Revolutionizing Chiral Amine Synthesis: Marine Strain Catalysis for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce high-value chiral intermediates, and patent CN103224963A presents a significant technological breakthrough in this domain. This intellectual property discloses a novel method for preparing chiral amines through asymmetric reduction catalyzed by a specific marine bacterial strain, offering a robust alternative to conventional synthetic routes. The core innovation lies in the utilization of a newly screened marine microorganism, identified as Pseudomonas kilonensis, which contains a highly selective NADH-dependent amine dehydrogenase. This biological catalyst enables the direct asymmetric reductive amination of ketones, bypassing the need for complex chemical resolving agents or expensive transition metal complexes that often plague traditional manufacturing. For R&D directors and procurement specialists, this technology represents a pivotal shift towards greener, more cost-effective production of critical pharmaceutical intermediates, ensuring both high optical purity and substantial yield improvements in industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis of chiral amines has long been hindered by inherent inefficiencies and environmental concerns that impact both cost and supply chain stability. Conventional methods often rely on the resolution of racemic mixtures using chiral acids or enzymes like lipases, which theoretically limits the maximum yield to 50%, thereby doubling the raw material consumption and waste generation for the discarded enantiomer. Furthermore, chemical asymmetric reduction frequently necessitates the use of precious metal catalysts such as rhodium or ruthenium, which not only drive up raw material costs significantly but also introduce stringent requirements for residual metal control in the final active pharmaceutical ingredients. The removal of these heavy metals requires additional purification steps, increasing processing time and complicating the regulatory approval process for drug substances. Additionally, many chemical processes operate under extreme temperatures or pressures, posing safety risks and requiring specialized, high-cost reactor infrastructure that limits flexibility in multi-purpose manufacturing facilities.
The Novel Approach
In stark contrast, the technology described in patent CN103224963A leverages the power of whole-cell biocatalysis to overcome these historical bottlenecks with remarkable efficiency. By employing a marine-derived strain, the process achieves yields exceeding 70% and optical purities greater than 90% ee, effectively breaking the 50% yield barrier associated with kinetic resolution methods. This approach utilizes readily available ketones as starting materials, which are converted directly into the desired chiral amines through an enzymatic pathway that is inherently stereoselective.  The versatility of this system is evident in its ability to accommodate a wide range of substrates, including aliphatic, aromatic, and hydroxy ketones, as illustrated by the general structural formula where R1 and R2 can vary significantly. This flexibility allows manufacturers to adapt the platform technology to diverse product portfolios without needing to develop entirely new synthetic routes for each molecule, thereby accelerating time-to-market for new drug candidates.
The versatility of this system is evident in its ability to accommodate a wide range of substrates, including aliphatic, aromatic, and hydroxy ketones, as illustrated by the general structural formula where R1 and R2 can vary significantly. This flexibility allows manufacturers to adapt the platform technology to diverse product portfolios without needing to develop entirely new synthetic routes for each molecule, thereby accelerating time-to-market for new drug candidates.
Mechanistic Insights into NADH-Dependent Asymmetric Reductive Amination
The scientific foundation of this process rests on the sophisticated enzymatic machinery within the Pseudomonas kilonensis cells, specifically the presence of NADH-dependent amine dehydrogenase. This enzyme facilitates the transfer of a hydride ion from the reduced cofactor NADH to the imine intermediate formed between the ketone substrate and the amino donor, resulting in the formation of the chiral amine with high stereocontrol. A critical component of this mechanistic cycle is the in-situ regeneration of the expensive NADH cofactor, which is achieved through the oxidation of an auxiliary substrate such as glucose. 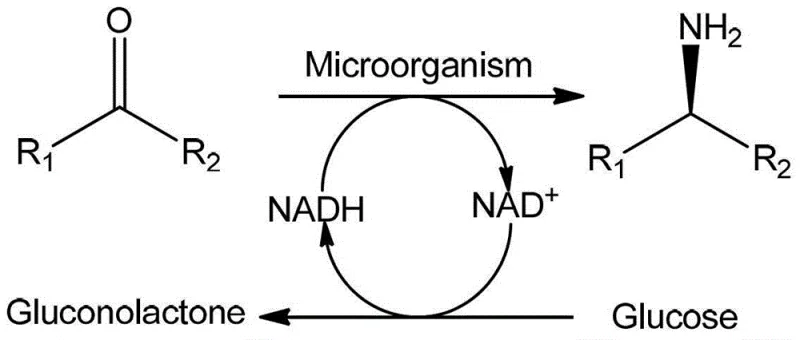 As depicted in the reaction mechanism, glucose is oxidized to gluconolactone, simultaneously reducing NAD+ back to NADH, which sustains the catalytic cycle without the need for stoichiometric amounts of external cofactors. This self-sustaining loop drastically reduces the cost of goods sold by minimizing the consumption of high-value reagents. Furthermore, the whole-cell format protects the delicate enzymes within the natural cellular environment, enhancing their stability and operational lifespan compared to isolated enzyme systems, which often suffer from rapid denaturation under industrial process conditions.
As depicted in the reaction mechanism, glucose is oxidized to gluconolactone, simultaneously reducing NAD+ back to NADH, which sustains the catalytic cycle without the need for stoichiometric amounts of external cofactors. This self-sustaining loop drastically reduces the cost of goods sold by minimizing the consumption of high-value reagents. Furthermore, the whole-cell format protects the delicate enzymes within the natural cellular environment, enhancing their stability and operational lifespan compared to isolated enzyme systems, which often suffer from rapid denaturation under industrial process conditions.
