Advanced Carbonylation Strategy for Scalable Thioester Production in Pharmaceutical Manufacturing
Advanced Carbonylation Strategy for Scalable Thioester Production in Pharmaceutical Manufacturing
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and fine chemical industries, particularly regarding safety, efficiency, and scalability. A significant breakthrough in this domain is detailed in patent CN113004181B, which discloses a novel method for preparing thioester compounds through a transition metal-catalyzed carbonylation reaction. Thioesters are pivotal structural motifs serving as stable acyl donors in complex organic syntheses and playing critical roles in biological processes such as native chemical ligation. However, traditional synthetic routes often rely on hazardous reagents or harsh conditions that limit their industrial applicability. This new methodology addresses these challenges by utilizing inexpensive and readily available benzyl chloride compounds and sulfonyl chlorides as starting materials, effectively bypassing the need for foul-smelling and toxic thiol reagents. By leveraging a palladium-catalyzed system with tungsten hexacarbonyl, this process offers a safer, more environmentally benign pathway to high-value intermediates, positioning it as a highly attractive option for reliable pharmaceutical intermediate suppliers seeking to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has been predominantly achieved through the direct acylation of thiols with carboxylic acids or their derivatives, a process fraught with significant operational and safety drawbacks. Thiols are notoriously malodorous, possessing a repulsive smell that persists even at extremely low concentrations, posing severe challenges for odor containment in large-scale manufacturing environments. Furthermore, thiols are prone to oxidation, leading to the formation of disulfide impurities that can complicate purification and reduce overall yield. Alternative methods involving the oxidative coupling of aldehydes or substitution reactions of halogenated alkanes often suffer from limited substrate scope or require stoichiometric amounts of oxidants that generate substantial chemical waste. Additionally, conventional carbonylation reactions typically necessitate the use of high-pressure carbon monoxide gas, which introduces significant safety risks related to toxicity and explosion hazards, requiring specialized and expensive high-pressure reactor infrastructure that many standard chemical plants lack.
The Novel Approach
In stark contrast to these legacy methods, the innovative process described in the patent utilizes a transition metal-catalyzed carbonylation strategy that fundamentally alters the reagent profile for thioester synthesis. By employing sulfonyl chlorides as the sulfur source instead of thiols, the method completely eliminates the handling of volatile, odorous sulfur compounds, thereby drastically improving the working environment and reducing the burden on waste gas treatment systems. The reaction leverages benzyl chlorides, which are stable, commercially abundant, and cost-effective building blocks, reacting them with sulfonyl chlorides in the presence of a palladium catalyst and a phosphine ligand. Crucially, the system uses tungsten hexacarbonyl (W(CO)6) as a solid source of carbon monoxide, which not only provides the necessary carbonyl group but also acts as a reducing agent, negating the need for external reducing agents or dangerous CO gas cylinders. This approach streamlines the reaction setup, allowing it to proceed under atmospheric pressure conditions in standard glassware or reactors, significantly lowering the barrier to entry for commercial adoption.
