Advanced Purification Technology for High-Purity Crisaborole Meeting ICH Standards
Introduction to Patent CN115850305A
The pharmaceutical industry continuously seeks robust methodologies to ensure the highest purity standards for active pharmaceutical ingredients, particularly for complex molecules like Crisaborole, a non-steroidal phosphodiesterase 4 inhibitor. Patent CN115850305A introduces a groundbreaking refining method specifically designed to produce high-purity Crisaborole, addressing critical challenges in impurity control and process scalability. This technology represents a significant leap forward from traditional purification techniques, offering a streamlined pathway to achieve pharmaceutical grade material that strictly adheres to ICH guidelines. By optimizing the crystallization dynamics through a unique combination of organic solvents and acidic conditions, this method ensures the consistent production of the desired crystal Form I while drastically minimizing the presence of toxic or genotoxic impurities. The strategic integration of activated carbon treatment within the hot filtration step further enhances the optical clarity and chemical purity of the final product.
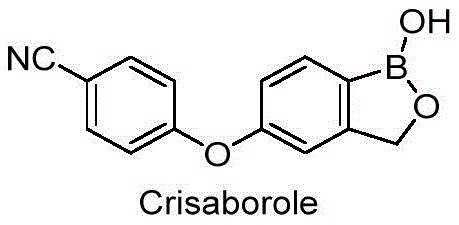
For R&D directors and process chemists, the ability to reliably produce material with unknown single impurities below 0.1% is paramount for regulatory approval and clinical success. The disclosed method not only simplifies the operational workflow by avoiding multi-step solvent exchanges but also significantly reduces the environmental footprint associated with waste solvent generation. This dual benefit of operational simplicity and environmental compliance makes it an attractive candidate for adoption by reliable API intermediate suppliers aiming to optimize their manufacturing portfolios. As we delve deeper into the technical specifics, it becomes evident that this refinement strategy solves long-standing bottlenecks in the commercial production of this vital dermatological agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Crisaborole has relied on labor-intensive and environmentally taxing techniques that struggle to meet modern regulatory demands. Traditional approaches often involve silica gel column chromatography, which, while effective on a laboratory scale, presents insurmountable challenges for commercial scale-up due to high solvent consumption and low throughput. Furthermore, prior art methods utilizing glacial acetic acid-water systems often result in products with residual solvent issues, as the high boiling point of acetic acid makes complete removal difficult and energy-intensive. Other recrystallization attempts using ethyl acetate and n-heptane systems have failed to consistently reduce unknown single impurities below the critical 0.1% threshold required by ICH standards. These conventional pathways frequently leave behind persistent process impurities such as dehalogenation byproducts and hydrolysis derivatives, compromising the safety profile of the final drug substance.
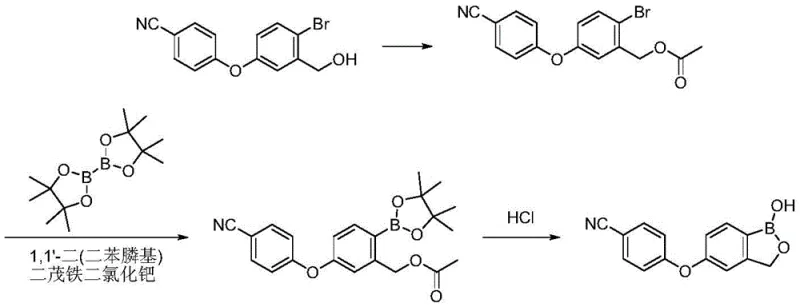
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated yet operationally simple recrystallization protocol that leverages the solubility differences between the target molecule and its impurities. By dissolving the crude product in a selected organic solvent like methanol or acetone in the presence of concentrated hydrochloric acid, the process creates a thermodynamic environment favorable for the exclusion of impurities. The addition of activated carbon acts as a powerful scavenger for colored bodies and trace organic contaminants, ensuring a pristine final appearance. Crucially, the controlled addition of water as an anti-solvent at elevated temperatures prevents premature precipitation, allowing for the formation of large, well-defined crystals of Form I upon cooling. This method effectively bypasses the need for complex ion exchange columns or multiple recrystallization cycles, delivering a product with purity levels reaching up to 100.0% in optimized embodiments.
Mechanistic Insights into Impurity Control and Crystal Engineering
The success of this refining method lies in its precise manipulation of intermolecular interactions and solubility parameters to selectively isolate the target compound. The presence of concentrated hydrochloric acid likely protonates basic impurities or modifies the solvation shell of the benzoxaborole ring, altering its solubility relative to neutral organic byproducts. This acid-mediated solubilization ensures that the target Crisaborole remains in solution during the hot filtration step, while insoluble particulate matter and polymeric side-products are retained on the filter cake or adsorbed onto the activated carbon. The subsequent anti-solvent addition of water reduces the dielectric constant of the medium, triggering nucleation specifically for the thermodynamically stable Form I crystal lattice. This controlled crystallization kinetics prevents the occlusion of mother liquor containing dissolved impurities, thereby yielding a solid with exceptional chemical homogeneity.
