Advanced Carbonylation Strategy for High-Purity Thioester Compounds and Commercial Scalability
Advanced Carbonylation Strategy for High-Purity Thioester Compounds and Commercial Scalability
The landscape of organic synthesis for sulfur-containing intermediates is undergoing a significant transformation driven by the need for safer, more efficient, and scalable processes. Patent CN113004181B introduces a groundbreaking methodology for the preparation of thioester compounds via a transition metal-catalyzed carbonylation reaction. This innovation addresses long-standing challenges in the field by utilizing inexpensive and readily available benzyl chloride compounds alongside sulfonyl chlorides as key starting materials. Unlike traditional routes that rely on hazardous gaseous carbon monoxide or malodorous thiols, this novel approach employs tungsten hexacarbonyl as a solid, manageable carbonyl source. For R&D directors and process chemists seeking reliable thioester supplier capabilities, this technology represents a paradigm shift towards greener and more controllable manufacturing protocols that maintain high purity standards while drastically simplifying operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has been fraught with significant operational and safety hurdles that impede large-scale commercialization. The most prevalent conventional method involves the direct acylation of thiols with carboxylic acids or their derivatives, a process that is inherently problematic due to the physicochemical properties of thiols. Thiol compounds are notorious for their extremely unpleasant, pervasive odors which pose severe health and safety risks in industrial settings, requiring specialized containment infrastructure. Furthermore, thiols act as potent catalyst poisons, often deactivating transition metal catalysts required for more sophisticated coupling reactions, thereby limiting the scope of applicable synthetic routes. Alternative methods involving the oxidative coupling of aldehydes or substitution reactions of halogenated alkanes often suffer from poor atom economy, harsh reaction conditions, or limited substrate tolerance, making them unsuitable for the cost reduction in pharmaceutical intermediate manufacturing demanded by modern supply chains.
The Novel Approach
The methodology disclosed in the patent data offers a robust solution by replacing volatile thiols with stable sulfonyl chlorides and utilizing benzyl chlorides instead of less reactive aryl halides in specific carbonylation contexts. This strategic substitution not only eliminates the olfactory hazards associated with thiols but also leverages the high reactivity of benzyl chlorides to drive the carbonylation forward efficiently. By employing a palladium catalyst system ligated with Xantphos, the reaction achieves high conversion rates under relatively mild thermal conditions. The use of sulfonyl chloride as a sulfur source provides a level of designability and functional group tolerance that was previously difficult to achieve, allowing for the synthesis of complex thioester architectures essential for advanced drug discovery. This approach effectively bypasses the need for high-pressure CO gas cylinders, enhancing the safety profile and scalability of the process for commercial production.
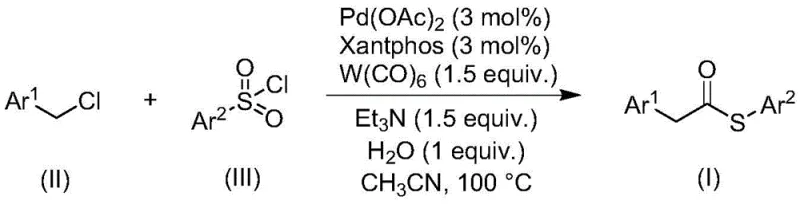
The general reaction pathway illustrates the convergence of benzyl chloride derivatives and sulfonyl chlorides in the presence of a palladium catalyst and tungsten hexacarbonyl. This transformation is pivotal for generating the thioester linkage (C(=O)-S) which is a critical motif in numerous bioactive molecules and polymer additives. The ability to execute this transformation in a single pot with high efficiency underscores the potential for this technology to become a standard operation in fine chemical facilities aiming for high-purity OLED material or pharmaceutical intermediate production.
Mechanistic Insights into Pd-Catalyzed Carbonylation with Sulfonyl Chlorides
At the heart of this synthetic breakthrough lies a sophisticated catalytic cycle mediated by palladium acetate and the bidentate phosphine ligand 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (Xantphos). The large bite angle of Xantphos is crucial for stabilizing the palladium center during the oxidative addition step, particularly when dealing with benzyl chloride substrates which can be prone to side reactions. The mechanism likely initiates with the oxidative addition of the benzyl chloride to the Pd(0) species, forming a benzyl-palladium(II) complex. Subsequently, carbon monoxide, released in situ from the decomposition of tungsten hexacarbonyl [W(CO)6], inserts into the palladium-carbon bond to generate an acyl-palladium intermediate. This step is facilitated by the unique ability of W(CO)6 to act as both a CO donor and a reducing agent, regenerating the active Pd(0) catalyst without the need for external reductants like zinc or manganese powder.
Following CO insertion, the sulfonyl chloride component interacts with the acyl-palladium species. While the exact mechanistic details of the sulfur transfer are complex, it is hypothesized that the sulfonyl chloride undergoes reduction or activation to generate a sulfur nucleophile in situ, which then attacks the electrophilic acyl carbon or undergoes transmetallation with the palladium center. The presence of triethylamine and water plays a critical role in buffering the reaction medium and facilitating the necessary proton transfers or hydrolysis steps that lead to the final thioester product. This intricate interplay of reagents ensures that the reaction proceeds with high selectivity, minimizing the formation of symmetric ketones or other carbonylation byproducts. The result is a clean reaction profile that simplifies downstream purification, a key factor for maintaining stringent purity specifications in regulated industries.