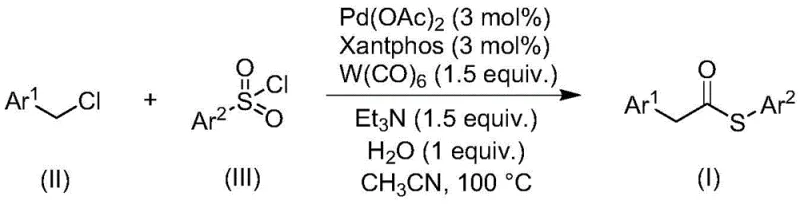
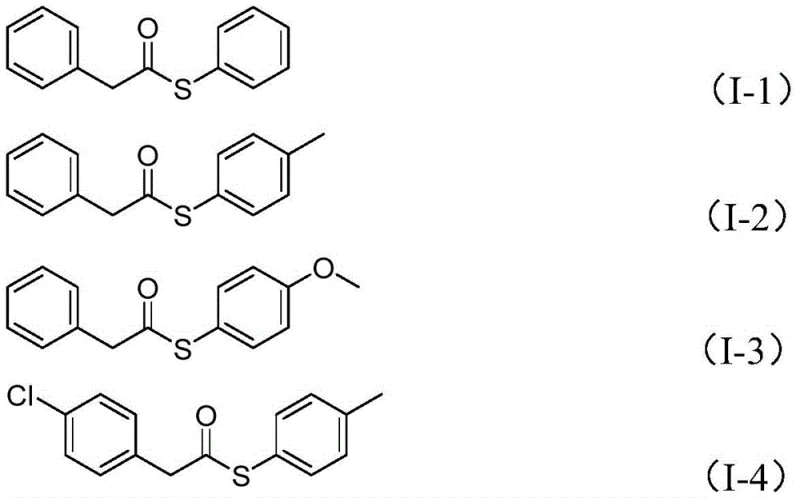
The versatility of this novel approach is further evidenced by its broad substrate tolerance, accommodating a wide range of electronic and steric environments on both the benzyl chloride and sulfonyl chloride components. As illustrated in the reaction scheme, the process successfully integrates various substituted aryl groups, demonstrating robustness against functional groups such as halogens, alkyls, and alkoxy groups. This flexibility is paramount for process chemists who require a adaptable synthetic route capable of generating diverse libraries of thioester derivatives for drug discovery and development programs without the need for extensive re-optimization for each new substrate.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this synthetic advancement lies in the intricate interplay between the palladium catalyst, the Xantphos ligand, and the tungsten hexacarbonyl additive. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species generated in situ from palladium acetate and the bidentate phosphine ligand 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (Xantphos). The use of Xantphos is particularly strategic due to its large bite angle, which facilitates the reductive elimination step and stabilizes the palladium center throughout the catalytic cycle. Following oxidative addition, the insertion of carbon monoxide—released slowly and controllably from the decomposition of tungsten hexacarbonyl—into the palladium-carbon bond forms an acyl-palladium intermediate. This in situ generation of CO ensures a steady, low concentration of the gas within the reaction mixture, minimizing side reactions and enhancing safety compared to bubbling high-pressure CO gas.
Subsequent nucleophilic attack by the sulfur species derived from the sulfonyl chloride, likely activated by the reducing environment provided by the tungsten species and triethylamine, leads to the formation of the thioester bond. The presence of water in the reaction mixture plays a subtle yet critical role, potentially assisting in the hydrolysis of intermediate sulfinate species or facilitating proton transfer steps necessary for catalyst turnover. From an impurity control perspective, this mechanism is highly favorable because it avoids the radical pathways often associated with thiol oxidation, thereby suppressing the formation of disulfide byproducts. The result is a cleaner reaction profile that simplifies downstream processing, a key consideration for manufacturing high-purity pharmaceutical intermediates where strict impurity thresholds must be met. The ability of tungsten hexacarbonyl to serve a dual function as both a carbonyl source and a reductant simplifies the reagent stoichiometry and reduces the overall chemical load, contributing to a more atom-economical process.
How to Synthesize Thioester Compounds Efficiently
Implementing this carbonylation protocol in a laboratory or pilot plant setting requires careful attention to reagent ratios and reaction conditions to maximize yield and purity. The standard procedure involves charging a reaction vessel with the benzyl chloride substrate, the sulfonyl chloride coupling partner, palladium acetate, Xantphos, tungsten hexacarbonyl, triethylamine, and a stoichiometric amount of water in an aprotic organic solvent such as acetonitrile. The mixture is then heated to approximately 100°C and stirred for a period of 24 hours to ensure complete conversion. The choice of acetonitrile as the solvent is preferred due to its ability to effectively dissolve all reaction components while promoting high conversion rates. Post-reaction workup is remarkably straightforward, involving simple filtration to remove insoluble tungsten residues, followed by mixing with silica gel and purification via column chromatography.
- Charge a reaction vessel with palladium acetate, Xantphos ligand, tungsten hexacarbonyl, triethylamine, water, benzyl chloride, and sulfonyl chloride in acetonitrile.
