Scalable One-Step Synthesis of Chiral Oxazoline Ligands for Asymmetric Catalysis
The landscape of asymmetric synthesis relies heavily on the availability of high-performance chiral ligands, and the technology disclosed in patent CN101519384A represents a significant advancement in the efficient production of chiral oxazoline derivatives. This intellectual property outlines a robust, one-step methodology for constructing bis-oxazoline scaffolds, which are critical components in the formation of metal-organic complexes used for enantioselective transformations such as the asymmetric synthesis of mandelic acid. For R&D directors and procurement specialists in the fine chemical sector, understanding the nuances of this synthetic route is essential for securing a reliable chiral ligand intermediate supplier capable of delivering consistent quality. The patent details the preparation of three distinct series of compounds—designated as formulas (I), (II), and (III)—which correspond to para, meta, and ortho substitutions on the central benzene ring, offering a versatile toolkit for catalyst design.
These chiral architectures are not merely academic curiosities but serve as foundational building blocks for industrial catalysis, where the steric and electronic properties of the ligand dictate the success of downstream pharmaceutical manufacturing. By leveraging the specific reaction parameters defined in this patent, manufacturers can access a library of ligands with varying R groups, including isobutyl, isopropyl, phenyl, and benzyl moieties, allowing for fine-tuning of catalytic activity. The ability to synthesize these complex heterocycles in a single operational step from readily available dicyanobenzenes and chiral amino alcohols marks a departure from more convoluted multi-step traditions, promising enhanced efficiency in cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of bis-oxazoline ligands has often involved laborious multi-step sequences that require the protection and deprotection of functional groups, leading to accumulated yield losses and increased waste generation. Traditional pathways might involve converting carboxylic acids to amides followed by cyclodehydration, processes that frequently demand harsh reagents like thionyl chloride or phosphorus pentachloride, which pose significant safety and environmental hazards on a commercial scale. Furthermore, achieving high enantiomeric purity in conventional syntheses often necessitates rigorous purification protocols, such as repeated recrystallizations or preparative chiral HPLC, which drastically inflate the cost of goods and extend lead times for high-purity chiral ligand intermediates. The reliance on sensitive intermediates that are prone to racemization under thermal stress further complicates the scale-up process, making it difficult for supply chain heads to guarantee batch-to-batch consistency.
The Novel Approach
In stark contrast, the methodology presented in CN101519384A streamlines the entire production workflow into a direct condensation-cyclization event, eliminating the need for pre-activation of the nitrile groups. This novel approach utilizes dicyanobenzene isomers reacting directly with chiral amino alcohols in the presence of a Lewis acid catalyst, effectively merging bond formation and ring closure into a single thermal operation. 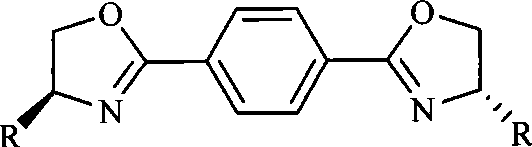 As illustrated by the structural diversity available, this single platform can generate twelve distinct high-value ligands simply by swapping the starting dicyanobenzene isomer and the chiral amino alcohol side chain. The process operates under relatively mild thermal conditions compared to pyrolytic methods, utilizing common organic solvents like chlorobenzene or xylene that are easily recovered and recycled, thereby supporting substantial cost savings and environmental compliance. This direct route significantly simplifies the supply chain by reducing the number of raw material inputs and processing units required, enhancing overall supply chain reliability for bulk production.
As illustrated by the structural diversity available, this single platform can generate twelve distinct high-value ligands simply by swapping the starting dicyanobenzene isomer and the chiral amino alcohol side chain. The process operates under relatively mild thermal conditions compared to pyrolytic methods, utilizing common organic solvents like chlorobenzene or xylene that are easily recovered and recycled, thereby supporting substantial cost savings and environmental compliance. This direct route significantly simplifies the supply chain by reducing the number of raw material inputs and processing units required, enhancing overall supply chain reliability for bulk production.
