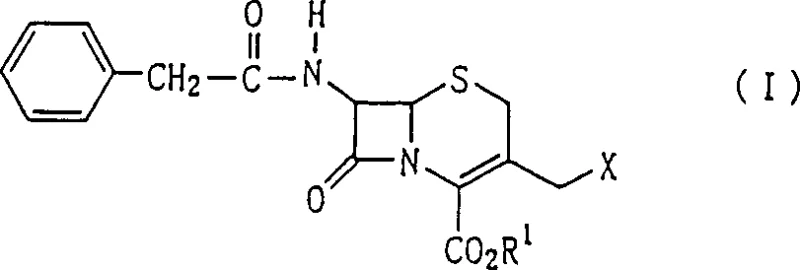 The subsequent steps involve a mild de-esterification using a phenol compound, followed by a highly specific enzymatic deacylation. This sequence ensures that the core cephem structure remains intact while the necessary functional groups are installed with precision. The novel approach effectively eliminates the need for complex intermediate purification, as the high selectivity of the reactions yields intermediates of sufficient purity for direct use in the final step. This streamlined workflow not only enhances the overall yield but also significantly simplifies the operational complexity, making it an attractive option for the commercial scale-up of complex pharmaceutical intermediates.
The subsequent steps involve a mild de-esterification using a phenol compound, followed by a highly specific enzymatic deacylation. This sequence ensures that the core cephem structure remains intact while the necessary functional groups are installed with precision. The novel approach effectively eliminates the need for complex intermediate purification, as the high selectivity of the reactions yields intermediates of sufficient purity for direct use in the final step. This streamlined workflow not only enhances the overall yield but also significantly simplifies the operational complexity, making it an attractive option for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Enzymatic Deacylation and Acylation
The core of this synthesis strategy lies in the precise manipulation of the 7-amino group protection and deprotection cycles, culminating in the final acylation. A critical mechanistic advantage is the use of immobilized penicillin G acylase (PGA) to remove the phenylacetyl protecting group from the intermediate. Unlike chemical hydrolysis, which can be non-specific and damaging to the beta-lactam ring, the enzymatic process operates under mild pH and temperature conditions, preserving the stereochemical integrity of the molecule. The enzyme specifically targets the amide bond at the 7-position, releasing the free amino group required for the final coupling without affecting the newly installed thiadiazole side chain. This biocatalytic step is crucial for achieving the high-purity Cefazolin specifications demanded by regulatory bodies. The resulting 7-amino intermediate, depicted below, is obtained in high yield and can be directly subjected to the final acylation without extensive isolation procedures. 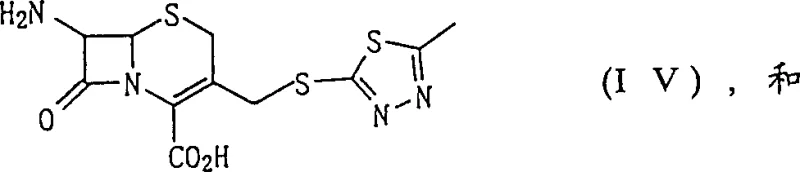 The final step involves the reaction of this amino intermediate with a mixed anhydride derived from 1H-tetrazole-1-acetic acid. The use of a mixed anhydride activates the carboxylic acid effectively, ensuring rapid and complete acylation. This mechanistic design minimizes the formation of regio-isomers and other impurities, ensuring a clean reaction profile that simplifies the final crystallization and drying processes.
The final step involves the reaction of this amino intermediate with a mixed anhydride derived from 1H-tetrazole-1-acetic acid. The use of a mixed anhydride activates the carboxylic acid effectively, ensuring rapid and complete acylation. This mechanistic design minimizes the formation of regio-isomers and other impurities, ensuring a clean reaction profile that simplifies the final crystallization and drying processes.
Impurity control is inherently built into the reaction design through the selection of high-purity starting materials and specific reagents. The use of a halogenomethyl precursor instead of an acetoxy precursor eliminates the formation of specific thioether by-products associated with the traditional route. Furthermore, the enzymatic deacylation step acts as a kinetic filter, where only the desired substrate is processed, leaving potential chemical impurities untouched and easier to remove in subsequent washes. The final acylation is performed at controlled low temperatures to prevent thermal degradation, which is a common source of impurities in cephalosporin synthesis. The process also incorporates a phenol-mediated de-esterification step that is highly selective, avoiding the use of strong acids or bases that could open the beta-lactam ring. By maintaining strict control over reaction parameters such as pH, temperature, and molar ratios, the process ensures that the impurity profile remains well within acceptable limits. This rigorous control mechanism is essential for meeting the stringent quality standards required for a reliable API intermediate supplier in the global market.
