Scalable Microwave-Assisted Synthesis of Ellipticine for Commercial API Production
Scalable Microwave-Assisted Synthesis of Ellipticine for Commercial API Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for potent antitumor agents, and ellipticine stands out as a critical cytotoxic alkaloid with significant therapeutic potential. Patent CN111100123B introduces a transformative synthetic methodology that addresses the longstanding bottlenecks associated with producing ellipticine and its substituted derivatives. Unlike traditional laboratory-scale procedures that rely heavily on tedious purification techniques, this invention delineates a streamlined six-step reaction sequence coupled with three strategic crystallization separations. The core innovation lies in the complete elimination of column chromatography, a factor that historically hindered the commercial viability of ellipticine production. By integrating microwave-assisted heating in key cyclization steps and utilizing readily available starting materials, this process achieves a high total yield while maintaining stringent purity specifications exceeding 95 percent. For global supply chain stakeholders, this represents a pivotal shift from fragile, low-yield academic protocols to a rugged, industrial-grade manufacturing platform capable of supporting the growing demand for topoisomerase II alpha inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ellipticine has been plagued by inefficiencies that render it unsuitable for large-scale commercial production. Prior art, such as the methods reported by Deane FM et al., typically suffers from an abysmal overall yield of approximately 14 percent, primarily due to the formation of difficult-to-separate isomers and the necessity for multiple silica gel column chromatography purifications. These conventional routes often involve harsh reaction conditions and expensive, non-commodity starting materials that drive up the cost of goods significantly. Furthermore, the reliance on column chromatography introduces massive volumes of organic solvents and silica waste, creating severe environmental compliance issues and extending production lead times. The inability to purify intermediates through simple crystallization means that impurities accumulate throughout the synthetic sequence, ultimately compromising the quality of the final active pharmaceutical ingredient. As illustrated in the literature pathways, the structural complexity of the pyrido[4,3-b]carbazole core often requires multi-step protection and deprotection strategies that further erode process efficiency.
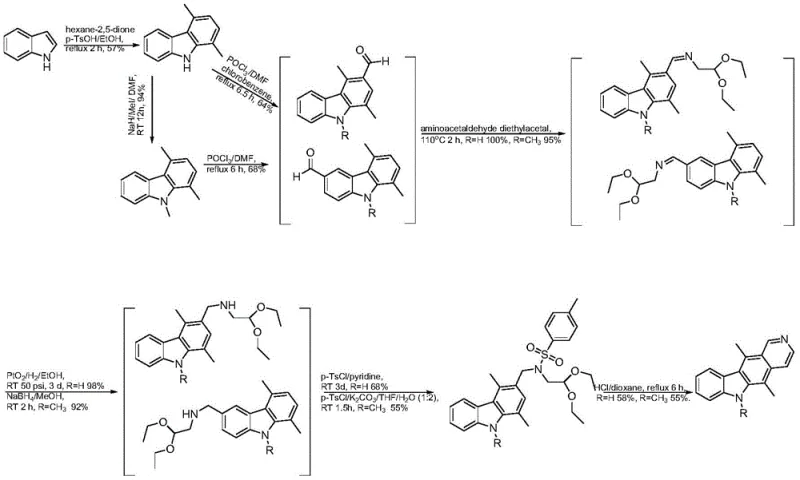
The Novel Approach
In stark contrast, the novel process disclosed in CN111100123B offers a paradigm shift by prioritizing operational simplicity and scalability from the outset. The new route leverages a microwave-assisted condensation of substituted indoles with 2,5-hexanedione to rapidly construct the carbazole scaffold with yields reaching up to 90 percent in the initial step. This is followed by a highly selective Vilsmeier-Haack formylation that installs the necessary aldehyde functionality without generating significant byproducts. Crucially, the subsequent steps involving condensation with aminoacetaldehyde diethyl acetal and final cyclization are optimized to proceed under solvent-free or microwave-enhanced conditions, drastically reducing reaction times. The strategic design allows every intermediate to be purified via recrystallization using common solvents like toluene, ethyl acetate, and ethanol, completely bypassing the need for chromatographic separation. This approach not only boosts the cumulative yield but also ensures that the process is inherently safer and more environmentally benign, aligning perfectly with modern green chemistry principles required by regulatory bodies.
