Advanced Iridium-Catalyzed Asymmetric Hydrogenation for High-Purity Chiral Amines
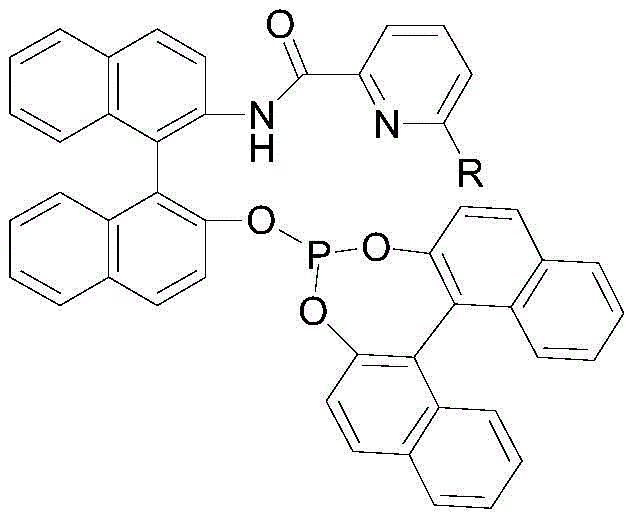
The landscape of chiral amine synthesis has been significantly advanced by the innovations detailed in patent CN110551036B, which discloses a highly efficient iridium/chiral phosphite ester-pyridine catalyzed imine asymmetric hydrogenation method. This technology represents a pivotal shift in the manufacturing of optically active amines, which serve as critical building blocks for bioactive compounds, particularly in the agrochemical sector. The core breakthrough involves the utilization of a novel chiral phosphite-pyridine (P, N) ligand that coordinates with a metal iridium precursor to form a complex in situ. This catalytic system addresses long-standing challenges in the industry, such as the need for excessive catalyst loading and the difficulty in achieving high enantiomeric excess (ee) values simultaneously. By enabling a substrate-to-catalyst (S/C) ratio of up to 500,000 in the synthesis of key intermediates like those for metolachlor, this method offers a compelling value proposition for manufacturers seeking to optimize both purity and production costs. The patent data indicates that this approach not only simplifies the operational workflow but also delivers exceptional stereocontrol, with ee values consistently exceeding 85 percent and yields reaching up to 95 percent under optimized conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of imines to produce chiral amines has been plagued by significant technical and economic hurdles. Early methodologies, such as those reported by Levi et al. in 1975, suffered from disappointingly low enantiomeric excess values, often hovering around merely 22 percent, which rendered them commercially unviable for high-specification applications. Subsequent advancements, including the industrialized process by Hans-Peter Jalett et al. utilizing ferrocene bisphosphine ligands, improved the ee value to approximately 76 percent but introduced new complexities. These conventional systems typically demand harsh reaction conditions, specifically requiring hydrogen pressures as high as 80 atmospheres and temperatures around 50°C, alongside the necessity for substantial amounts of acid additives and iodide salts. Furthermore, the synthesis of the ferrocene-based ligands themselves is notoriously difficult and costly, involving multi-step procedures that complicate the supply chain. The requirement for corrosive acid additives also imposes stringent demands on reactor materials, increasing capital expenditure for equipment capable of withstanding such aggressive environments. Consequently, these legacy methods often result in higher operational costs and limited flexibility regarding substrate scope.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a chiral phosphite-pyridine ligand system that fundamentally alters the efficiency profile of the hydrogenation process. This new catalytic architecture allows for the generation of the active iridium species in situ under much milder conditions, eliminating the absolute necessity for the large quantities of acid additives required by previous generations of catalysts. The structural design of the ligand, featuring a binaphthyl backbone coupled with a pyridine-amide moiety, creates a highly defined chiral pocket that effectively discriminates between prochiral faces of the imine substrate. This results in a dramatic improvement in stereoselectivity, with documented enantiomeric excess values surpassing 85 percent and reaching as high as 91 percent for specific agrochemical intermediates. Moreover, the operational simplicity is enhanced by the ability to conduct the reaction in common organic solvents like dichloromethane or toluene without the need for exotic additives. The robustness of this system is further evidenced by its tolerance to a wide range of reaction parameters, maintaining high activity across a temperature window of 20-100°C and pressure range of 20-100 bar, thereby providing process engineers with greater flexibility in optimizing throughput and safety.
