Scalable Synthesis of Optically Active 1-Aryl Indole Derivatives for Advanced Pharmaceutical Applications
The pharmaceutical industry is constantly seeking robust and efficient pathways to access complex chiral scaffolds that serve as the backbone for next-generation therapeutics. Patent CN110183373B introduces a groundbreaking methodology for the synthesis of optically active 1-aryl indole derivatives, a class of compounds renowned for their presence in bioactive natural products and their utility as chiral ligands. This innovation addresses a critical gap in the market by providing a metal-free, organocatalytic route that achieves exceptional enantioselectivity under mild conditions. Unlike traditional methods that often struggle with poor stereocontrol or require harsh reagents, this patented process utilizes a chiral spirophosphoric acid catalyst to drive a three-component cascade reaction. The resulting compounds, characterized by the general structural formula (I) shown below, exhibit significant potential in medicinal chemistry, particularly in the development of agents for inhibiting glioma.
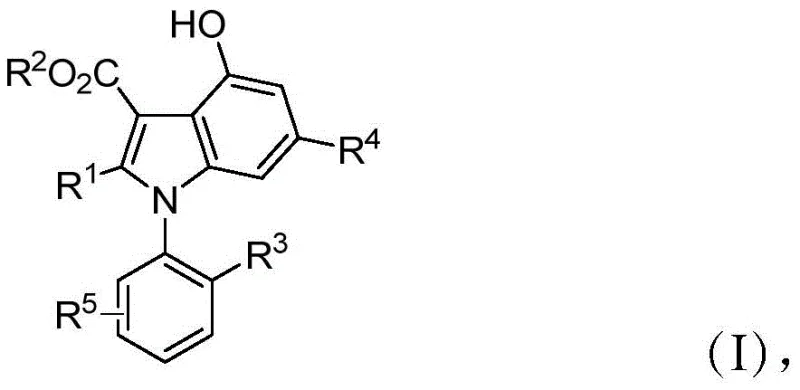
For R&D directors and procurement specialists, the significance of this technology lies in its ability to generate high-purity pharmaceutical intermediates with defined axial chirality. The structural diversity allowed by the variable substituents (R1-R5) means that a wide library of analogs can be generated from a common synthetic platform. This flexibility is crucial for lead optimization campaigns where subtle changes in steric and electronic properties can dramatically alter biological efficacy. Furthermore, the absence of transition metals in the catalytic cycle removes a major bottleneck in downstream processing, ensuring that the final active pharmaceutical ingredient (API) candidates are free from problematic metal residues that often require costly removal steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-aryl indole derivatives with axial chirality has relied heavily on transition metal catalysis or resolution of racemic mixtures. Literature precedents, such as those cited in Tetrahedron (2016), indicate that metal-chiral ligand systems often suffer from mediocre enantioselectivity, necessitating multiple recrystallization steps that erode overall yield. Additionally, the use of heavy metals like palladium or rhodium introduces significant supply chain risks and environmental compliance burdens. The removal of trace metals to meet ICH Q3D guidelines adds substantial cost and complexity to the manufacturing process. Moreover, many conventional routes require elevated temperatures or strong bases that can degrade sensitive functional groups, limiting the scope of substrates that can be utilized. These factors collectively hinder the rapid scale-up required for clinical trial material production.
The Novel Approach
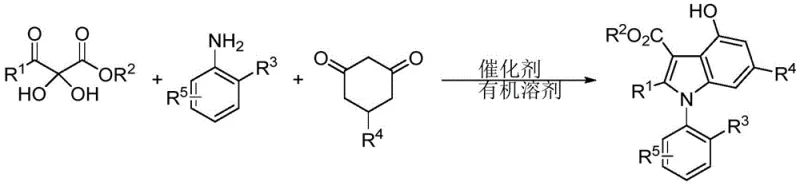
The patented methodology offers a transformative solution by employing an organocatalytic three-component tandem reaction. As illustrated in the reaction scheme below, the process combines a 2,3-diketone ester, an aromatic amine, and a 1,3-cyclohexanedione derivative in a single pot. This convergent strategy not only improves atom economy but also drastically simplifies the operational workflow. The reaction proceeds smoothly at moderate temperatures ranging from 40°C to 100°C in common organic solvents like dichloromethane or toluene. The use of a chiral spirophosphoric acid catalyst ensures high stereocontrol, with experimental data demonstrating enantiomeric excess (ee) values reaching up to 99%. This level of precision eliminates the need for chiral separation columns, directly translating to lower production costs and faster time-to-market for new drug candidates.
