Advanced Phosphonate Chemistry for Scalable Loratadine Intermediate Production
Advanced Phosphonate Chemistry for Scalable Loratadine Intermediate Production
The pharmaceutical landscape for antihistamine production has been significantly reshaped by the innovations disclosed in patent CN1334810A, which introduces a robust and chemically elegant process for preparing tricyclic compounds with antihistaminic activity, most notably loratadine and its metabolite descarboethoxyloratadine (DCL). This patent represents a critical pivot away from legacy synthetic routes that relied on hazardous reagents and unstable intermediates, offering instead a pathway grounded in phosphonate chemistry that ensures higher purity and operational safety. For R&D directors and process chemists, the transition from Grignard-based methodologies to this phosphonate carbanion approach solves long-standing issues regarding conjugate addition side reactions and thermal instability. The core innovation lies in the reaction of a specific tricyclic ketone with a stabilized phosphonate carbanion, followed by a thermal decomposition step that cleanly generates the requisite exocyclic double bond without the need for harsh acidic dehydration or toxic halogenating agents. This technical breakthrough not only enhances the chemical integrity of the final API intermediate but also streamlines the manufacturing workflow, making it an ideal candidate for reliable loratadine intermediate supplier networks seeking to modernize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in U.S. Patent 4,659,716, relied heavily on the coupling of a tricyclic ketone with a Grignard reagent derived from 4-chloro-N-methylpiperidine. This historical approach is fraught with significant chemical and logistical disadvantages that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Firstly, the precursor 4-chloro-N-methylpiperidine itself requires a cumbersome five-step synthesis, introducing multiple points of failure and yield loss before the key coupling even begins. Furthermore, this halide is thermally unstable at temperatures above room temperature, necessitating strict cold chain management and immediate consumption upon preparation, which creates severe bottlenecks in large-scale manufacturing environments. The subsequent Grignard reaction with the tricyclic ketone typically suffers from low yields, often hovering around 60%, due to competing conjugate addition reactions where the nucleophile attacks the pyridine ring instead of the carbonyl group. Additionally, the dehydration of the resulting alcohol intermediate is highly sensitive, often leading to isomerization of the double bond, while the final Von Braun reaction to install the carbamate group generates toxic methyl chloride gas, imposing heavy costs for gas scrubbing and environmental compliance.
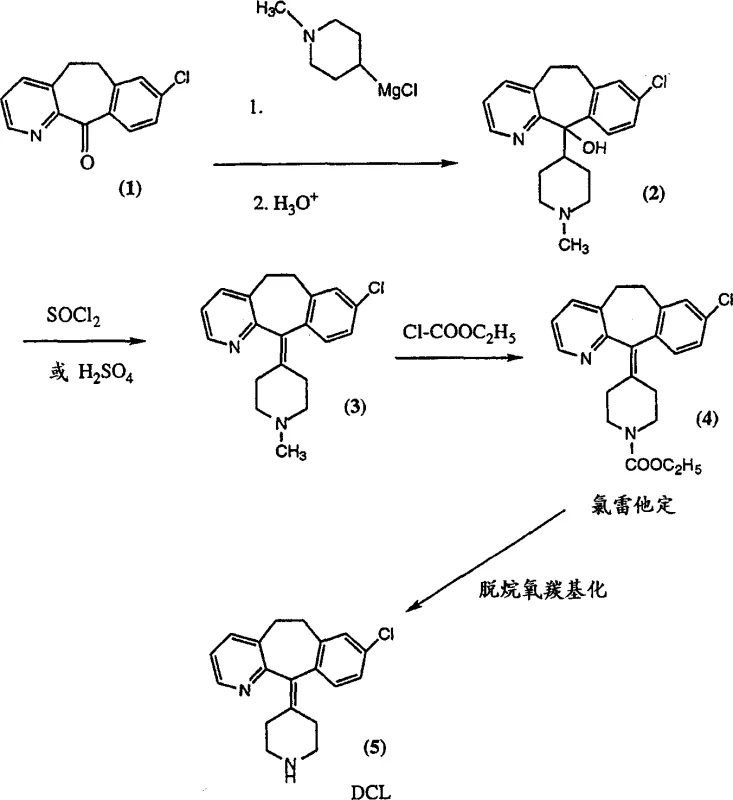
The Novel Approach
In stark contrast, the novel process disclosed in CN1334810A circumvents these pitfalls by utilizing a phosphonate-based carbanion derived from a thermally stable piperidine-phosphonate intermediate. This new route allows for the direct conversion of the starting ketone to the desired loratadine structure in a potential one-pot procedure, effectively eliminating the need to isolate the sensitive beta-hydroxy intermediate. The phosphonate reagent is significantly more stable than its Grignard counterpart, allowing for greater flexibility in reaction timing and temperature control. By replacing the acidic dehydration and toxic Von Braun steps with a single thermal decomposition phase, the process inherently improves the impurity profile by avoiding isomerization and eliminating the generation of hazardous methyl chloride. This shift not only simplifies the reactor train requirements but also drastically reduces the environmental footprint of the synthesis, aligning perfectly with modern green chemistry initiatives and cost reduction in API manufacturing strategies.
