Advanced Enzymatic Resolution for High-Purity Chiral Alcohol Intermediates
Advanced Enzymatic Resolution for High-Purity Chiral Alcohol Intermediates
The pharmaceutical industry continuously seeks robust and scalable methodologies for producing chiral building blocks, which are critical for the synthesis of active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN108192932B, which discloses an enzyme-catalyzed preparation method for chiral alcohols. This technology addresses the longstanding challenges associated with synthesizing key intermediates for drugs such as myeloid leukemia 1 (MCL1) inhibitors. Unlike traditional chemical resolutions that often suffer from harsh conditions and low efficiency, this novel approach leverages the specificity of biological catalysts to achieve exceptional stereocontrol. The process utilizes a screened lipase, specifically PS-SD, to kinetically resolve racemic precursors into high-value enantiomers with outstanding purity and yield. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more cost-effective manufacturing strategies that align with modern sustainability goals while ensuring a reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alcohol intermediates, particularly those required for MCL1 inhibitors, has relied on cumbersome chemical pathways that pose significant operational risks and economic burdens. Prior art methods, such as those described in Nature (2016) and WO2016207225, typically involve a multi-step sequence starting with a Wohl-Ziegler reaction to generate a brominated species, followed by a Grignard reaction with ethyl glyoxylate. These conventional routes are plagued by severe limitations, including the strict requirement for anhydrous conditions during the Grignard step, which demands specialized equipment and increases safety hazards. Furthermore, the overall yield of the racemate in these two-step processes is dismally low, often reported around only 23%. To obtain the single-configuration target product, manufacturers must resort to chiral chromatographic column separation, a technique that is notoriously expensive, difficult to scale, and results in substantial material loss. These factors collectively render traditional chemical synthesis unsuitable for industrial amplification, creating bottlenecks in the supply chain for critical drug substances.
The Novel Approach
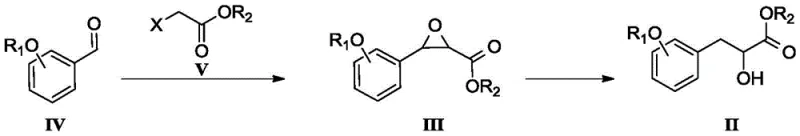
In stark contrast to the inefficiencies of legacy methods, the enzymatic preparation method disclosed in the patent offers a streamlined and highly efficient alternative. The new strategy involves the synthesis of a racemic precursor (Compound II) via a base-catalyzed reaction between a substituted benzaldehyde (Compound IV) and an alpha-halo ester (Compound V), followed by a palladium-catalyzed ring-opening hydrogenation. This chemical pre-step is robust and avoids the extreme sensitivity of Grignard reagents. The true innovation lies in the subsequent enzymatic resolution step, where the racemic mixture is subjected to kinetic resolution using PS-SD lipase. This biological catalyst operates under mild conditions, typically between 5°C and 40°C, in a biphasic system comprising a buffer and an organic solvent. The result is a dramatic improvement in process metrics, with yields for the desired ester enantiomer reaching up to 94% and enantiomeric excess (ee) values soaring to 99.79%. This approach not only simplifies post-treatment but also eliminates the need for costly chiral columns, marking a substantial advancement in cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into PS-SD Lipase Catalyzed Resolution
The core of this technological advancement is the precise mechanistic action of the PS-SD lipase within a carefully controlled biphasic reaction system. The enzyme functions by selectively recognizing and hydrolyzing the ester bond of one specific enantiomer of the racemic substrate (Compound II), converting it into the corresponding carboxylic acid (Compound Ib), while leaving the other enantiomer (Compound Ia) untouched as an ester. This kinetic resolution is highly dependent on the micro-environment of the enzyme, which is maintained by a phosphate buffer system with a pH strictly controlled between 6.5 and 8.5, preferably 7.0 to 8.0. During the reaction, the pH is dynamically managed by the supplementary addition of sodium or potassium hydroxide to neutralize the acid formed, ensuring the enzyme remains in its optimal active state throughout the process. The choice of organic solvent is also critical; solvents such as tert-butyl methyl ether, toluene, or n-hexane are employed to dissolve the hydrophobic substrate while maintaining enzyme stability at the interface. This delicate balance allows for the continuous conversion of the substrate without denaturing the biocatalyst, facilitating a clean separation of the two enantiomers based on their differing chemical properties (acid vs. ester) post-reaction.
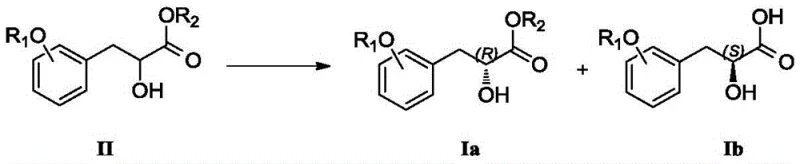
Furthermore, the impurity profile of the final product is significantly improved due to the high specificity of the enzymatic reaction. Unlike chemical catalysts that may promote side reactions such as elimination or over-reduction, the lipase exhibits exquisite chemoselectivity, targeting only the ester functionality. The post-treatment process further enhances purity; the reaction mixture is filtered to remove the immobilized enzyme, and the phases are separated. The organic phase containing the unreacted ester (Compound Ia) is washed and concentrated, while the aqueous phase containing the acid product (Compound Ib) is acidified and extracted. Both crude products can be purified simply by pulping in non-polar solvents like petroleum ether or n-hexane, avoiding the need for complex column chromatography. This mechanism ensures that the final compounds possess purities greater than 98% and high optical purity, meeting the stringent quality standards required for reliable pharmaceutical intermediate suppliers.