Impurity control is another area where this biocatalytic mechanism excels, providing a cleaner reaction profile that simplifies downstream processing. The high enantioselectivity of the marine strain's amine dehydrogenase ensures that the formation of the unwanted enantiomer is minimized from the outset, reducing the burden on crystallization or chromatographic purification steps later in the process. The reaction conditions, typically maintained between pH 7 and 13 and temperatures ranging from 0°C to 70°C, are mild enough to prevent the degradation of sensitive functional groups that might be present on complex pharmaceutical intermediates. This gentle reaction environment also suppresses side reactions such as over-reduction or polymerization, which are common pitfalls in chemical hydrogenation processes. Consequently, the crude product stream contains fewer by-products, allowing for higher overall recovery rates and reducing the volume of organic solvents required for extraction and purification, aligning with modern green chemistry principles.
How to Synthesize Chiral Amines Efficiently
Implementing this synthesis route requires a structured approach to fermentation and biocatalysis to ensure consistent quality and yield at scale. The process begins with the precise cultivation of the Pseudomonas kilonensis strain in a specialized 216L medium, where parameters such as pH, temperature, and agitation speed are tightly controlled to maximize cell density and enzyme expression. Following fermentation, the cells are harvested and prepared as a concentrated biocatalyst, which is then introduced to the reaction vessel containing the ketone substrate and amino donor. The detailed standardized synthesis steps, including specific concentrations of buffers and co-substrates required for optimal performance, are outlined in the technical guide below to assist process engineers in replicating these results.
- Cultivate Pseudomonas kilonensis strains in 216L medium containing peptone and yeast powder at 30°C for 72 hours to obtain fermentation broth.
- Centrifuge the fermentation broth to harvest cells, then resuspend and wash them with Tris-HCl buffer to prepare the concentrated cell liquid.
- Add ketone substrate, amino donor, and glucose for coenzyme regeneration to the cell liquid, maintaining pH 7-13 and temperature 0-70°C for asymmetric reductive amination.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this marine strain technology offers tangible strategic advantages that extend beyond mere technical feasibility. The shift from chemical synthesis to biocatalysis fundamentally alters the cost structure of chiral amine manufacturing by eliminating the dependency on volatile precious metal markets and reducing the complexity of waste management. Since the process operates in an aqueous environment with biodegradable by-products, the costs associated with hazardous waste disposal and environmental compliance are significantly lowered. This transition not only improves the margin profile of the final product but also mitigates regulatory risks associated with heavy metal residues, ensuring a smoother path through quality audits and customer approvals.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and chiral resolving agents leads to substantial cost savings in raw material procurement. By utilizing glucose for cofactor regeneration, the process avoids the need for stoichiometric quantities of costly reducing agents, further driving down the variable costs per kilogram. Additionally, the high yield and optical purity reduce the loss of valuable starting materials, meaning less feedstock is required to produce the same amount of saleable product. These efficiencies compound over large production volumes, resulting in a more competitive pricing structure that can be passed on to customers or retained as improved gross margin.
- Enhanced Supply Chain Reliability: Relying on fermentation-based production diversifies the supply chain away from petrochemical-dependent synthetic routes that are susceptible to oil price fluctuations and geopolitical instability. The raw materials for this bioprocess, such as glucose and ammonium salts, are commodity chemicals with stable and abundant global supplies, ensuring continuity of production even during market disruptions. Furthermore, the scalability of fermentation technology allows for rapid capacity expansion by simply increasing fermenter volume, providing the agility needed to respond to sudden spikes in demand from downstream pharmaceutical clients without long lead times for new equipment installation.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system simplifies the scale-up process from laboratory to commercial production, as heat and mass transfer issues are more manageable compared to exothermic chemical reductions. This ease of scale-up reduces the time and capital investment required for process validation and technology transfer. From an environmental perspective, the reduction in organic solvent usage and the absence of toxic metal waste align with increasingly stringent global environmental regulations, future-proofing the manufacturing site against tighter emission standards. This sustainability profile enhances the brand value of the supply chain, appealing to end-users who prioritize green sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this technology, we have compiled a set of questions based on the specific data and claims found within the patent documentation. These answers provide clarity on the performance metrics, substrate scope, and operational parameters that define the capabilities of this marine strain catalytic system. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What is the optical purity achievable with this marine strain method?
A: According to patent CN103224963A, the optical purity of the obtained chiral amine products consistently reaches above 90% ee, with specific examples demonstrating values as high as 99.0% ee.
Q: How does this biocatalytic method compare to traditional chemical synthesis?
A: Unlike traditional chemical methods that often require harsh conditions and expensive metal catalysts, this marine strain method operates under mild aqueous conditions, eliminates heavy metal residues, and achieves yields exceeding 70%.
Q: What types of ketone substrates are compatible with this process?
A: The process demonstrates broad substrate tolerance, successfully converting aliphatic ketones, aromatic ketones, and hydroxy ketones with carbon chain lengths ranging from 2 to 10 into their corresponding chiral amines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the marine strain catalysis technology disclosed in patent CN103224963A for the production of high-value chiral amines. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are successfully translated into robust industrial realities. Our facilities are equipped with state-of-the-art fermentation and downstream processing units capable of handling sensitive biocatalytic reactions with stringent purity specifications. We maintain rigorous QC labs that employ advanced analytical techniques to verify optical purity and impurity profiles, guaranteeing that every batch meets the exacting standards required by global pharmaceutical regulators.
We invite you to collaborate with us to leverage this advanced biocatalytic route for your next project, optimizing your supply chain for both cost and quality. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this technology can improve your bottom line. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics rather than theoretical projections. Together, we can accelerate the development of your chiral intermediates and secure a competitive advantage in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