Mechanistic Insights into Phosphonate Carbanion Addition and Thermal Elimination
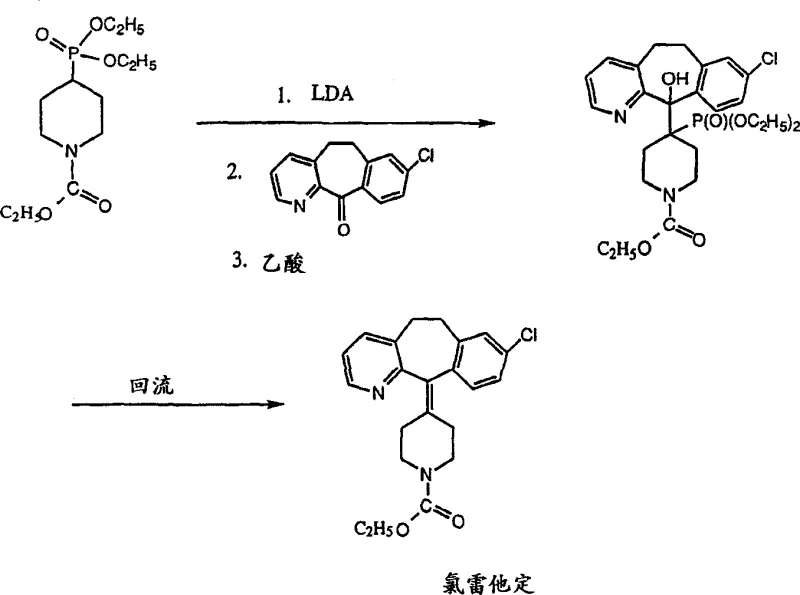
The mechanistic elegance of this process begins with the generation of the nucleophile. The phosphonate intermediate, specifically N-ethoxycarbonylpiperidine-4-phosphonate, is treated with a strong non-nucleophilic base, preferably lithium diisopropylamide (LDA), in an aprotic solvent system such as tetrahydrofuran (THF) or a mixture of THF and xylene. This deprotonation occurs optimally at cryogenic temperatures ranging from -40°C to -20°C, ensuring the formation of the reactive carbanion without inducing premature decomposition or side reactions. Once formed, this carbanion attacks the carbonyl carbon of the tricyclic ketone (8-chloro-11,12-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one) to form an unstable alkoxide species. Unlike the Grignard addition which is prone to 1,4-conjugate addition on the pyridine ring, the phosphonate carbanion exhibits high chemoselectivity for the 1,2-addition to the ketone, thereby preserving the aromatic integrity of the pyridine moiety and maximizing the yield of the desired adduct.
Following the nucleophilic attack, the reaction mixture is quenched with a protonating agent, with anhydrous acetic acid being particularly preferred for one-pot operations. This protonation converts the alkoxide into a beta-hydroxy phosphonate intermediate. The final transformation involves the thermal decomposition of this beta-hydroxy species. By heating the reaction mixture to reflux temperatures between 110°C and 160°C, preferably 130°C to 140°C in a high-boiling solvent like xylene, the molecule undergoes an elimination reaction. This thermal step cleaves the phosphonate group and the hydroxyl group simultaneously to form the exocyclic double bond characteristic of loratadine. This mechanism avoids the harsh acidic conditions required in traditional dehydration, which are known to cause double bond migration. Furthermore, the purification is straightforward; recrystallization from acetonitrile effectively removes trace amounts of unreacted ketone and phosphonate oxides, ensuring the delivery of high-purity antihistamine intermediates suitable for downstream pharmaceutical processing.
How to Synthesize Loratadine Efficiently
The synthesis of loratadine via this phosphonate route is designed for operational simplicity and high throughput, making it an attractive option for contract development and manufacturing organizations. The process leverages the stability of the phosphonate reagent to enable a streamlined workflow that minimizes unit operations and solvent exchanges. By combining the carbanion generation, nucleophilic addition, and thermal elimination into a cohesive sequence, manufacturers can reduce cycle times and improve overall equipment effectiveness. The detailed standardized synthesis steps for implementing this advanced protocol are outlined in the guide below, providing a clear roadmap for technical teams looking to adopt this superior methodology.
