Scalable Six-Step Synthesis of Ellipticine: A Technical Breakthrough for Commercial Manufacturing
Scalable Six-Step Synthesis of Ellipticine: A Technical Breakthrough for Commercial Manufacturing
The pharmaceutical industry continuously seeks robust, scalable pathways for complex alkaloids, particularly those with potent antitumor activity like ellipticine. A significant advancement in this domain is detailed in patent CN111100123A, which discloses a simple, high-yield method for preparing ellipticine and its substituted derivatives. This technology represents a paradigm shift from traditional laboratory-scale syntheses to processes viable for industrial manufacturing. By leveraging microwave-assisted organic synthesis (MAOS) and eliminating the bottleneck of column chromatography, this route offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to secure the supply chain for oncology drug development. The process achieves high purity (>95.0%) through crystallization alone, addressing critical pain points regarding solvent consumption and operational complexity in large-scale production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ellipticine has been plagued by inefficiencies that hinder commercial viability. Prior art, such as the work by Deane FM et al., reports synthetic routes with an overall yield of merely 14% and requires rigorous purification via silica gel column chromatography to separate isomers with a 5:1 ratio. Furthermore, other literature, including methods by Modi SP and Backvall JE, describes processes that rely on harsh reaction conditions and starting materials that are not readily available or are prohibitively expensive. These conventional approaches often involve multiple protection-deprotection sequences and tedious work-up procedures that generate substantial chemical waste. The reliance on column chromatography is particularly detrimental at scale, as it limits batch sizes, increases solvent usage drastically, and introduces variability in product quality, making these methods unsuitable for the commercial scale-up of complex pharmaceutical intermediates.
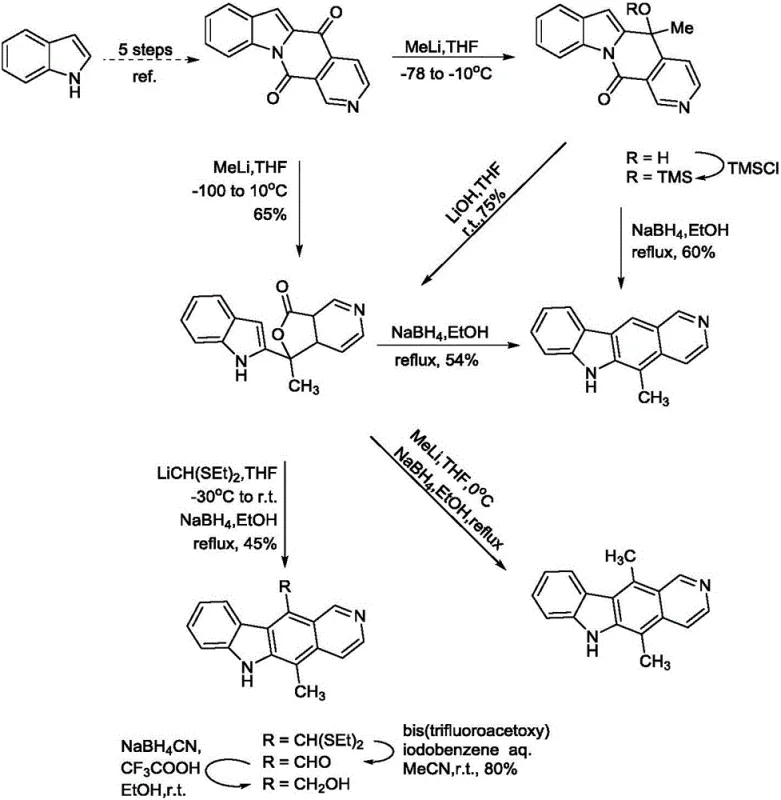
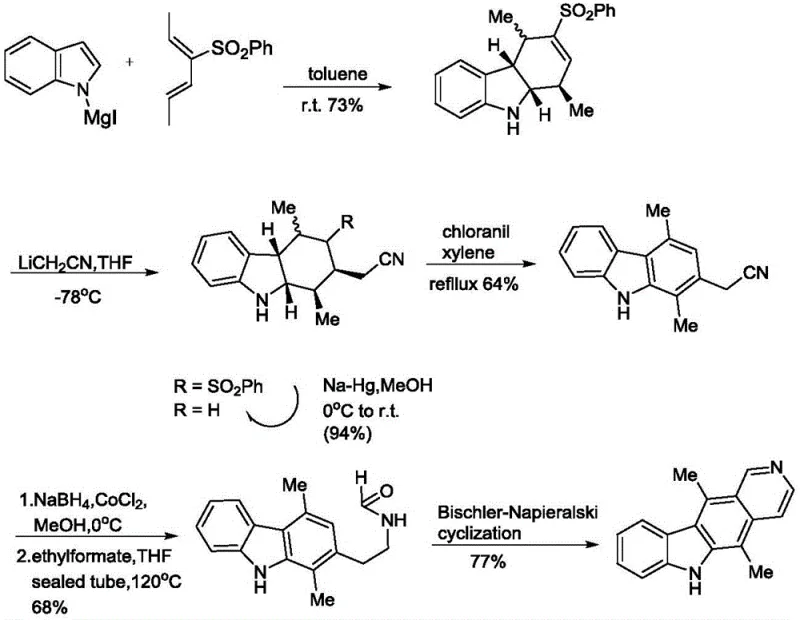
The Novel Approach
In stark contrast, the methodology outlined in patent CN111100123A streamlines the synthesis into a concise six-step sequence that prioritizes operational simplicity and yield optimization. The core innovation lies in the strategic use of microwave irradiation for both the initial carbazole formation and the final cyclization step, dramatically reducing reaction times from hours or days to mere minutes. Crucially, the process is designed to avoid column chromatography entirely; instead, intermediates are purified through straightforward recrystallization using common solvents like toluene and ethyl acetate. This approach not only simplifies the workflow but also ensures that the intermediate products and the target ellipticine are obtained with high purity suitable for downstream applications. The versatility of this route is further demonstrated by its ability to accommodate various substituents, enabling the efficient production of derivatives like 9-methoxyellipticine without compromising the overall efficiency of the synthetic pathway.