How to Synthesize Chiral Alcohol Efficiently
The synthesis of these high-value chiral alcohols is designed to be operationally simple yet chemically rigorous, making it accessible for adoption in standard pilot and production facilities. The process begins with the preparation of the racemic precursor, which can be executed in a telescoped manner to minimize handling time. Once the precursor is secured, the enzymatic resolution is performed in a stirred tank reactor equipped with pH control capabilities. The reaction parameters, including temperature and enzyme loading (0.015-0.06 g/g of substrate), are optimized to maximize throughput while maintaining high stereoselectivity. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and work-up procedures derived from the patent examples, are outlined below to guide process engineers in replicating this high-efficiency route.
- Synthesize the racemic precursor (Compound II) by reacting a substituted benzaldehyde (Compound IV) with an alpha-halo ester (Compound V) under alkaline conditions to form an epoxide intermediate (Compound III), followed by catalytic hydrogenation to open the ring.
- Prepare the enzymatic reaction system by dispersing PS-SD lipase into a phosphate buffer solution (pH 6.5-8.5) under inert gas protection, ensuring stable enzyme activity.
- Add the racemic precursor dissolved in an organic solvent (e.g., tert-butyl methyl ether) to the enzyme mixture, maintain temperature between 5-40°C, and control pH during reaction to separate the desired (R)-ester and (S)-acid enantiomers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic methodology offers profound strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the production workflow, which directly translates to enhanced supply chain reliability and reduced lead times. By eliminating the dependency on chiral chromatography—a process often limited by column capacity and resin costs—manufacturers can significantly increase batch sizes and throughput. This scalability ensures a consistent and uninterrupted supply of critical intermediates, mitigating the risk of production delays that can plague complex synthetic routes. Moreover, the use of commercially available and economical enzymes, combined with common organic solvents, reduces the vulnerability of the supply chain to fluctuations in the availability of exotic reagents or specialized catalysts.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive purification steps and the improvement in overall yield. Traditional methods that rely on chiral columns incur high operational expenditures due to the cost of chiral stationary phases and the low recovery rates associated with chromatographic separation. In contrast, the enzymatic route achieves high yields (up to 94% for the ester and 82% for the acid) and high purity through simple crystallization or pulping. Additionally, the mild reaction conditions (ambient pressure and moderate temperatures) reduce energy consumption compared to high-pressure or cryogenic chemical processes. The removal of heavy metal catalysts from the final resolution step also lowers the cost associated with metal scavenging and waste disposal, contributing to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the enzymatic process contributes to a more resilient supply chain. The reagents used, such as substituted benzaldehydes and alpha-halo esters, are commodity chemicals that are readily available from multiple global sources, reducing the risk of single-source bottlenecks. The enzyme itself, PS-SD, is a stable, commercially available biocatalyst that does not require complex cold-chain logistics for short-term storage. The simplicity of the work-up procedure, which involves standard liquid-liquid extraction and filtration, means that the process can be easily transferred between different manufacturing sites without the need for highly specialized equipment. This flexibility ensures that production can be scaled or shifted rapidly in response to market demand, securing the continuity of supply for downstream API manufacturing.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this green chemistry approach aligns perfectly with modern sustainability mandates. The process operates in a biphasic system that minimizes the volume of organic solvents required, and the aqueous waste streams are generally less hazardous than those generated by heavy metal-catalyzed reactions. The high atom economy and reduced waste generation simplify the treatment of effluents, lowering the environmental compliance burden. Furthermore, the patent explicitly highlights the suitability of this method for industrial amplification, indicating that the kinetics and mass transfer characteristics are favorable for large-scale reactors. This scalability allows manufacturers to meet growing global demand for chiral drugs without compromising on environmental standards or facing regulatory hurdles associated with toxic waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the advantages of this enzymatic method over traditional chiral chromatography?
A: Traditional methods often rely on chiral chromatographic columns which are expensive, have low loading capacities, and result in low overall yields (around 23% in prior art). This enzymatic method achieves yields up to 94% with ee values exceeding 99%, significantly reducing production costs and eliminating the need for complex column separation equipment.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the process is suitable for scale-up production. It utilizes mild reaction conditions (5-40°C), common solvents, and robust enzyme catalysts that do not require strict anhydrous environments, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Q: How is the stereoselectivity controlled in this reaction?
A: Stereoselectivity is achieved through the specific substrate recognition of the PS-SD lipase. By carefully controlling the pH of the buffer system (preferably 7.0-8.0) and the reaction temperature, the enzyme selectively hydrolyzes one enantiomer while leaving the other intact, allowing for easy physical separation of the high-purity chiral products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic resolution technology described in patent CN108192932B for the production of high-purity chiral alcohols. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped with advanced bioreactors capable of maintaining the precise pH and temperature controls required for optimal lipase activity. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral intermediate meets the highest international standards, providing our partners with the confidence needed to advance their drug development programs.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains using this cutting-edge technology. By leveraging our expertise in biocatalysis and process chemistry, we can help you achieve significant efficiencies in your manufacturing operations. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. Our team is ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can support your journey from bench to market with reliable, high-quality chiral intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