- Generate the phosphonate carbanion by treating N-ethoxycarbonylpiperidine-4-phosphonate with LDA in a THF and xylene mixture at temperatures between -40°C and -20°C.
- React the carbanion with the tricyclic ketone precursor and quench the resulting alkoxide with anhydrous acetic acid to form the beta-hydroxy phosphonate intermediate.
- Thermally decompose the intermediate by refluxing the mixture at 130°C to 140°C to eliminate the phosphonate group and form the final double bond of loratadine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the process described in CN1334810A offers tangible benefits that extend beyond mere chemical yield improvements. The primary advantage lies in the drastic simplification of the raw material supply chain. The phosphonate intermediate can be synthesized from pyridine in just two steps with high efficiency, compared to the five-step synthesis required for the unstable chloro-piperidine used in legacy methods. This reduction in synthetic complexity translates directly into a more resilient supply chain, as there are fewer precursors to source and qualify. Moreover, the thermal stability of the phosphonate reagent means it can be produced in large batches, stored, and shipped without the degradation risks associated with the prior art Grignard precursors, thereby enhancing supply chain reliability and reducing the risk of production stoppages due to reagent spoilage.
- Cost Reduction in Manufacturing: The elimination of the Von Braun reaction removes the need for expensive and hazardous chloroformate reagents and the associated infrastructure for handling toxic methyl chloride gas. By replacing this with a thermal decomposition step, the process significantly lowers the capital expenditure required for gas scrubbing systems and waste treatment facilities. Furthermore, the ability to run the reaction in a one-pot fashion reduces solvent consumption and labor hours associated with intermediate isolation and drying. These qualitative efficiencies drive down the cost of goods sold (COGS) without compromising on quality, allowing for more competitive pricing in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The inherent stability of the key phosphonate intermediate mitigates the risks of supply disruption. Unlike the thermally unstable 4-chloro-N-methylpiperidine which must be used immediately after preparation, the phosphonate reagent can be stockpiled, providing a buffer against upstream supply fluctuations. This stability also simplifies logistics, as the reagent does not require specialized cold-chain transport or immediate processing upon arrival at the manufacturing site. Consequently, production planning becomes more predictable, and lead times for high-purity pharmaceutical intermediates can be consistently met, fostering stronger relationships with downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is vastly superior. The absence of toxic methyl chloride emissions simplifies regulatory compliance and reduces the environmental liability of the manufacturing site. The thermal decomposition step is clean and generates benign phosphorus byproducts that are easier to manage than halogenated waste streams. This green chemistry profile facilitates easier permitting for capacity expansion and aligns with the sustainability goals of major pharmaceutical companies. The robustness of the reaction conditions also ensures that the process can be scaled from pilot plant to multi-ton commercial production with minimal re-optimization, ensuring a smooth technology transfer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphonate-based synthesis route. These answers are derived directly from the experimental data and specifications provided in the patent literature, ensuring accuracy for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: How does the phosphonate route improve safety compared to the traditional Grignard method?
A: The traditional Von Braun reaction produces toxic methyl chloride gas requiring complex scrubbing systems. The phosphonate route utilizes thermal decomposition, eliminating hazardous gaseous byproducts and significantly reducing environmental compliance burdens.
Q: Why is the phosphonate intermediate considered more stable for supply chain logistics?
A: Unlike the 4-chloro-N-methylpiperidine Grignard precursor which is thermally unstable and requires immediate use, the phosphonate intermediate is thermally stable and can be stored or transported, allowing for flexible batch scheduling and reduced waste.
Q: Can the beta-hydroxy intermediate be isolated if necessary?
A: Yes, the process allows for the isolation of the beta-hydroxy phosphonate intermediate using water extraction and crystallization, although the preferred industrial method is a one-pot procedure to maximize efficiency and yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Loratadine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the phosphonate-based synthesis route for producing high-value antihistamine intermediates like loratadine. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal conditions required for this chemistry, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the global pharmaceutical industry. We are committed to delivering consistent quality and supply continuity for your critical API needs.
We invite you to collaborate with us to optimize your supply chain for loratadine and related tricyclic compounds. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this safer, more efficient process. Contact us today to request specific COA data and route feasibility assessments tailored to your production volume requirements, and let us demonstrate how our expertise can drive value and efficiency in your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