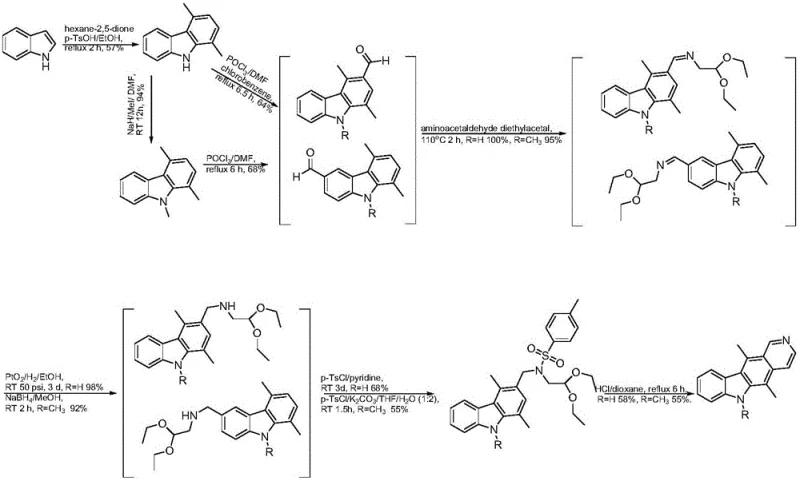
Mechanistic Insights into Microwave-Assisted Cyclization and Formylation
The success of this synthetic route hinges on two critical mechanistic features: the efficiency of the Vilsmeier-Haack formylation and the rapid kinetics of the microwave-assisted cyclization. In the second step, the reaction of the 1,4-dimethylcarbazole derivative with N-methyl-N-phenylformamide and phosphorus oxychloride (POCl3) generates the essential aldehyde intermediate. This electrophilic aromatic substitution is carefully controlled at room temperature over 24 to 48 hours to ensure regioselectivity, preventing the formation of unwanted byproducts that could complicate downstream purification. The subsequent condensation with aminoacetaldehyde diethyl acetal under solvent-free conditions at elevated temperatures (110-150°C) creates the necessary side chain for ring closure. This solvent-free approach minimizes waste and drives the equilibrium towards the desired imine intermediate, setting the stage for the final transformation.
The final ring-closing step utilizes microwave heating in the presence of hydrochloric acid to effect the Bischler-Napieralski type cyclization. Microwave energy provides uniform and rapid heating, which is essential for overcoming the activation energy barrier of the cyclization without degrading the sensitive alkaloid structure. This rapid thermal input facilitates the dehydration and aromatization required to form the pyrido[4,3-b]carbazole core of ellipticine. The mechanism ensures that the nitrogen atom is correctly incorporated into the pyridine ring, maintaining the planar structure essential for the molecule's biological activity, specifically its ability to intercalate DNA and inhibit topoisomerase IIα. By optimizing these specific reaction conditions, the process achieves a total yield that is significantly higher than historical benchmarks, validating the robustness of the chemical design.
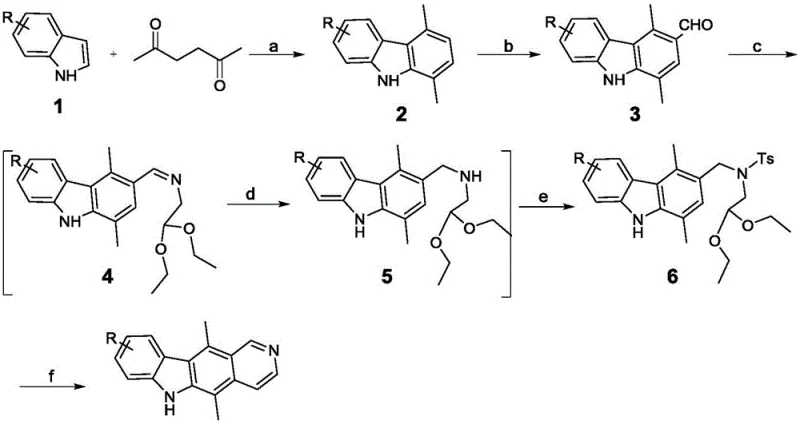
How to Synthesize Ellipticine Efficiently
The synthesis of ellipticine via this patented route involves a logical progression of functional group transformations that are amenable to standard chemical manufacturing equipment. The process begins with the condensation of indole and hexanedione, followed by formylation, side-chain installation, reduction, tosylation, and finally, microwave-assisted cyclization. Each step has been optimized to maximize yield while minimizing the need for complex purification techniques. For R&D teams looking to implement this chemistry, the detailed standardized synthetic steps provided in the patent documentation serve as a comprehensive guide for replication and optimization. The following section outlines the critical operational parameters required to achieve the reported high yields and purity specifications.