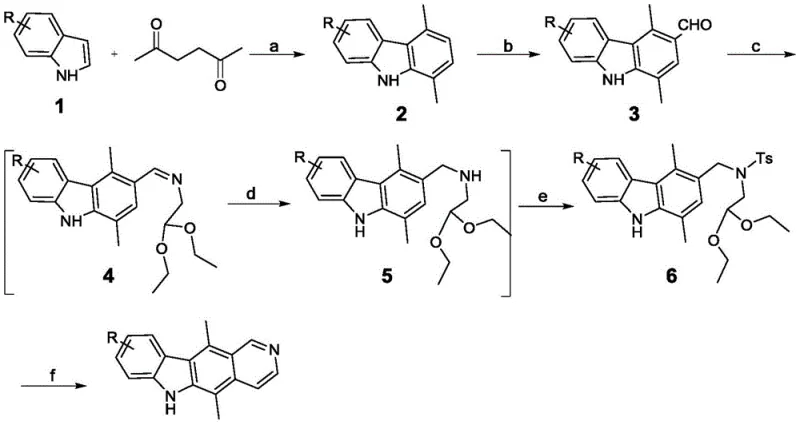
Mechanistic Insights into Microwave-Assisted Cyclization and Formylation
The success of this synthetic route hinges on the precise control of reaction kinetics, particularly during the construction of the tetracyclic core. The initial formation of the 1,4-dimethylcarbazole derivative utilizes a solid acid catalyst, specifically p-toluenesulfonic acid supported on montmorillonite, under microwave irradiation. This heterogeneous catalytic system facilitates rapid proton transfer and dehydration, driving the equilibrium towards the desired carbazole product while minimizing polymerization side reactions. The microwave energy provides uniform and instantaneous heating, which is critical for overcoming the activation energy barrier of the cyclization without subjecting the sensitive indole nucleus to prolonged thermal stress. Following this, the Vilsmeier-Haack reaction employs phosphorus oxychloride and N-methyl-N-phenylformamide to generate the reactive iminium species in situ. This electrophile attacks the electron-rich position of the carbazole ring with high regioselectivity, ensuring that the formyl group is installed exclusively at the position required for the subsequent ring closure, thereby preventing the formation of regioisomers that would complicate downstream purification.
Impurity control is meticulously managed through the physical properties of the intermediates rather than complex chemical scavenging. The patent highlights that intermediate 3, the formylated carbazole, can be recrystallized from toluene to obtain a refined, needle-like powder with high purity, effectively removing unreacted starting materials and phosphorylated byproducts. Similarly, the final cyclization step, which involves treating the tosylated precursor with hydrochloric acid under microwave conditions, is designed to be self-purifying. The resulting ellipticine base precipitates out of the acidic solution upon pH adjustment, allowing for a straightforward filtration. Any remaining organic impurities, such as unreacted amine precursors or hydrolysis products, remain soluble in the aqueous or alcoholic wash phases. This reliance on phase separation and crystallization thermodynamics ensures that the final product consistently meets the purity threshold of greater than 95 percent, as confirmed by HPLC analysis, without the need for additional polishing steps that would impact throughput.
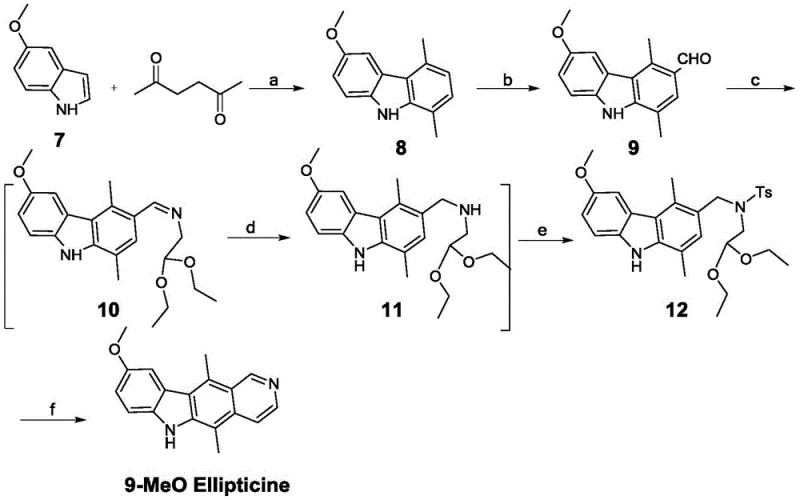
How to Synthesize Ellipticine Efficiently
The implementation of this six-step protocol requires careful attention to reaction parameters, particularly regarding microwave power and stoichiometry, to replicate the high yields reported in the patent data. The process begins with the microwave-assisted condensation, followed by formylation, acetal condensation, reduction, tosylation, and finally, acid-mediated cyclization. Each step has been optimized to minimize workup complexity, often allowing crude products to proceed directly to the next stage or requiring only simple filtration. For process chemists looking to adopt this technology, the detailed standardized synthesis steps provided below outline the specific molar ratios, solvent choices, and temperature profiles necessary to achieve consistent results. Adhering to these parameters ensures that the benefits of the microwave acceleration and crystallization-based purification are fully realized in a production environment.