How to Synthesize Cefazolin Efficiently
The synthesis of Cefazolin via this patented route involves a sequence of four distinct chemical and enzymatic transformations that must be carefully orchestrated to maximize yield and purity. The process begins with the nucleophilic substitution of the halogen atom on the GCLE starting material, followed by de-esterification to expose the carboxylic acid. The third and most critical step involves the enzymatic removal of the 7-phenylacetyl group using immobilized PGA, which requires precise pH control to maintain enzyme activity. Finally, the free amino group is acylated using a pre-formed mixed anhydride of tetrazole acetic acid to complete the molecular structure. Detailed standardized synthesis steps, including specific solvent volumes, temperature ranges, and reaction times, are essential for replicating this high-efficiency process in a commercial setting. The following guide outlines the procedural framework necessary for successful implementation.
- React 7-phenylacetylamino-3-halogenomethyl-cephem carboxylic acid ester with 2-methyl-5-mercapto-1,3,4-thiadiazole in the presence of a base.
- Perform de-esterification of the reaction product using a phenol compound to obtain the carboxylic acid intermediate.
- Utilize immobilized penicillin G acylase to remove the phenylacetyl group, yielding the 7-amino cephalosporin intermediate.
- React the amino intermediate with a mixed anhydride of 1H-tetrazole-1-acetic acid to finalize the Cefazolin structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits regarding cost stability and production reliability. The elimination of low-yield chemical steps and the reduction of purification requirements directly translate to a more efficient use of raw materials and reduced waste generation. The use of immobilized enzymes allows for catalyst reusability, which significantly lowers the operational expenditure associated with biocatalysts over time. This process design inherently supports a more sustainable manufacturing model, aligning with modern environmental compliance standards without sacrificing output volume. By simplifying the production workflow, manufacturers can reduce the risk of batch failures and ensure a more consistent supply of high-quality product. These factors collectively contribute to a more resilient supply chain capable of meeting the demanding schedules of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive and complex purification of intermediates, which traditionally accounts for a significant portion of production costs. By achieving high yields in the initial substitution and enzymatic steps, the consumption of starting materials is optimized, leading to substantial cost savings in raw material procurement. The reusability of the immobilized enzyme further reduces the recurring cost of catalysts, providing a long-term economic advantage over chemical deacylation methods. Additionally, the simplified workflow reduces energy consumption and labor hours required for monitoring and processing, contributing to overall operational efficiency. These qualitative improvements in process economics make the manufacturing of high-purity Cefazolin more financially viable in a competitive market.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like GCLE and stable immobilized enzymes reduces the risk of supply disruptions associated with specialized reagents. The robustness of the enzymatic step ensures consistent batch-to-batch quality, minimizing the need for reprocessing or rejection of off-spec material. This consistency allows for more accurate production planning and inventory management, ensuring that delivery commitments to downstream pharmaceutical partners are met reliably. The reduced complexity of the process also means that scale-up activities can be executed with greater confidence, ensuring that supply volumes can be increased rapidly in response to market demand. This reliability is crucial for maintaining the continuity of antibiotic supply chains globally.
- Scalability and Environmental Compliance: The mild reaction conditions employed throughout the synthesis, particularly the enzymatic step, facilitate easier scale-up from pilot to commercial production without significant re-engineering. The reduction in hazardous chemical waste, due to the avoidance of harsh deacylation reagents, simplifies waste treatment and disposal, ensuring compliance with stringent environmental regulations. The process design supports the principles of green chemistry by utilizing biocatalysts and minimizing solvent usage through efficient crystallization steps. This environmental compatibility not only reduces regulatory risk but also enhances the corporate sustainability profile of the manufacturer. Such attributes are increasingly important for pharmaceutical companies seeking to partner with environmentally responsible suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented Cefazolin synthesis process. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility and advantages of adopting this manufacturing route. The information provided clarifies the mechanistic benefits and the practical implications for large-scale production.
Q: How does the enzymatic step improve purity compared to chemical deacylation?
A: The use of immobilized penicillin G acylase offers high regioselectivity, minimizing side reactions and by-product formation that are common in harsh chemical deacylation methods, thus ensuring a cleaner impurity profile.
Q: What are the advantages of using GCLE as a starting material?
A: Starting with 7-phenylacetylamino-3-halogenomethyl-cephem carboxylic acid ester (GCLE) allows for direct nucleophilic substitution at the 3-position, bypassing the low-yield acetoxy replacement steps associated with traditional 7-ACA routes.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes immobilized enzymes which are reusable and stable, and the reaction conditions are mild, making it highly scalable for industrial manufacturing with consistent quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefazolin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for essential antibiotics like Cefazolin. Our technical team has extensively analyzed this patented methodology and possesses the expertise to implement these complex pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs and advanced manufacturing facilities. Our experience in scaling diverse pathways ensures that we can adapt this high-yield process to meet your specific volume requirements while maintaining the highest quality standards. We understand the nuances of enzymatic reactions and mixed anhydride acylations, allowing us to troubleshoot and optimize the process for maximum efficiency.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced process chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments for Cefazolin and related intermediates. By partnering with us, you gain access to a reliable supply source that combines technical innovation with commercial reliability. Let us help you secure a stable and cost-effective supply of high-quality pharmaceutical ingredients for your global operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
