Revolutionizing Amide Synthesis: Rare Earth Catalysis for High-Purity Pharmaceutical Intermediates at Commercial Scale
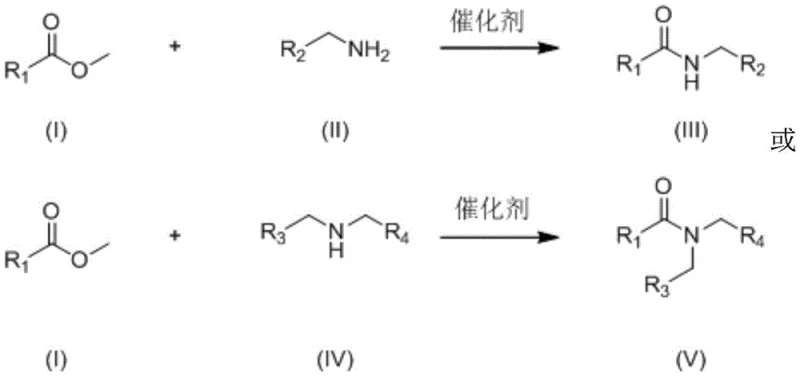
Patent CN107915653B introduces a groundbreaking method for synthesizing amides through the catalytic reaction of esters and amines using alkoxy rare earth metal cluster compounds containing alkali metal sodium. This innovative approach eliminates the need for solvents while achieving high yields across a broad substrate scope representing a significant advancement in green chemistry for pharmaceutical intermediate production. The technology addresses critical industry challenges in amide synthesis by providing a more efficient environmentally friendly and scalable alternative to conventional methods that typically require stoichiometric amounts of base and generate substantial waste. With reaction conditions operating between 40-80°C and completion within 3-12 hours this method offers pharmaceutical manufacturers a compelling solution for producing high-purity amide intermediates with enhanced process economics and reduced environmental impact. The patent demonstrates exceptional versatility across diverse ester and amine substrates making it particularly valuable for the synthesis of complex pharmaceutical building blocks where purity and yield are paramount concerns for R&D directors and supply chain managers alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for amide synthesis typically rely on the use of acyl halides or acid anhydrides with stoichiometric amounts of base generating significant waste streams that require extensive purification and disposal procedures. These conventional approaches often suffer from limited substrate compatibility particularly with sensitive functional groups commonly found in pharmaceutical intermediates leading to reduced yields and increased impurity profiles that complicate downstream processing. The requirement for large quantities of organic or inorganic bases not only increases raw material costs but also necessitates additional neutralization and waste treatment steps substantially extending production timelines and increasing the environmental footprint of the manufacturing process. Furthermore many existing catalytic systems require specialized solvents that must be carefully removed post-reaction adding complexity to the purification process and potentially introducing solvent residues that compromise product purity requirements for pharmaceutical applications. The narrow substrate scope of conventional methods also restricts their applicability in the synthesis of structurally diverse amide-containing compounds forcing manufacturers to develop multiple specialized routes for different product families rather than implementing a unified scalable production platform.
The Novel Approach
The patented method overcomes these limitations through the innovative use of alkoxy rare earth metal cluster compounds containing alkali metal sodium as catalysts enabling direct reaction between esters and amines without solvent addition. This breakthrough approach operates under mild conditions (40-80°C) with remarkably low catalyst loading (0.0025-0.01 mol%) achieving high yields (up to 99%) across an exceptionally broad substrate scope that includes various substituted benzoates cinnamates and heterocyclic esters with diverse primary and secondary amines. The solvent-free nature of the reaction eliminates the need for solvent removal steps significantly reducing processing time and energy consumption while avoiding potential solvent contamination issues that could compromise product purity. The catalyst system demonstrates excellent functional group tolerance maintaining high efficiency even with substrates containing halogens trifluoromethyl groups and heterocyclic moieties that typically pose challenges for conventional methods. This novel approach not only simplifies the manufacturing process but also enhances sustainability by generating alcohol as the sole byproduct aligning perfectly with green chemistry principles while delivering superior process economics through reduced raw material consumption and waste generation.
Mechanistic Insights into Rare Earth Metal Cluster Catalysis
The catalytic mechanism centers on the unique structure of the alkoxy rare earth metal cluster compounds containing alkali metal sodium which activates both the ester carbonyl group and the amine nucleophile through a cooperative dual activation pathway. In the presence of La2Na8(OCH2CF3)14(THF)6 the rare earth metal centers coordinate with the ester oxygen while the sodium cations interact with the amine nitrogen lowering the activation energy barrier for nucleophilic attack. This dual activation creates a highly organized transition state that facilitates direct alcohol elimination without requiring additional base explaining the remarkable efficiency observed even at low catalyst loadings. The cluster structure provides multiple active sites that work in concert to stabilize key intermediates preventing undesired side reactions and ensuring high selectivity toward the desired amide product. The precise geometric arrangement within the cluster catalyst also enables optimal positioning of reactants which is particularly advantageous for sterically demanding substrates that typically show poor reactivity in conventional systems.
