Advanced Biocatalytic Deracemization for High-Purity Chiral Alpha-Hydroxy Acids and Commercial Scalability
Advanced Biocatalytic Deracemization for High-Purity Chiral Alpha-Hydroxy Acids and Commercial Scalability
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective methods for producing optically active compounds, which serve as critical building blocks for a vast array of bioactive molecules. A significant breakthrough in this domain is detailed in patent CN102660624A, which discloses a novel method for preparing chiral alpha-hydroxy acids through a double bacteria coupled catalysis system. This technology addresses the longstanding challenges associated with the production of high-value intermediates like mandelic acid and its derivatives, offering a robust alternative to traditional chemical resolution techniques. By leveraging specific microbial strains capable of stereoselective oxidation and reduction, this process achieves exceptional conversion rates and optical purity under mild reaction conditions. For global manufacturers, understanding the nuances of this biocatalytic approach is essential for optimizing supply chains and ensuring the consistent availability of reliable pharmaceutical intermediate supplier materials that meet stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
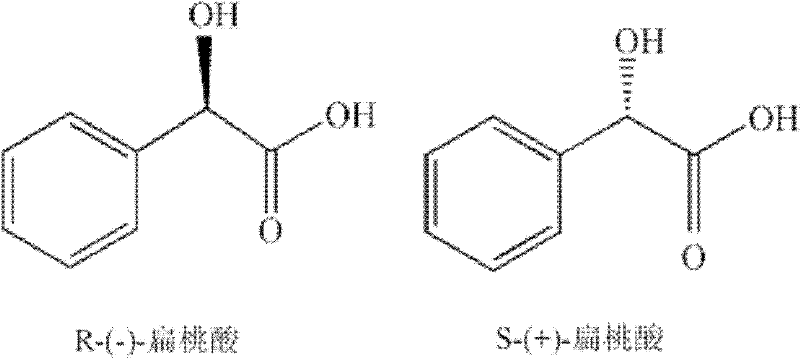
Historically, the preparation of optically active mandelic acid and its derivatives has relied heavily on the chemical resolution of racemates, a process fraught with significant economic and environmental inefficiencies. The most common method, diastereomeric salt crystallization, necessitates the use of expensive chiral resolving agents which often possess toxicity profiles that complicate downstream processing and waste management. Furthermore, this technique inherently suffers from a maximum theoretical yield of only 50% for the desired enantiomer, leading to substantial material wastage and increased raw material costs. Alternative methods such as chromatographic separation, while effective for analytical purposes, are prohibitively expensive for large-scale commercial production due to high equipment costs and low throughput capacities. Similarly, chiral extraction and capillary electrophoresis, though promising in laboratory settings, have not yet matured into viable industrial solutions, leaving a critical gap in the market for a scalable, high-yield production method that can support the growing demand for cost reduction in pharma intermediates manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the biocatalytic deracemization strategy outlined in the patent utilizes a sophisticated dual-enzyme system to dynamically convert the entire racemic mixture into a single desired enantiomer. By employing an S-selective alpha-hydroxy acid dehydrogenase alongside an R-selective keto acid reductase, the process effectively recycles the unwanted S-enantiomer into the desired R-form via a keto acid intermediate. This dynamic kinetic resolution approach theoretically allows for 100% conversion of the starting material, dramatically improving atom economy and reducing the overall carbon footprint of the synthesis. The reaction proceeds in an aqueous buffer system at moderate temperatures ranging from 20 to 50 degrees Celsius, eliminating the need for harsh organic solvents or extreme pressure conditions often required in chemical catalysis. This shift towards biocatalysis not only enhances the safety profile of the manufacturing process but also simplifies the purification workflow, thereby facilitating the commercial scale-up of complex fine chemicals with greater operational ease and reliability.