- React substituted indole with 2,5-hexanedione under microwave conditions using a solid acid catalyst to form the carbazole core.
- Perform Vilsmeier-Haack formylation using POCl3 and DMF to introduce the aldehyde functionality at the specific position.
- Condense the aldehyde intermediate with aminoacetaldehyde diethyl acetal, followed by reduction and tosylation to prepare the cyclization precursor.
- Execute the final ring-closing reaction using hydrochloric acid under microwave irradiation to yield the target ellipticine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from chromatography-dependent synthesis to this crystallization-based workflow offers profound economic and logistical benefits. The primary driver of cost reduction is the complete removal of silica gel and the associated large volumes of elution solvents, which traditionally constitute a significant portion of the variable costs in alkaloid manufacturing. By replacing these with recyclable solvents like ethanol and ethyl acetate, the process drastically lowers waste disposal fees and raw material expenditure. Furthermore, the high yield of the initial carbazole formation step means that less expensive indole starting material is wasted, improving the overall atom economy of the synthesis. The ability to purify intermediates through crystallization also shortens the batch cycle time, as filtration and drying are significantly faster than column loading, elution, and solvent evaporation. This efficiency gain translates directly into increased production capacity without the need for capital investment in additional reactor volume.
- Cost Reduction in Manufacturing: The elimination of column chromatography removes the need for expensive chromatography grade solvents and disposable silica media, leading to substantial operational cost savings. Additionally, the use of commodity chemicals such as 2,5-hexanedione and phosphorus oxychloride ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with specialty reagents. The high convergence of the synthesis, where complex functionality is built early in the sequence, minimizes the loss of value-added intermediates in later stages, further optimizing the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: By utilizing robust, non-sensitive reagents and avoiding air- or moisture-sensitive catalysts that require special handling, the process enhances operational reliability and reduces the risk of batch failures. The simplified purification protocol reduces the dependency on skilled labor for column packing and fraction collection, making the process easier to transfer between manufacturing sites or contract manufacturing organizations. This standardization ensures a consistent supply of high-quality ellipticine, mitigating the risk of stockouts that can occur with erratic, low-yield legacy processes.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the physical limitations of column chromatography, which is difficult to implement at multi-ton scales. The reliance on microwave heating can be adapted using continuous flow reactors for even greater throughput. From an environmental perspective, the reduction in solvent usage and the absence of silica waste align with strict international environmental regulations, simplifying the permitting process for new manufacturing lines and reducing the carbon footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ellipticine synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on yield expectations, purity profiles, and adaptability. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this ellipticine synthesis process require column chromatography?
A: No, the patented process (CN111100123B) is specifically designed to eliminate the need for silica gel column chromatography. Purification is achieved entirely through crystallization and filtration steps, which significantly reduces solvent consumption and processing time suitable for large-scale manufacturing.
Q: What is the overall yield advantage of this new method compared to literature procedures?
A: Traditional literature methods often report overall yields as low as 14% with significant isomer formation. This novel approach utilizes optimized microwave conditions and efficient crystallization to achieve a substantially higher total yield, with individual steps like the initial carbazole formation reaching up to 90% efficiency.
Q: Can this process be adapted for substituted ellipticine derivatives?
A: Yes, the process is highly versatile. The patent explicitly demonstrates the successful synthesis of 9-methoxyellipticine using the same six-step protocol, indicating that various substituents (such as halogens, nitro, or alkoxy groups) on the starting indole can be tolerated to produce diverse analogues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ellipticine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for high-value oncology intermediates like ellipticine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering ellipticine and its derivatives with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the >95 percent purity benchmark required for downstream drug development. Our state-of-the-art facilities are equipped to handle the specific microwave and crystallization requirements of this novel process, guaranteeing a consistent and high-quality supply for our global partners.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology for their antitumor drug programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this chromatography-free route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive pricing structures that reflect the efficiencies of this next-generation manufacturing process.