Mechanistic Insights into Lewis Acid Catalyzed Cyclization
The core of this synthetic innovation lies in the activation of the nitrile functionality by a Lewis acid catalyst, which facilitates the nucleophilic attack by the amine group of the chiral amino alcohol. 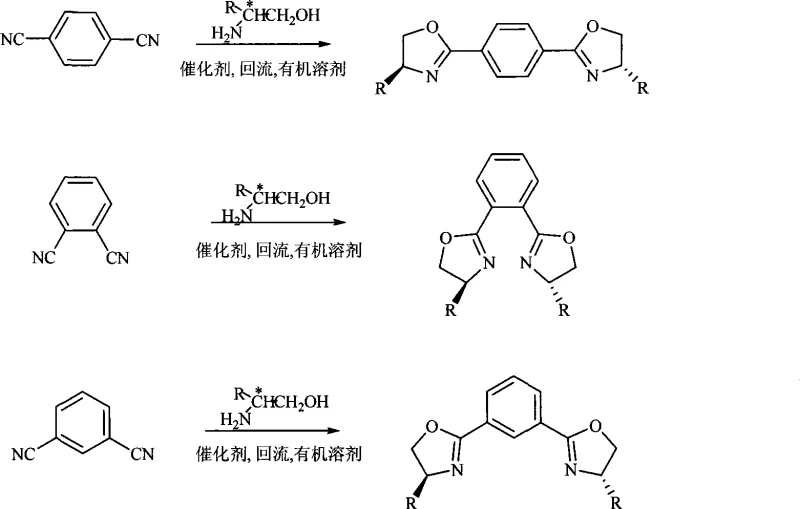 The mechanism likely proceeds through the coordination of the metal center (such as Zinc, Nickel, or Rare Earth elements) to the nitrogen lone pair of the nitrile group, increasing the electrophilicity of the nitrile carbon. This activation allows the primary amine of the amino alcohol to attack, forming an amidine intermediate which subsequently undergoes intramolecular cyclization via the hydroxyl group attacking the imine carbon. The choice of catalyst is critical; the patent highlights the efficacy of transition metal chlorides like ZnCl2 and NiCl2, as well as rare earth chlorides, which provide the optimal balance of Lewis acidity to drive the reaction forward without promoting side reactions or racemization of the chiral center. The reaction is typically conducted under anhydrous and anaerobic conditions to prevent catalyst deactivation and hydrolysis of the nitrile starting material, ensuring high conversion rates.
The mechanism likely proceeds through the coordination of the metal center (such as Zinc, Nickel, or Rare Earth elements) to the nitrogen lone pair of the nitrile group, increasing the electrophilicity of the nitrile carbon. This activation allows the primary amine of the amino alcohol to attack, forming an amidine intermediate which subsequently undergoes intramolecular cyclization via the hydroxyl group attacking the imine carbon. The choice of catalyst is critical; the patent highlights the efficacy of transition metal chlorides like ZnCl2 and NiCl2, as well as rare earth chlorides, which provide the optimal balance of Lewis acidity to drive the reaction forward without promoting side reactions or racemization of the chiral center. The reaction is typically conducted under anhydrous and anaerobic conditions to prevent catalyst deactivation and hydrolysis of the nitrile starting material, ensuring high conversion rates.
Impurity control is inherently managed by the thermodynamic stability of the oxazoline ring formed under these reflux conditions, which drives the equilibrium towards the product. The use of specific solvents like chlorobenzene or ethylbenzene, which have boiling points matching the required reaction temperature range of 110-145°C, ensures that the system remains in a liquid phase with sufficient thermal energy to overcome the activation barrier for cyclization. The stereocenter derived from the chiral amino alcohol is preserved throughout the process because the reaction conditions do not involve strong bases or acidic protons that could facilitate enolization or epimerization at the alpha-carbon. This mechanistic robustness translates directly to commercial viability, as it reduces the burden on downstream purification teams who would otherwise need to separate diastereomeric impurities, thus supporting the delivery of high-purity OLED material or pharmaceutical precursors with minimal additional processing.