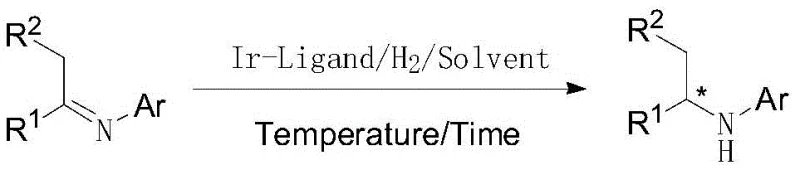
Mechanistic Insights into Iridium/Chiral Phosphite-Pyridine Catalyzed Hydrogenation
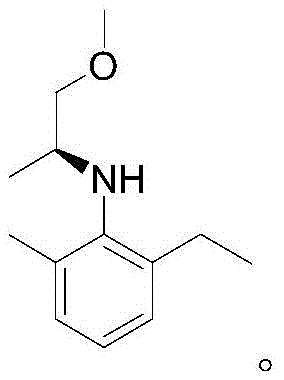
The exceptional performance of this catalytic system can be attributed to the unique electronic and steric properties of the chiral phosphite-pyridine (P, N) ligand. The ligand is constructed from a rigid binaphthyl scaffold, which provides the necessary chirality, linked to a pyridine ring via an amide bond and a phosphite group. This specific arrangement facilitates a bidentate coordination to the iridium center, forming a stable yet reactive cationic or neutral complex depending on the counterion used. The phosphite moiety acts as a strong sigma-donor and pi-acceptor, modulating the electron density at the metal center to facilitate the oxidative addition of hydrogen. Simultaneously, the pyridine nitrogen provides a secondary coordination point that locks the ligand into a specific conformation, preventing unwanted isomerization and ensuring that the chiral information is effectively transferred to the substrate during the hydride transfer step. The steric bulk of the binaphthyl groups creates a confined environment around the active site, forcing the imine substrate to approach in a specific orientation that favors the formation of one enantiomer over the other. This precise spatial control is crucial for achieving the high ee values observed in the experimental data, particularly for sterically demanding substrates like 2-methyl-6-ethylaniline derivatives.
Regarding impurity control, the mechanism inherently minimizes side reactions due to the high specificity of the catalyst-substrate interaction. In traditional hydrogenation processes, non-selective reduction or over-reduction can lead to complex impurity profiles that are difficult and expensive to separate. However, the well-defined coordination sphere of the iridium-phosphite-pyridine complex ensures that the activation of hydrogen and the subsequent insertion into the C=N bond occur with high fidelity. The mild reaction conditions further contribute to a cleaner reaction profile by reducing thermal degradation of sensitive functional groups on the substrate or product. Additionally, the in situ generation of the catalyst allows for immediate consumption of the active species, reducing the likelihood of catalyst decomposition products contaminating the final mixture. The patent data highlights that even at extremely high turnover numbers (S/C = 500,000), the conversion remains above 99 percent with minimal byproduct formation, indicating a highly robust catalytic cycle that resists deactivation. This level of purity is paramount for downstream applications in pharmaceutical and agrochemical synthesis, where strict regulatory limits on impurities must be met.
How to Synthesize Chiral Phosphite-Pyridine Ligand Efficiently
The synthesis of the key chiral ligand is a critical upstream process that determines the overall cost and viability of the catalytic method. The patent outlines a streamlined two-step procedure that avoids the use of hazardous reagents and complex purification techniques often associated with chiral ligand preparation. The first step involves the condensation of picolinic acid with (S)-NOBIN in the presence of a dehydration agent like DMTMM, forming an amide intermediate with high efficiency. This is followed by a phosphitylation reaction with (S)-MonoPhos in refluxing toluene, which precipitates the final ligand as a white solid, simplifying isolation. This straightforward synthetic route ensures that the ligand can be produced on a kilogram scale with consistent quality, supporting the continuous operation capabilities mentioned in the patent. For detailed standardized synthesis steps and specific molar ratios, please refer to the guide below.
- Condense picolinic acid with (S)-NOBIN using DMTMM in THF at room temperature to form the amide intermediate, followed by aqueous workup and silica gel chromatography.
- React the amide intermediate with (S)-MonoPhos in refluxing toluene under nitrogen to generate the chiral phosphite-pyridine ligand via precipitation.