Mechanistic Insights into Chiral Spirophosphoric Acid Catalysis
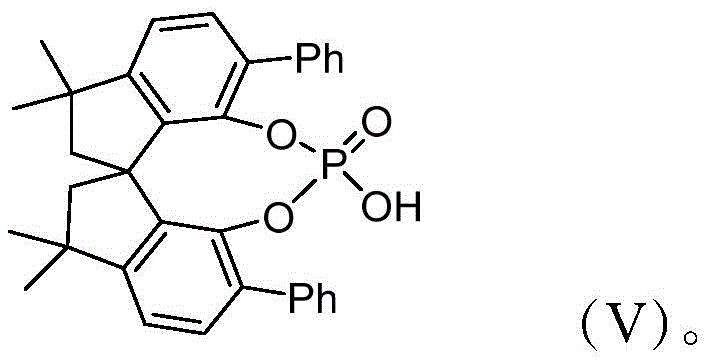
The success of this synthesis hinges on the unique activation mode provided by the chiral spirophosphoric acid catalyst, specifically the 3,3,3',3'-tetramethyl spirocyclic phosphoric acid depicted in structure (V). The mechanism initiates with the condensation of the aromatic amine and the 1,3-cyclohexanedione to form an enamine intermediate. This nucleophilic species then attacks the electrophilic carbonyl of the 2,3-diketone ester. The chiral phosphate anion acts as a Brønsted acid base pair, simultaneously activating the electrophile and organizing the transition state through hydrogen bonding networks. This precise spatial arrangement forces the reaction to proceed through a specific trajectory, thereby establishing the N-C axial chirality with high fidelity. Following the initial coupling, the intermediate undergoes cyclization and dehydration, followed by aromatization to yield the final stable indole core.
From an impurity control perspective, this mechanism offers distinct advantages. Because the reaction is driven by specific non-covalent interactions rather than radical pathways or high-energy intermediates, the formation of side products is minimized. The mild acidic nature of the catalyst prevents the decomposition of acid-sensitive groups that might be present on the aromatic rings. Furthermore, the catalyst loading is relatively low, typically between 1 to 10 mol%, which reduces the burden on the purification process. The final workup involves simple column chromatography, which effectively separates the product from any unreacted starting materials or minor byproducts. This clean reaction profile is essential for maintaining high purity standards required for pharmaceutical grade intermediates, ensuring consistent batch-to-batch quality.
How to Synthesize Optically Active 1-Aryl Indole Derivatives Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins by charging the reaction vessel with equimolar amounts of the three key components: the 2,3-diketone ester, the substituted aniline, and the cyclic diketone. Anhydrous sodium sulfate is often added as a drying agent to drive the equilibrium forward by removing water generated during the condensation steps. The chiral catalyst is then introduced, and the mixture is heated to the optimal temperature, typically around 65°C, for a duration of approximately 16 hours. Monitoring the reaction progress via TLC or HPLC ensures complete conversion before proceeding to isolation. The detailed standardized synthesis steps, including specific solvent ratios and purification parameters, are outlined in the guide below.
- Combine 2,3-diketone ester, aromatic amine, and 1,3-cyclohexanedione in an organic solvent with a chiral spirophosphoric acid catalyst.
- Heat the reaction mixture to 40-100°C for approximately 16 hours to facilitate enamine formation and subsequent cyclization.
- Purify the crude product via silica gel column chromatography using ethyl acetate and petroleum ether to isolate the high-purity optical active derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route presents compelling economic and logistical benefits. The shift away from precious metal catalysts immediately reduces the raw material cost volatility associated with commodities like palladium or rhodium. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents, leading to a smaller environmental footprint and lower waste disposal costs. The robustness of the reaction conditions means that the process is less susceptible to variations in utility supply, such as steam pressure fluctuations, enhancing overall manufacturing reliability. These factors combine to create a more resilient supply chain capable of meeting the rigorous demands of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated scavenging resins results in significant cost savings per kilogram of product. Additionally, the high yields and enantioselectivity reduce the loss of valuable chiral building materials, optimizing the overall material balance. The use of commodity chemicals as starting materials further insulates the production cost from market fluctuations, ensuring stable pricing for long-term contracts.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted anilines and diketones, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors, facilitating easy technology transfer between different manufacturing sites. This flexibility ensures continuous supply even in the event of regional disruptions or equipment maintenance at a specific facility.
- Scalability and Environmental Compliance: The metal-free nature of the process simplifies regulatory filings and environmental impact assessments, accelerating the approval timeline for new drug applications. The reduced generation of heavy metal waste aligns with green chemistry principles and corporate sustainability goals. The process is inherently scalable, having been demonstrated effectively from milligram to gram scales in the patent examples, providing a clear path for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral indole derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is crucial for integrating this technology into your existing drug discovery pipelines.
Q: What is the primary advantage of this organocatalytic method over traditional metal catalysis?
A: The primary advantage is the elimination of toxic heavy metals, which simplifies purification and ensures the final pharmaceutical intermediate meets stringent regulatory limits for residual metals, while achieving high enantioselectivity (up to 99% ee).
Q: Can this synthesis method be scaled for commercial production of API intermediates?
A: Yes, the process utilizes readily available starting materials and mild reaction conditions (40-100°C) without requiring specialized high-pressure equipment, making it highly suitable for commercial scale-up from kilogram to tonnage levels.
Q: What represents the key structural feature of the catalyst used in this patent?
A: The catalyst is an optically active 3,3,3',3'-tetramethyl spirocyclic phosphoric acid, which provides the necessary chiral environment to induce axial chirality at the N-C bond of the indole derivative during the three-component cascade reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aryl Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of life-saving medications. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-aryl indole derivatives meets the highest international standards. Our commitment to quality extends beyond mere compliance; we strive to be a strategic partner in your innovation journey.
We invite you to contact our technical procurement team to discuss how this advanced organocatalytic technology can benefit your specific projects. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this metal-free route. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your target molecules. Let us help you accelerate your drug development timeline with reliable, high-performance chemical solutions.