How to Synthesize Chiral Bis-Oxazoline Efficiently
The practical execution of this synthesis requires careful attention to stoichiometry and thermal management to maximize yield and optical purity. The patent provides explicit guidance on mixing the dicyanobenzene substrate with a slight excess of the chiral amino alcohol in a dry organic solvent, followed by the addition of the catalyst at a loading of 1-3 wt%. The detailed standardized synthesis steps below outline the precise operational parameters validated in the experimental examples, ensuring reproducibility from gram-scale laboratory trials to multi-kilogram pilot runs.
- Mix dicyanobenzene (para, meta, or ortho) with chiral amino alcohol in an inert organic solvent such as chlorobenzene or xylene.
- Add a Lewis acid catalyst (e.g., ZnCl2, rare earth chloride) at 1-3 wt% loading and heat the mixture to 110-145°C.
- Reflux for 20-30 hours, then perform aqueous workup and column chromatography to isolate the pure chiral oxazoline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits regarding cost structure and operational flexibility. The elimination of complex multi-step sequences reduces the total number of unit operations, which directly correlates to lower capital expenditure on equipment and reduced labor costs per kilogram of finished product. By utilizing commodity chemicals like dicyanobenzenes and commercially available chiral amino alcohols as starting materials, the process mitigates the risk of supply bottlenecks associated with exotic or custom-synthesized precursors. This reliance on widely available feedstocks enhances supply chain reliability, ensuring that production schedules can be maintained even during periods of raw material volatility in the global chemical market.
- Cost Reduction in Manufacturing: The one-pot nature of this reaction eliminates the need for intermediate isolation and purification steps, which are traditionally the most expensive phases of fine chemical manufacturing. By avoiding the use of precious metal catalysts like palladium or platinum in favor of abundant transition metals like zinc or nickel, the process achieves significant cost optimization without sacrificing catalytic performance. Furthermore, the ability to recover and reuse high-boiling solvents like chlorobenzene or xylene through standard distillation techniques contributes to substantial cost savings in raw material consumption and waste disposal fees.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate standard industrial heating and stirring equipment, means that this chemistry can be transferred to contract manufacturing organizations (CMOs) with minimal technical friction. The simplicity of the workup procedure, involving basic aqueous extraction and standard column chromatography or crystallization, ensures that turnaround times are minimized, effectively reducing lead time for high-purity chiral ligand intermediates. This operational simplicity allows for agile response to market demand fluctuations, securing a steady flow of critical materials for downstream asymmetric synthesis applications.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous reagents like phosgene or thionyl chloride, aligning with modern green chemistry principles. The waste profile is significantly cleaner compared to traditional amide coupling methods, primarily consisting of aqueous salts and recoverable organic solvents, which simplifies wastewater treatment and regulatory compliance. This environmental advantage not only reduces the ecological footprint but also lowers the long-term liability and permitting costs associated with operating large-scale chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral oxazoline synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific application needs.
Q: What catalysts are suitable for this oxazoline synthesis?
A: The patent specifies a range of Lewis acids including transition metal chlorides like ZnCl2, NiCl2, and CuCl2, as well as rare earth chlorides and aluminum chlorides, typically used at 1-3 wt% loading.
Q: What are the typical reaction conditions for high yield?
A: Optimal results are achieved by refluxing the reaction mixture in solvents like chlorobenzene or ethylbenzene at temperatures between 120°C and 140°C for approximately 22 to 28 hours.
Q: Can this method produce different isomeric forms?
A: Yes, by selecting para-, meta-, or ortho-dicyanobenzene as the starting material, the process can selectively generate 1,4-, 1,3-, or 1,2-substituted bis-oxazoline ligands respectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Oxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral ligands play in the development of next-generation pharmaceuticals and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral HPLC and NMR instrumentation to guarantee that every batch of chiral oxazoline meets the exacting standards required for asymmetric catalysis.
We invite you to collaborate with us to leverage this innovative synthetic technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your R&D and commercial goals with reliability and precision.