- Mix the ligand with an iridium-cyclooctadiene precursor in solvent to form the catalyst in situ, then hydrogenate the imine substrate at 20-100 bar and 20-100°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iridium-catalyzed technology translates into tangible strategic advantages beyond mere technical performance. The primary driver for cost optimization lies in the unprecedented turnover number (TON) achieved by the catalyst system. With a demonstrated substrate-to-catalyst ratio of up to 500,000:1, the consumption of the expensive iridium metal and the chiral ligand is drastically reduced compared to conventional methods that might operate at ratios of 1,000:1 or lower. This reduction in precious metal loading directly lowers the variable cost per kilogram of the final chiral amine product. Furthermore, the elimination of large quantities of acid additives and iodide salts simplifies the downstream processing workflow. Traditional methods often require extensive washing and neutralization steps to remove these corrosive additives, generating significant volumes of aqueous waste. By operating with a cleaner catalytic system, the new method reduces the burden on waste treatment facilities and lowers the consumption of auxiliary chemicals, contributing to a more sustainable and cost-effective manufacturing process.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the drastic reduction in catalyst loading. Since the catalyst is effective at parts-per-million levels relative to the substrate, the direct material cost associated with the noble metal and chiral ligand becomes negligible in the overall bill of materials. Additionally, the simplified workup procedure, which does not require aggressive acid quenching or complex salt removal, reduces the operational time and utility consumption (steam, water, electricity) per batch. The high yield of 95 percent further maximizes the utilization of raw materials, minimizing the loss of valuable starting imines to side reactions or incomplete conversion. These factors combined create a significantly leaner cost structure for the production of high-value chiral intermediates.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of the ligand synthesis ensures a stable source of the catalytic system. The starting materials, such as (S)-NOBIN and picolinic acid derivatives, are commercially available and do not rely on obscure or single-source suppliers. The ligand synthesis itself is high-yielding and produces a stable solid that can be stored and transported without special handling requirements, unlike some air-sensitive phosphine ligands. This stability reduces the risk of supply disruptions caused by catalyst degradation during logistics. Moreover, the ability of the catalyst to perform well across a broad range of temperatures and pressures provides operational resilience; if there are fluctuations in utility availability (e.g., cooling water temperature), the process can be adjusted within the wide operating window without compromising product quality, ensuring consistent delivery schedules.
- Scalability and Environmental Compliance: The method is explicitly designed for industrial scalability, having been validated for the synthesis of metolachlor intermediates which are produced in massive volumes globally. The use of standard solvents like toluene and dichloromethane, combined with moderate pressure requirements (20-100 bar), means the process can be implemented in existing hydrogenation infrastructure without the need for specialized high-pressure autoclaves rated for extreme conditions. Environmentally, the high atom economy and reduced waste generation align with modern green chemistry principles. The absence of heavy metal contaminants in the product (due to low catalyst loading) simplifies the purification process, potentially eliminating the need for expensive scavenging resins or activated carbon treatments often required to meet strict residual metal specifications in agrochemical and pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy for R&D and process development teams evaluating this route. Understanding these nuances is essential for assessing the feasibility of integrating this catalytic system into existing production lines or new product development pipelines.
Q: What represents the primary advantage of this iridium catalyst system over traditional ferrocene-based methods?
A: The primary advantage lies in the significantly higher turnover number (TON) and enantioselectivity. While traditional ferrocene bisphosphine systems often require harsh conditions (80 atm) and achieve lower ee values (around 76%), this novel iridium/phosphite-pyridine system operates efficiently with a substrate-to-catalyst ratio as high as 500,000:1, achieving up to 91% ee and 95% yield under milder conditions.
Q: Is this catalytic method suitable for large-scale industrial production of metolachlor intermediates?
A: Yes, the method is explicitly designed for industrial practicability. The patent demonstrates exceptional performance in the synthesis of the metolachlor intermediate (2-ethyl-6-methylaniline derivative), achieving high conversion rates (>99%) and excellent enantioselectivity. The simplicity of the ligand synthesis and the robustness of the catalyst under standard hydrogenation pressures make it highly scalable.
Q: What are the typical reaction conditions required for this asymmetric hydrogenation?
A: The reaction typically proceeds in solvents such as dichloromethane, 1,2-dichloroethane, or toluene. The hydrogen pressure ranges from 20 to 100 bar, and the temperature is maintained between 20°C and 100°C. The catalyst is generated in situ by stirring the iridium precursor and ligand at room temperature for 2 to 5 hours prior to substrate addition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iridium/chiral phosphite-pyridine catalyzed hydrogenation method for the production of high-purity chiral amines. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory success into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity and yields observed in the patent are maintained at an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced chiral HPLC and GC capabilities to verify the optical purity of every batch, guaranteeing that our chiral amine intermediates meet the exacting standards required by global agrochemical and pharmaceutical companies.
We invite you to collaborate with us to leverage this advanced technology for your specific supply chain needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-TON catalytic system for your specific molecule. We encourage you to contact our technical procurement team today to request specific COA data for our chiral amine portfolio and to discuss route feasibility assessments tailored to your project timelines. Let us help you secure a reliable, cost-effective, and high-quality supply of critical chiral intermediates.
