Scalable Synthesis of Axially Chiral 1-Aryl Indoles via Organocatalysis for Pharmaceutical Applications
Scalable Synthesis of Axially Chiral 1-Aryl Indoles via Organocatalysis for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign methods to construct complex chiral scaffolds, particularly those with axial chirality which are increasingly recognized for their unique biological profiles. Patent CN110183373B introduces a groundbreaking methodology for the synthesis of optically active 1-aryl indole derivatives, a class of compounds that serves as a critical backbone for various bioactive molecules and chiral ligands. This technology leverages a sophisticated three-component tandem reaction catalyzed by an optically active 3,3,3',3'-tetramethyl spirocyclic phosphoric acid, offering a distinct advantage over traditional transition metal-catalyzed routes. The resulting structures, defined generally by Formula (I), possess a stable C-N chiral axis and demonstrate potent pharmacological activities, including significant inhibition of glioma cells. For R&D teams focused on oncology and neurodegenerative diseases, accessing these high-purity intermediates through a reliable pharmaceutical intermediate supplier is essential for accelerating lead optimization campaigns.
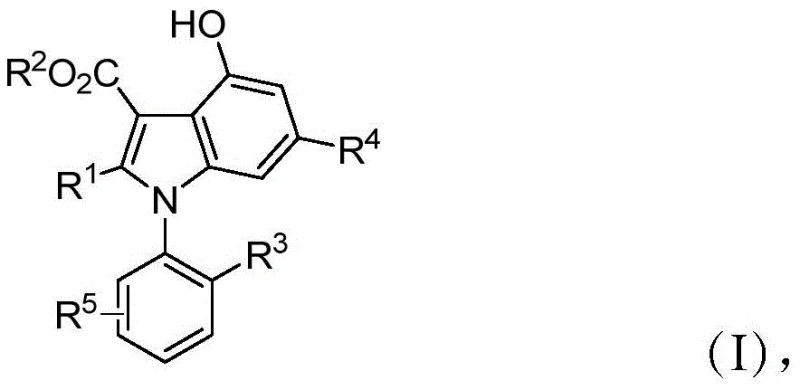
As illustrated in the general structure above, the versatility of this synthetic platform allows for extensive diversification at the R1 through R5 positions, enabling medicinal chemists to rapidly explore structure-activity relationships (SAR). The presence of functional groups such as halogens, alkyls, and aryls provides ample handles for further derivatization, making these molecules invaluable building blocks in modern drug discovery. Furthermore, the ability to access both levorotatory and dextrorotatory forms ensures that researchers can evaluate the specific biological impact of each enantiomer, a critical requirement for regulatory approval of new chiral drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral 1-aryl indoles has relied heavily on transition metal catalysis, often involving palladium, copper, or rhodium complexes paired with chiral ligands. While effective in some contexts, these conventional methods suffer from significant drawbacks that hinder their utility in large-scale API manufacturing. Firstly, the removal of trace metal residues to meet stringent ICH Q3D guidelines requires additional purification steps, such as scavenging or recrystallization, which drastically reduces overall yield and increases production costs. Secondly, many metal-catalyzed protocols require harsh reaction conditions, including high temperatures or strong bases, which can lead to the decomposition of sensitive functional groups and the formation of difficult-to-remove impurities. Additionally, the enantioselectivity reported in earlier literature, such as in Tetrahedron 72 (2016), was often moderate, necessitating costly chiral resolution steps to obtain the desired optical purity.
The Novel Approach
In stark contrast, the methodology disclosed in CN110183373B utilizes a metal-free organocatalytic strategy that fundamentally reshapes the efficiency profile of this transformation. By employing a chiral spirophosphoric acid catalyst, the reaction proceeds through a highly ordered transition state that ensures excellent stereocontrol without the need for toxic heavy metals. The process involves a one-pot three-component coupling of a 2,3-diketone ester, an aromatic amine, and a 1,3-cyclohexanedione derivative, as depicted in the reaction scheme below. This convergent approach not only simplifies the operational workflow but also significantly enhances atom economy. The mild reaction conditions, typically ranging from 40°C to 100°C in common organic solvents like dichloromethane or toluene, preserve the integrity of sensitive substrates and minimize energy consumption, representing a substantial step forward in cost reduction in pharmaceutical intermediate manufacturing.
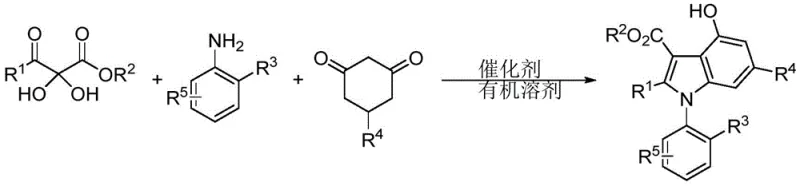
Mechanistic Insights into Chiral Spirophosphoric Acid Catalyzed Cyclization
The success of this transformation lies in the unique dual-activation mode of the chiral spirophosphoric acid catalyst, structurally represented by Formula (V). This organocatalyst acts as a Brønsted acid, simultaneously activating the electrophilic carbonyl species and stabilizing the nucleophilic enamine intermediate formed in situ from the condensation of the aromatic amine and 1,3-cyclohexanedione. The rigid spirocyclic backbone of the catalyst creates a well-defined chiral pocket that directs the approach of the reacting partners, thereby enforcing the formation of the N-C axial chirality with high fidelity. This precise spatial arrangement prevents the formation of racemic byproducts and ensures that the cyclization and subsequent aromatization steps proceed with exceptional enantioselectivity, often exceeding 90% ee and reaching up to 99% ee in optimized examples.