The mechanism inherently minimizes impurity formation through its highly selective activation pathway which avoids common side reactions such as ester hydrolysis or amine oxidation that plague traditional methods requiring strong bases or harsh conditions. The absence of solvent eliminates potential solvent-derived impurities while the mild reaction temperature (40-80°C) prevents thermal degradation pathways that could generate colored impurities or decomposition products. The catalyst's ability to operate effectively across diverse substrate combinations without requiring adjustment of reaction parameters ensures consistent product quality regardless of structural variations in starting materials addressing a critical concern for pharmaceutical manufacturers who must maintain stringent purity specifications across multiple product lines. This inherent selectivity translates directly to reduced purification burden and higher overall process efficiency making it particularly valuable for producing high-purity pharmaceutical intermediates where impurity profiles directly impact final drug quality and regulatory approval.
How to Synthesize Amides Efficiently
This patented synthesis route represents a significant advancement in amide production methodology offering pharmaceutical manufacturers a streamlined pathway to high-purity intermediates with exceptional yield and substrate flexibility. The process eliminates traditional limitations through its innovative catalyst system and solvent-free operation providing a robust foundation for commercial-scale manufacturing of complex amide-containing compounds. Detailed standardized synthesis procedures have been developed based on extensive optimization studies documented in the patent ensuring consistent results across diverse production scales. The following step-by-step guide outlines the essential protocol for implementing this technology in manufacturing environments with specific parameters tailored to maximize yield and purity while minimizing processing time.
- Prepare reaction mixture with methyl ester and amine at molar ratio of 1: 1.2 under anhydrous conditions
- Add La₂Na₈(OCH₂CF₃)₁₄(THF)₆ catalyst at 0.5 mol% concentration under argon atmosphere
- Heat mixture to 80°C for 6 hours without solvent addition followed by column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative catalytic approach delivers substantial commercial benefits that directly address critical pain points in pharmaceutical intermediate manufacturing offering procurement and supply chain teams a compelling solution for enhancing operational efficiency while maintaining stringent quality standards. The elimination of solvent requirements and reduction in processing steps translate to significant operational improvements that impact both cost structure and supply chain resilience. By simplifying the manufacturing workflow while expanding substrate compatibility this technology provides manufacturers with greater flexibility to respond to changing market demands without requiring major process revalidation or equipment modifications.
- Cost Reduction in Manufacturing: The solvent-free process eliminates substantial costs associated with solvent purchase handling recovery and disposal while reducing energy consumption through simplified processing workflows. The highly efficient catalyst system operates at low loadings (0.5 mol%) with excellent recyclability potential significantly reducing raw material costs compared to conventional methods requiring stoichiometric amounts of expensive reagents. The elimination of base addition steps removes associated neutralization requirements and waste treatment costs creating substantial cost savings through process intensification without compromising product quality or yield.
- Enhanced Supply Chain Reliability: The use of readily available starting materials (methyl esters and amines) with broad commercial availability reduces dependency on specialized or restricted reagents that often cause supply chain disruptions. The simplified process with fewer unit operations minimizes potential failure points in manufacturing enhancing production reliability and consistency across batches. The robust nature of the catalytic system maintains high performance across diverse substrate combinations without requiring significant process adjustments providing manufacturers with greater flexibility to accommodate changing raw material availability while maintaining consistent output quality.
- Scalability and Environmental Compliance: The straightforward reaction profile without complex workup procedures enables seamless scale-up from laboratory to commercial production volumes while maintaining consistent yield and purity profiles. The environmentally benign nature of the process (generating only alcohol as byproduct) aligns with increasingly stringent regulatory requirements for sustainable manufacturing practices reducing compliance risks and potential future retrofitting costs. The elimination of hazardous solvents and reagents simplifies waste management protocols while improving workplace safety contributing to overall operational sustainability without sacrificing manufacturing efficiency.
Frequently Asked Questions (FAQ)
The following questions address common concerns from procurement R&D and supply chain professionals regarding implementation of this patented technology in commercial manufacturing environments. Each response is grounded in specific technical details documented in patent CN107915653B providing evidence-based insights into process performance scalability considerations and quality assurance aspects critical for decision-making in pharmaceutical intermediate production.
Q: How does this rare earth metal cluster catalysis improve upon traditional amide synthesis methods?
A: The rare earth metal cluster catalyst enables solvent-free reaction conditions with higher yields up to 99% and broader substrate scope compared to conventional methods requiring stoichiometric base amounts and generating substantial waste streams
Q: What are the key advantages of the solvent-free process for commercial manufacturing?
A: The solvent-free process eliminates solvent removal and recovery steps significantly reducing processing time energy consumption and waste generation while maintaining high product purity essential for pharmaceutical applications
Q: How does this technology address supply chain challenges for pharmaceutical intermediates?
A: The simplified process with readily available starting materials enhances supply chain reliability by reducing dependency on specialized reagents enabling consistent production of high-purity amide intermediates across diverse structural variants
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex amide intermediates using this patented rare earth metal cluster catalysis technology. Our stringent purity specifications and rigorous QC labs ensure consistent delivery of high-quality products that meet exacting pharmaceutical standards while leveraging the process advantages documented in CN107915653B for optimal cost-effectiveness. With dedicated technical teams experienced in implementing solvent-free catalytic processes at commercial scale we provide comprehensive support from route validation through full-scale production ensuring seamless technology transfer and reliable supply chain performance for your critical intermediate needs.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your requirements. Take advantage of our Customized Cost-Saving Analysis service to evaluate how this innovative technology can optimize your amide intermediate supply chain while maintaining uncompromising quality standards essential for pharmaceutical applications.