- Heat the reaction mixture to 100°C and maintain stirring for 24 hours to ensure complete conversion of the starting materials.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible operational improvements and risk mitigation strategies that extend beyond mere chemical yield. The shift away from gaseous carbon monoxide and liquid thiols represents a fundamental upgrade in process safety, eliminating the need for specialized high-pressure equipment and complex odor scrubbing systems. This reduction in infrastructure requirements allows for greater flexibility in manufacturing site selection and significantly lowers capital expenditure associated with reactor certification and maintenance. Furthermore, the reliance on solid, shelf-stable reagents like tungsten hexacarbonyl and commercially ubiquitous benzyl chlorides enhances supply chain resilience, reducing the vulnerability to disruptions often associated with hazardous gas logistics or specialty thiol suppliers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction setup and the elimination of costly safety measures. By removing the necessity for high-pressure CO autoclaves, manufacturers can utilize standard glass-lined or stainless steel reactors, drastically reducing equipment depreciation and maintenance costs. Additionally, the avoidance of thiols eliminates the expenses related to specialized personal protective equipment (PPE), odor neutralization chemicals, and the disposal of hazardous sulfur-containing waste streams. The dual role of tungsten hexacarbonyl as both a reagent and a reducing agent further streamlines the bill of materials, reducing the number of distinct chemicals that need to be sourced, stored, and managed, leading to substantial overall cost savings in fine chemical manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the raw material supply chain is significantly improved by the use of commodity chemicals. Benzyl chlorides and sulfonyl chlorides are produced on a massive scale globally for various industrial applications, ensuring a stable and competitive market price with multiple qualified vendors available. This contrasts sharply with specialized thiols or high-pressure gas cylinders, which may have limited suppliers and longer lead times. The stability of the solid reagents also allows for bulk purchasing and long-term storage without significant degradation, enabling manufacturers to buffer against market volatility and ensure continuous production schedules even during periods of raw material scarcity, thereby securing the continuity of supply for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the homogeneous nature of the reaction and the absence of gas-liquid mass transfer limitations typical of traditional carbonylations. The reaction proceeds efficiently in common solvents like acetonitrile, which are easily recovered and recycled, aligning with green chemistry principles. The elimination of malodorous thiols and toxic CO gas greatly simplifies environmental compliance, reducing the regulatory burden and permitting timelines for new production lines. The straightforward workup procedure, involving filtration and chromatography, is amenable to automation and continuous processing technologies, offering a clear pathway for increasing throughput without compromising product quality or environmental standards.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction parameters and scope. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a realistic overview of what can be expected during process development. Understanding these nuances is essential for accurately projecting timelines and resource allocation for potential scale-up projects.
Q: What is the primary advantage of using sulfonyl chloride over thiols in this synthesis?
A: The use of sulfonyl chloride eliminates the handling of malodorous and toxic thiol compounds, significantly improving workplace safety and simplifying odor control measures in large-scale manufacturing facilities.
Q: Can this carbonylation method tolerate diverse functional groups on the aromatic rings?
A: Yes, the patented process demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens such as chlorine and fluorine.
Q: Is high-pressure carbon monoxide gas required for this reaction?
A: No, the method utilizes tungsten hexacarbonyl (W(CO)6) as a solid carbonyl source, which acts as both the CO donor and a reducing agent, thereby removing the need for hazardous high-pressure CO gas cylinders.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced carbonylation technologies in modernizing the production of high-value chemical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113004181B can be seamlessly translated into robust, GMP-compliant manufacturing processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay. Our facility is designed to handle complex organometallic chemistry safely, with dedicated containment systems for palladium recovery and solvent recycling, maximizing efficiency while minimizing environmental impact.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage potential partners to contact us directly to obtain specific COA data for similar thioester structures and to discuss route feasibility assessments that could accelerate your development timeline. Let us help you secure a sustainable, cost-effective, and high-quality supply of thioester intermediates for your global operations.