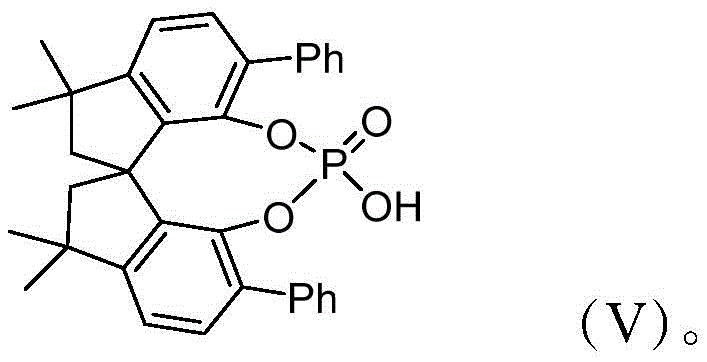
From an impurity control perspective, this mechanism offers distinct advantages over radical or metal-mediated pathways. The absence of single-electron transfer processes minimizes the generation of polymeric tars or uncontrolled oligomers, which are common issues in indole synthesis. Furthermore, the dehydration step required for aromatization is facilitated by the acidic nature of the catalyst and the presence of drying agents like anhydrous sodium sulfate, driving the equilibrium towards the desired product. This clean reaction profile simplifies downstream processing, allowing for the isolation of high-purity pharmaceutical intermediates via standard silica gel chromatography without the need for complex aqueous workups to remove metal salts.
How to Synthesize Optically Active 1-Aryl Indole Derivatives Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory equipment, making it an attractive option for process development teams aiming to secure a reliable supply chain for chiral indoles. The protocol typically involves charging the three components in equimolar ratios along with a catalytic amount (1-10 mol%) of the chiral phosphoric acid in a dry organic solvent. The reaction mixture is then heated under inert atmosphere, monitored by TLC or HPLC, and upon completion, the catalyst can potentially be recovered or the product directly purified. For detailed operational parameters, stoichiometry, and specific workup procedures tailored to your specific substrate combination, please refer to the standardized synthesis guide below.
- Combine 2,3-diketone ester, aromatic amine, and 1,3-cyclohexanedione in an organic solvent with a chiral spirophosphoric acid catalyst.
- Heat the reaction mixture to 40-100°C for approximately 16 hours to facilitate enamine formation and cyclization.
- Purify the crude product via silica gel column chromatography using ethyl acetate and petroleum ether to isolate the high-purity chiral indole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic route presents a compelling value proposition centered on risk mitigation and cost efficiency. The elimination of precious metal catalysts removes a major source of supply chain volatility associated with fluctuating prices of palladium or rhodium, while also eradicating the regulatory burden of proving low residual metal levels in the final drug substance. This translates directly into a more predictable and stable manufacturing timeline, reducing the risk of batch failures due to metal contamination. Moreover, the use of commodity chemicals such as substituted anilines and beta-keto esters as starting materials ensures that raw material sourcing is robust and geographically diverse, enhancing the overall resilience of the supply network against regional disruptions.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the purification train. Since no metal scavengers or specialized filtration media are required, the operational expenditure (OPEX) per kilogram of product is significantly lowered. The high yields reported (often exceeding 80%) combined with the high optical purity mean that less starting material is wasted, and the need for recycling or reprocessing off-spec material is minimized. This efficiency gain allows for a more competitive pricing structure for the final high-purity pharmaceutical intermediate, providing a clear margin advantage for downstream drug developers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to superior batch-to-batch consistency, a critical factor for maintaining continuous supply agreements. The tolerance of the catalyst to various functional groups means that a single catalytic system can be applied to a wide library of derivatives, reducing the need for multiple specialized production lines. This flexibility enables manufacturers to respond quickly to changing demand patterns for different analogues, ensuring that clinical trial materials and commercial stocks can be delivered with reduced lead times and greater reliability.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this metal-free methodology aligns perfectly with green chemistry principles. The absence of heavy metals simplifies waste stream treatment and disposal, lowering the environmental compliance costs associated with hazardous waste management. The mild thermal requirements also reduce the energy footprint of the process. These factors collectively facilitate easier commercial scale-up of complex pharmaceutical intermediates from pilot plant to multi-ton production, ensuring that the technology remains viable and sustainable as volume requirements increase during late-stage clinical development.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific projects, we have compiled answers to common inquiries regarding the synthesis and application of these chiral indoles. The following insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, addressing key concerns about scalability, purity, and biological relevance. Understanding these details is crucial for integrating this novel chemistry into your existing drug discovery pipelines.
Q: What is the primary advantage of this organocatalytic method over traditional metal catalysis?
A: The primary advantage is the complete elimination of transition metal residues, which simplifies purification and ensures compliance with strict pharmaceutical heavy metal limits, while achieving high enantiomeric excess (up to 99% ee).
Q: Can this synthesis be scaled for commercial production of API intermediates?
A: Yes, the reaction utilizes mild temperatures (40-100°C) and commercially available starting materials like beta-keto esters and anilines, making it highly suitable for scale-up from laboratory to multi-ton production.
Q: What is the biological significance of these 1-aryl indole derivatives?
A: These compounds exhibit significant cytotoxicity against glioma cell lines (such as C6), showing better activity than reference standards like doxorubicin in specific configurations, highlighting their potential in oncology drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aryl Indole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating the delivery of life-saving therapies to patients. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We are committed to delivering stringent purity specifications for all our products, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify optical purity and chemical identity. Whether you require custom synthesis of specific analogues or bulk supply of the core scaffold, our infrastructure is designed to meet the demanding timelines of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge organocatalytic technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis to quantify the potential economic benefits of switching to this metal-free route for your specific target molecule. Please contact us today to discuss your requirements, obtain specific COA data for our available inventory, and receive comprehensive route feasibility assessments tailored to your development goals.
