Advanced Aluminum Halide Deprotection Strategy for Scalable Beta-Lactam Antibiotic Manufacturing
The pharmaceutical industry constantly seeks robust methodologies for the synthesis of beta-lactam antibiotics, where the management of protecting groups is a critical bottleneck. Patent CN1113885C introduces a transformative approach for the preparation of beta-lactam derivatives, specifically addressing the deprotection of carboxyl groups without compromising the sensitive beta-lactam ring. This technology leverages aluminum halides in aliphatic ether solvents to achieve high-yield deprotection while enabling the recovery of the protecting group. For R&D directors and process chemists, this represents a significant shift away from traditional methods that rely on costly noble metals or harsh acidic conditions that degrade product quality. The ability to isolate the final carboxylic acid through simple precipitation or extraction underscores the practical utility of this invention for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the removal of carboxyl protecting groups from beta-lactam intermediates has been fraught with chemical and economic challenges. Traditional catalytic hydrogenation using noble metals is often ineffective because the sulfur atoms inherent in cephalosporin and penicillin structures act as potent catalyst poisons, necessitating excessive catalyst loading and driving up costs. Furthermore, acidolytic methods utilizing trifluoroacetic acid or formic acid require vast quantities of expensive reagents and pose significant difficulties in solvent recovery. In many cases, the harsh acidic environment leads to the decomposition of the acid-labile beta-lactam core, drastically reducing the overall yield of the active pharmaceutical ingredient. Additionally, methods employing scavengers like anisole result in the irreversible consumption of the protecting group, preventing its recycling and increasing the material cost per batch.
The Novel Approach
The methodology described in CN1113885C circumvents these issues by utilizing aluminum halides, such as aluminum trichloride, within aliphatic ether solvents like dioxane or tetrahydrofuran. This system creates a controlled Lewis acidic environment that selectively cleaves the ester bond of protecting groups like p-methoxybenzyl or diphenylmethyl without attacking the beta-lactam ring. A distinct advantage of this approach is the fate of the leaving group; instead of forming irreversible adducts, the protecting group is released as an alcohol (X-OH) which can be easily separated and converted back into the halide for reuse. This closed-loop capability significantly enhances the atom economy of the process. Moreover, the choice of ether solvents facilitates the precipitation of the product directly from the reaction mixture, allowing for isolation via simple filtration rather than complex chromatographic purification.
Mechanistic Insights into Aluminum Halide Mediated Deprotection
The core of this innovation lies in the specific interaction between the aluminum halide Lewis acid and the carbonyl oxygen of the protected ester. In the presence of aliphatic ethers, the aluminum species coordinates with the ester, enhancing the electrophilicity of the carbonyl carbon and facilitating the cleavage of the C-O bond associated with the benzyl or diphenylmethyl group. Unlike protic acids which generate high concentrations of free protons that can open the beta-lactam ring, this Lewis acid pathway offers a milder, more selective activation. The reaction conditions, typically maintained between 0°C and 50°C, are sufficiently gentle to preserve the stereochemical integrity of the chiral centers at the 6- or 7-positions of the antibiotic scaffold. This selectivity is paramount for maintaining the biological activity of the final drug substance.
Furthermore, the mechanism supports a unique impurity profile that favors high purity. Because the reaction drives the precipitation of the beta-lactam carboxylic acid product from the ether solution, the equilibrium is shifted towards completion, and soluble impurities remain in the supernatant. This phenomenon effectively acts as an in-situ purification step. The structural versatility of this method is evident in its applicability to various beta-lactam residues, including cephalosporins, penicillins, and carbacephems, as defined by the general formulas in the patent documentation. 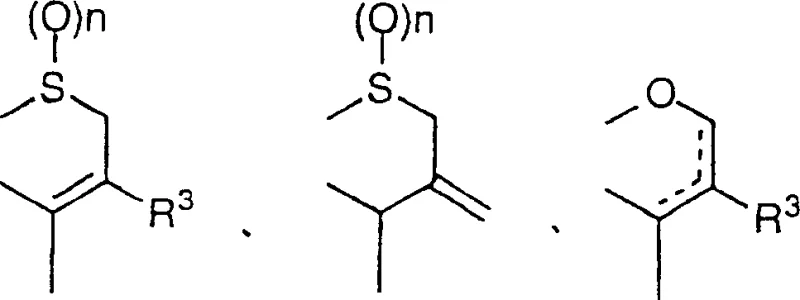
This broad substrate scope ensures that the process is not limited to a single molecule but serves as a platform technology for a wide array of antibiotic intermediates. The ability to handle diverse substituents at the 3-position of cephalosporins or the 2-position of penicillins without side reactions demonstrates the robustness of the aluminum halide system against nucleophilic attack or rearrangement.