- React substituted indole with 2,5-hexanedione under microwave irradiation using a solid acid catalyst to form the carbazole core.
- Perform Vilsmeier-Haack formylation using POCl3 and N-methyl-N-phenylformamide to introduce the aldehyde group.
- Condense the aldehyde intermediate with aminoacetaldehyde diethyl acetal under solvent-free conditions followed by reduction and tosylation.
- Execute the final ring-closing cyclization using hydrochloric acid under microwave conditions to yield the target ellipticine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from column chromatography-dependent methods to this crystallization-based process offers profound economic and logistical benefits. The elimination of silica gel and the massive volumes of elution solvents associated with column chromatography directly translates to cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the use of microwave reactors for specific steps reduces energy consumption and cycle times, enhancing the throughput of the production facility. The reliance on commodity chemicals such as phosphorus oxychloride, sodium borohydride, and p-toluenesulfonyl chloride ensures that raw material sourcing is stable and not subject to the volatility of specialized reagent markets. This stability is crucial for maintaining supply chain reliability and ensuring continuous production schedules without interruption due to material shortages.
- Cost Reduction in Manufacturing: The most significant cost driver in fine chemical synthesis is often the purification process. By replacing column chromatography with recrystallization, the process drastically reduces the consumption of silica gel and high-purity organic solvents. This reduction in material usage lowers the variable cost per kilogram of the final product. Additionally, the high overall yield means that less starting material is wasted, further improving the cost efficiency of the campaign. The simplified work-up procedures also reduce labor hours and waste disposal costs, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: The synthetic route utilizes widely available starting materials and reagents that are produced on a global scale. Unlike prior art methods that may require obscure or custom-synthesized precursors, this process relies on bulk chemicals like indole, hexane-2,5-dione, and standard acids. This accessibility mitigates the risk of supply disruptions and allows for flexible sourcing strategies. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, ensuring consistent output even when sourcing from different vendors.
- Scalability and Environmental Compliance: Scaling a process that relies on column chromatography is notoriously difficult and often requires a complete redesign of the purification train for pilot or commercial plant scales. In contrast, crystallization is a unit operation that scales linearly and predictably from the gram to the tonne level. The reduced solvent load also simplifies solvent recovery and waste treatment, aiding in compliance with increasingly stringent environmental regulations. The use of microwave technology, while requiring specific equipment, offers a path to intensified processing that can reduce the physical footprint of the reactor train.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ellipticine synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN111100123A, providing clarity on purity, scalability, and derivative synthesis. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline requirements.
Q: Does this synthesis process require column chromatography for purification?
A: No, a key advantage of the process described in patent CN111100123A is that it eliminates the need for silica gel column chromatography. Instead, it utilizes recrystallization techniques (using solvents like toluene, ethyl acetate, and ethanol) to purify intermediates and the final product, which significantly enhances scalability and reduces production costs.
Q: What is the overall yield advantage of this method compared to prior art?
A: Traditional methods reported in literature often suffer from low overall yields, such as the 14% yield cited in earlier studies involving complex isomer separations. The novel six-step process achieves a high total yield through efficient microwave-assisted steps and high-yielding individual transformations, making it far superior for industrial application.
Q: Can this process be adapted for substituted ellipticine derivatives?
A: Yes, the process is highly versatile. The patent explicitly demonstrates the successful synthesis of 9-methoxyellipticine by starting with 5-methoxyindole. The methodology accommodates various substituents (R = hydrogen, OH, OCH3, formyl, nitro, halogen) on the indole ring, allowing for the production of diverse analogues for structure-activity relationship studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ellipticine Supplier
The technical advantages of the six-step microwave-assisted synthesis position ellipticine as a viable candidate for large-scale production, yet translating patent chemistry into commercial reality requires specialized expertise. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless. Our facilities are equipped with advanced microwave reactors and crystallization units capable of handling the specific thermal and purification demands of this process. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ellipticine meets the high standards required for pharmaceutical applications, including oncology research and drug development.
We invite potential partners to engage with our technical team to explore how this optimized synthesis can benefit your specific projects. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your supply of high-purity ellipticine is secure, cost-effective, and aligned with your long-term strategic goals.