Mechanistic Insights into Dual-Bacteria Coupled Catalysis
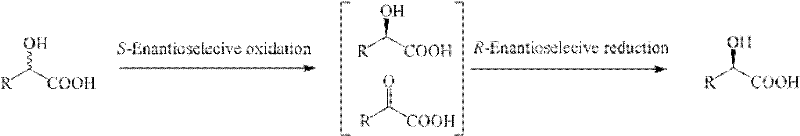
The core of this technological advancement lies in the precise orchestration of two distinct microbial activities within a single reaction vessel. The process begins with the asymmetric oxidation of the racemic alpha-hydroxy acid substrate by an S-selective dehydrogenase, which specifically targets the S-enantiomer and converts it into the corresponding alpha-keto acid. This step is crucial as it removes the unwanted isomer from the equilibrium, driving the reaction forward. Subsequently, the generated alpha-keto acid serves as the substrate for the second enzymatic activity, an R-selective keto acid reductase, which stereospecifically reduces the keto group back to a hydroxyl group, but exclusively forming the R-configured alpha-hydroxy acid. This tandem reaction sequence ensures that regardless of the initial ratio of enantiomers in the racemic feedstock, the final product pool becomes increasingly enriched with the desired R-enantiomer. The synergy between these two biocatalysts creates a self-correcting system that maximizes yield and minimizes the formation of byproducts, representing a paradigm shift in how chiral building blocks are synthesized for high-purity chiral building blocks applications.
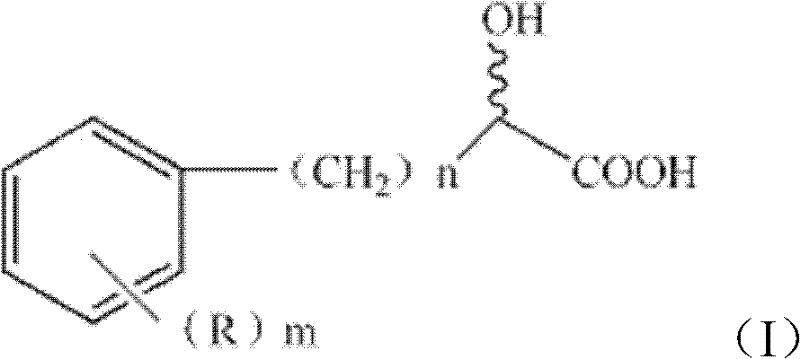
The versatility of this biocatalytic system is further demonstrated by its broad substrate scope, accommodating a wide variety of substituted alpha-hydroxy acids as defined by Formula I in the patent documentation. The method is effective for substrates where the aromatic ring bears various substituents such as hydroxyl groups, halogens like fluorine or chlorine, or alkyl chains, allowing for the production of diverse derivatives including 2-chloromandelic acid and 4-fluoromandelic acid. The tolerance of the enzyme system to these structural variations is a key factor in its commercial viability, as it enables a single platform technology to service multiple product lines within a pharmaceutical portfolio. Moreover, the inclusion of glycerol as an auxiliary substrate plays a vital role in maintaining the redox balance necessary for continuous enzymatic turnover, ensuring that the cofactors required for the oxidation and reduction steps are efficiently regenerated throughout the reaction cycle. This mechanistic robustness is essential for maintaining consistent quality and reducing lead time for high-purity active pharmaceutical ingredients in a competitive market environment.
How to Synthesize Chiral Alpha-Hydroxy Acids Efficiently
Implementing this deracemization protocol requires careful attention to the cultivation of the specific microbial strains and the optimization of the transformation conditions to ensure maximal enzymatic activity. The process involves the separate fermentation of the dehydrogenase-producing strains, such as Pseudomonas aeruginosa or Sinorhizobium sp., and the reductase-producing strain, Saccharomyces ellipsoideus, followed by their combined application in the bioconversion step. Detailed standard operating procedures regarding media composition, induction strategies, and cell harvesting are critical for reproducibility, and the patent provides specific parameters for achieving optimal wet cell mass and enzyme titer. For R&D teams looking to adopt this technology, understanding the interplay between pH control, temperature stability, and substrate concentration is paramount to scaling the process from benchtop to pilot plant without losing the high enantiomeric excess values reported in the examples.
- Cultivate specific bacterial strains including Pseudomonas aeruginosa or Sinorhizobium sp. for S-selective dehydrogenase and Saccharomyces ellipsoideus for R-selective reductase in optimized fermentation media.
- Perform the deracemization reaction by mixing the racemic substrate with the wet cell masses of both strains in a buffered solution containing glycerol as a co-substrate at 20-50°C.