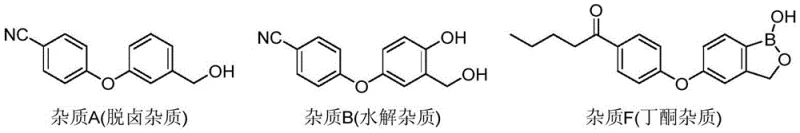
Furthermore, the mechanism effectively targets the specific structural analogues that typically plague Crisaborole synthesis, such as Impurity A, Impurity B, and Impurity F. Impurity A, a dehalogenation byproduct often arising from palladium-catalyzed coupling, and Impurity B, a hydrolysis product, possess different polarity profiles compared to the nitrile-substituted target. The chosen solvent system exploits these polarity differences, keeping these polar impurities in the aqueous-organic mother liquor during the precipitation phase. Similarly, Impurity F, a ketone derivative associated with organolithium routes, is effectively suppressed through the rigorous washing and crystallization steps. The result is a refined product where key impurities are driven down to levels lower than 0.05%, often becoming undetectable by HPLC, which is a critical metric for ensuring patient safety and regulatory compliance in topical formulations.
How to Synthesize Crisaborole Efficiently
The implementation of this refining protocol requires careful attention to temperature control and reagent ratios to maximize yield and purity. The process begins with the dissolution of the crude material in a warm organic solvent mixture containing hydrochloric acid, followed by a critical decolorization step. Operators must maintain the filtrate temperature between 55°C and 75°C during the water addition to prevent 'oiling out' or amorphous precipitation, which could trap impurities. Detailed standard operating procedures regarding the specific mass-to-volume ratios of solvents and the cooling ramp rates are essential for reproducibility. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized steps outlined below.
- Mix crude Crisaborole with an organic solvent (such as methanol, acetone, DMF, or acetonitrile) and concentrated hydrochloric acid, then heat to 55-75°C to fully dissolve the mixture.
- Add activated carbon (3-5% of crude mass) to the hot solution, stir for decolorization and impurity adsorption, and perform hot filtration to remove solid particulates.
- Add purified water to the hot filtrate as an anti-solvent, cool the mixture to below 40°C to precipitate the pure crystal form I, and filter to obtain the finished product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this refining technology offers transformative benefits that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By eliminating the need for silica gel chromatography and reducing the number of recrystallization cycles from two to one, the process drastically cuts down on solvent procurement costs and waste disposal fees. The simplified workflow reduces the overall processing time, allowing facilities to increase batch turnover rates without requiring additional capital investment in specialized equipment. This efficiency gain translates into a more agile supply chain capable of responding rapidly to market demand fluctuations for high-purity API intermediates. Moreover, the use of common, commercially available solvents like methanol and acetone mitigates the risk of supply disruptions associated with exotic or highly regulated reagents.
- Cost Reduction in Manufacturing: The elimination of expensive chromatography resins and the reduction in total solvent volume lead to substantial cost savings in raw material expenditure. By streamlining the purification into a single pot operation with hot filtration, labor costs and energy consumption for solvent recovery are significantly minimized. The high yield achieved in this process ensures that valuable crude material is not lost to excessive mother liquor retention or multiple handling steps, maximizing the return on investment for every kilogram of input material.
- Enhanced Supply Chain Reliability: The robustness of this method against variations in crude quality ensures consistent output, reducing the risk of batch failures that can disrupt supply schedules. Since the process relies on standard unit operations like dissolution, filtration, and crystallization, it can be easily transferred between different manufacturing sites or scaled up from pilot to commercial production with minimal tech transfer friction. This flexibility empowers procurement managers to diversify their supplier base without compromising on product quality specifications.
- Scalability and Environmental Compliance: The significant reduction in waste solvent generation aligns perfectly with green chemistry principles and increasingly stringent environmental regulations. The process avoids the use of chlorinated solvents often associated with extraction workflows, simplifying the permitting process for manufacturing facilities. The ease of scaling this recrystallization method means that production capacity can be expanded linearly to meet growing global demand for Crisaborole-based dermatological treatments without encountering the bottlenecks typical of chromatographic purifications.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel refining technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy techniques. Understanding these nuances is crucial for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines.
Q: How does this refining method improve upon traditional silica gel chromatography?
A: Traditional silica gel chromatography is difficult to scale industrially and generates significant solvent waste. This novel method utilizes a simple recrystallization technique with common organic solvents and hydrochloric acid, eliminating the need for column chromatography and enabling easy industrial scale-up while meeting strict ICH impurity standards.
Q: Which specific impurities are effectively removed by this process?
A: The process is highly effective at removing key process-related impurities including Impurity A (dehalogenation impurity), Impurity B (hydrolysis impurity), and Impurity F (butanone impurity). The refined product consistently achieves a single impurity content lower than 0.05%, often resulting in undetectable levels of these specific contaminants.
Q: What solvents are compatible with this purification protocol?
A: The method demonstrates flexibility with several polar organic solvents. Validated embodiments include methanol, acetone, N,N-Dimethylformamide (DMF), and acetonitrile. The choice of solvent can be optimized based on availability and cost, with methanol and acetone showing particularly high yields and purity profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Crisaborole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of complex pharmaceutical intermediates like Crisaborole. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity Crisaborole that meets stringent purity specifications, leveraging advanced refining technologies similar to the one described in CN115850305A to guarantee product quality. Our rigorous QC labs employ state-of-the-art analytical instrumentation to verify that every batch conforms to ICH guidelines, providing our partners with the confidence they need to advance their drug development pipelines.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through our tailored solutions. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates the potential economic benefits of switching to our refined production methods. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how our capabilities align with your project requirements. Let us help you secure a stable, high-quality supply of Crisaborole that supports your long-term commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