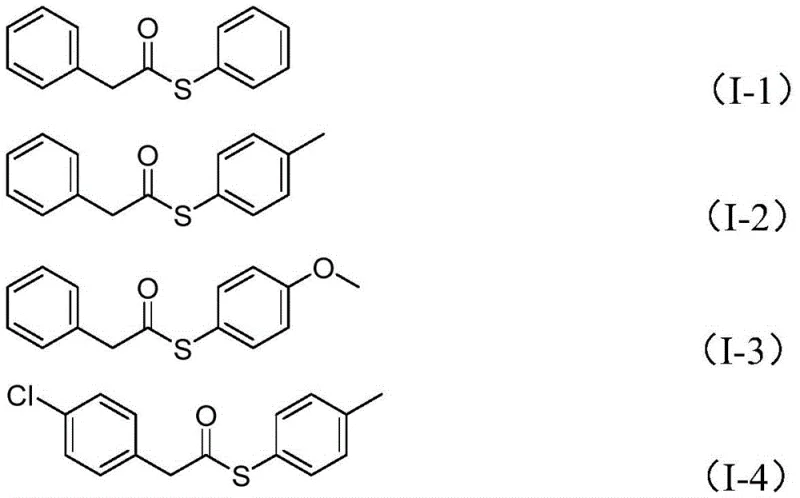
The scope of this reaction is further evidenced by the successful synthesis of various substituted thioesters, as shown in the structural examples. The system tolerates electron-donating groups like methoxy and methyl, as well as electron-withdrawing halogens, demonstrating the versatility required for diverse medicinal chemistry campaigns. This broad functional group compatibility is essential for the commercial scale-up of complex polymer additives and specialty chemicals where molecular diversity is paramount.
How to Synthesize Thioester Compounds Efficiently
Implementing this carbonylation protocol requires precise control over stoichiometry and reaction parameters to maximize yield and minimize impurities. The process is designed to be operationally simple, utilizing standard laboratory glassware or stainless steel reactors capable of maintaining temperatures up to 100°C. The following section outlines the generalized procedure derived from the patent examples, serving as a foundational guide for process development teams looking to adapt this chemistry for larger batches. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety compliance.
- Charge a reaction vessel with palladium acetate (3 mol%), Xantphos (3 mol%), tungsten hexacarbonyl (1.5 equiv.), triethylamine (1.5 equiv.), water (1 equiv.), benzyl chloride derivative, and sulfonyl chloride derivative in acetonitrile.
- Heat the reaction mixture to 100°C and maintain stirring for 24 hours to ensure complete conversion of the starting materials into the target thioester structure.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this carbonylation technology offers substantial strategic benefits that extend beyond mere chemical novelty. The shift away from malodorous thiols and high-pressure carbon monoxide gas significantly reduces the regulatory burden and infrastructure costs associated with hazardous material handling. Facilities can operate with greater flexibility and lower insurance premiums, directly impacting the bottom line. Furthermore, the reliance on commercially available and inexpensive starting materials such as benzyl chlorides and sulfonyl chlorides ensures a stable and resilient supply chain, mitigating the risks of raw material shortages that often plague the fine chemical sector.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents translates directly into lower production costs. By using tungsten hexacarbonyl as a combined CO source and reductant, the process removes the need for separate reducing agents and specialized high-pressure gas equipment, leading to significant capital expenditure savings. Additionally, the simplified workup procedure, which involves basic filtration and chromatography, reduces solvent consumption and labor hours, driving down the overall cost of goods sold (COGS) for high-value thioester intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for consistent batch-to-batch quality, which is critical for maintaining long-term contracts with pharmaceutical clients. The use of stable solid reagents rather than volatile gases or liquids minimizes the risk of shipping delays or storage degradation. This reliability ensures that downstream customers receive their orders on time, fostering stronger partnerships and reducing the lead time for high-purity pharmaceutical intermediates needed for clinical trials and commercial drug launches.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently in standard organic solvents like acetonitrile. The absence of toxic thiol waste streams simplifies wastewater treatment and aligns with increasingly strict environmental regulations regarding volatile organic compounds (VOCs) and sulfur emissions. This environmental compatibility facilitates easier permitting for plant expansion and supports corporate sustainability goals, making it an attractive option for green chemistry initiatives within the specialty chemical industry.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is vital for stakeholders evaluating its potential integration into existing production lines. The following questions address common inquiries regarding safety, scalability, and substrate compatibility based on the experimental data provided in the patent documentation. These insights are intended to clarify the operational advantages and assist in decision-making processes for technical procurement teams.
Q: What is the primary advantage of using sulfonyl chlorides over thiols in thioester synthesis?
A: The primary advantage is the avoidance of unpleasant odors and catalyst poisoning associated with volatile thiol compounds. Sulfonyl chlorides serve as a stable, odorless sulfur source that simplifies handling and improves workplace safety.
Q: Can this carbonylation method tolerate diverse functional groups on the aromatic rings?
A: Yes, the method demonstrates wide substrate tolerance. It successfully accommodates substituents such as methyl, methoxy, tert-butyl, halogens (Cl, F, Br), and ester groups on both the benzyl chloride and sulfonyl chloride components without significant yield loss.
Q: What role does tungsten hexacarbonyl play in this reaction system?
A: Tungsten hexacarbonyl [W(CO)6] serves a dual function as both the carbon monoxide source for the carbonylation step and as a reducing agent, eliminating the need for additional external reducing agents or high-pressure CO gas equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
As the demand for high-quality sulfur-containing intermediates continues to rise in the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO is essential for navigating the complexities of process development. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our state-of-the-art facilities are equipped to handle sensitive carbonylation reactions with the utmost safety and precision, adhering to stringent purity specifications and rigorous QC labs to guarantee product integrity.
We invite you to leverage our technical expertise to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific thioester requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced carbonylation capabilities can meet your critical deadlines and quality standards.