- Isolate the final optically pure product by adjusting pH to 1.0, extracting with ethyl acetate, and recrystallizing the solid residue to achieve high enantiomeric purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this biocatalytic deracemization method offers transformative benefits that extend far beyond simple technical performance metrics. Traditional chemical resolution methods often rely on volatile organic solvents and stoichiometric amounts of chiral auxiliaries, which introduce significant volatility in raw material pricing and supply security. By transitioning to a fermentation-based process that utilizes renewable biological catalysts and aqueous buffers, manufacturers can decouple their production costs from the fluctuating petrochemical markets that drive the price of traditional reagents. This shift not only stabilizes the cost base but also mitigates regulatory risks associated with solvent residues and heavy metal contamination, which are increasingly scrutinized by global health authorities. Consequently, this technology empowers supply chain leaders to build more resilient sourcing strategies that are less susceptible to geopolitical disruptions or raw material shortages, ensuring a steady flow of critical intermediates for downstream drug synthesis.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and the ability to convert 100% of the racemic starting material into the desired product fundamentally alters the cost structure of chiral acid production. Traditional methods discard half of the starting material or require costly recycling loops, whereas this biological approach maximizes raw material utilization, leading to substantial cost savings in the overall manufacturing budget. Additionally, the mild reaction conditions reduce energy consumption associated with heating, cooling, and high-pressure operations, further contributing to a lower cost of goods sold. The simplified downstream processing, which avoids complex chromatographic separations, also reduces labor and equipment maintenance costs, making the final product more price-competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Reliance on specialized chemical reagents often creates single points of failure in the supply chain, especially when those reagents are sourced from limited geographic regions. In contrast, the biological catalysts used in this process can be propagated indefinitely from master cell banks, providing a self-sustaining source of catalytic activity that is not subject to the same supply constraints as synthetic chemicals. This inherent renewability of the biocatalyst ensures long-term supply continuity, allowing procurement managers to negotiate longer-term contracts with greater confidence. Furthermore, the robustness of the microbial strains against substrate variations means that the process can accommodate different grades of raw materials without significant loss of efficiency, adding another layer of flexibility to the supply chain.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent, the ability to manufacture chemicals with a reduced environmental footprint is a significant competitive advantage. This biocatalytic process generates minimal hazardous waste compared to traditional chemical resolution, which often produces large volumes of saline waste streams from salt formation and breaking steps. The use of water as the primary solvent and the biodegradability of the microbial biomass simplify waste treatment protocols and reduce the burden on effluent treatment plants. This alignment with green chemistry principles not only facilitates easier permitting for new production facilities but also enhances the corporate sustainability profile of the manufacturer, appealing to environmentally conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dual-bacteria catalytic system for chiral synthesis. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within existing manufacturing frameworks. Understanding these details is crucial for technical teams assessing the feasibility of technology transfer and for commercial teams evaluating the potential return on investment. The answers highlight the specific operational parameters and performance benchmarks that distinguish this method from conventional approaches, offering clarity on its practical application in an industrial setting.
Q: What are the primary advantages of this biocatalytic method over traditional chemical resolution?
A: Unlike traditional diastereomeric salt crystallization which requires expensive resolving agents and generates significant waste, this dual-bacteria method operates under mild conditions with yields exceeding 90% and enantiomeric excess greater than 99%, drastically reducing environmental impact and processing costs.
Q: Which specific substrates can be processed using this deracemization technology?
A: The technology is highly versatile and applicable to a broad range of alpha-hydroxy acids including mandelic acid and its halogenated derivatives such as 2-chloromandelic acid, 4-fluoromandelic acid, and phenyllactic acid, making it suitable for various pharmaceutical intermediate syntheses.
Q: How does the dual-bacteria system ensure high stereoselectivity?
A: The system couples an S-selective alpha-hydroxy acid dehydrogenase that oxidizes the unwanted S-enantiomer to a keto acid, with an R-selective keto acid reductase that reduces the keto acid back to the desired R-enantiomer, effectively recycling the unwanted isomer into the product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mandelic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to meet the evolving demands of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the biocatalytic deracemization of alpha-hydroxy acids can be successfully translated from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral intermediate meets the highest international standards for enantiomeric excess and chemical purity. Our commitment to technical excellence allows us to offer customized solutions that optimize both performance and cost, positioning us as a strategic partner for companies seeking to enhance their supply chain resilience.
We invite you to engage with our technical procurement team to discuss how this cutting-edge deracemization technology can be integrated into your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits tailored to your volume needs. We encourage you to contact us to obtain specific COA data and route feasibility assessments, enabling you to make informed decisions about upgrading your supply chain for high-purity active pharmaceutical ingredients. Let us help you navigate the complexities of chiral synthesis and secure a reliable source of high-quality intermediates for your future projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