How to Synthesize Deprotected Beta-Lactam Carboxylic Acids Efficiently
Implementing this synthesis route requires precise control over stoichiometry and temperature to maximize the recovery of both the product and the protecting group. The standard protocol involves dissolving the protected precursor in a dry ether solvent, followed by the gradual addition of the aluminum halide reagent. Maintaining the reaction within the optimal temperature window prevents thermal degradation while ensuring complete conversion. Following the reaction period, the workup procedure is notably simplified compared to traditional acidolysis; the product can often be filtered directly or extracted into an aqueous bicarbonate layer, leaving neutral organic impurities behind.
- Dissolve the carboxyl-protected beta-lactam derivative (Formula 1) in an aliphatic ether solvent such as dioxane or tetrahydrofuran.
- Add aluminum halide, preferably aluminum trichloride, to the solution at a temperature range of 0°C to 50°C and stir until the reaction is complete.
- Filter the precipitated product or perform an extraction with aqueous bicarbonate followed by acidification to isolate the pure beta-lactam carboxylic acid (Formula 2).
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the transition to this aluminum halide-based deprotection method offers substantial operational improvements over legacy technologies. The elimination of noble metal catalysts removes a major cost driver and supply risk, as the market for palladium and platinum can be volatile. Additionally, the avoidance of bulk trifluoroacetic acid reduces the handling of hazardous corrosive materials, lowering safety compliance costs and waste disposal fees. The recovery of the protecting group as a reusable alcohol further decouples the process from the continuous purchase of expensive benzylating agents, creating a more circular and cost-efficient material flow.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with commodity aluminum halides results in a drastic reduction in raw material costs. Since the catalyst does not suffer from sulfur poisoning, there is no need for excessive loading or frequent replacement, leading to significant savings in reagent expenditure. Furthermore, the ability to recover and recycle the protecting group precursor minimizes the consumption of high-value starting materials, directly improving the gross margin of the manufacturing process.
- Enhanced Supply Chain Reliability: By utilizing common aliphatic ether solvents like dioxane and THF, the process relies on widely available commodities rather than specialized or restricted reagents. This ensures a stable supply of inputs and reduces the lead time associated with sourcing niche chemicals. The simplified isolation procedure, which often allows for direct filtration, shortens the production cycle time, enabling faster turnaround for batch releases and improved responsiveness to market demand.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to methods requiring large volumes of strong acids or nitromethane. The ease of solvent recovery in ether systems reduces the environmental footprint and aligns with green chemistry principles. The robustness of the reaction conditions allows for seamless scale-up from laboratory to commercial production without the need for complex engineering controls typically required for high-pressure hydrogenation or cryogenic acid treatments.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this deprotection technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, highlighting the practical advantages for industrial application.
Q: Why is the aluminum halide method superior to noble metal catalysis for beta-lactams?
A: Conventional noble metal catalysis often suffers from catalyst poisoning due to the sulfur atoms present in beta-lactam antibiotics. The aluminum halide method avoids this issue entirely, eliminating the need for expensive precious metals and ensuring consistent reaction kinetics.
Q: Can the protecting group be recovered in this process?
A: Yes, a key advantage of this patent is the ability to recover the leaving group X as a reusable alcohol (X-OH). Unlike methods using anisole scavengers which form irreversible adducts, this process allows the protecting group precursor to be regenerated and reused.
Q: What solvents are compatible with this deprotection strategy?
A: The process specifically utilizes aliphatic ether solvents such as dioxane, dioxolane, or tetrahydrofuran. These solvents facilitate the precipitation of the product, simplifying isolation, and are easier to recover compared to nitromethane or large volumes of trifluoroacetic acid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Lactam Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the aluminum halide deprotection process to deliver high-quality pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1113885C are fully realized in a GMP environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-lactam intermediate meets the exacting standards required for downstream API synthesis.
We invite global partners to leverage our expertise for their next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis for your specific beta-lactam synthesis route. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall manufacturing costs.
