Scalable Metal-Free Nitrile Synthesis Strategy for High-Purity Pharmaceutical Intermediates
The global demand for high-purity nitrile compounds continues to surge, driven by their critical role as versatile intermediates in the synthesis of active pharmaceutical ingredients (APIs), agrochemicals, and advanced functional materials. However, traditional manufacturing routes often face significant hurdles regarding safety, environmental compliance, and operational complexity. A groundbreaking technical solution has emerged through patent CN114409571B, which discloses a highly efficient, metal-free methodology for converting aldehydes directly into nitriles. This innovation leverages imidazole hydrochloride as a key reaction auxiliary in a one-pot process, effectively bypassing the need for hazardous cyanide sources or corrosive strong acids. For procurement leaders and R&D teams seeking a reliable nitrile compound supplier, this technology represents a paradigm shift towards greener, safer, and more cost-effective manufacturing protocols that align with modern sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of nitrile compounds has relied heavily on methodologies that pose substantial risks to both personnel and the environment. Classic approaches frequently utilize剧毒 cyanation reagents such as sodium cyanide or potassium cyanide, which require stringent safety protocols, specialized containment infrastructure, and complex waste treatment systems to neutralize toxic byproducts. Furthermore, alternative dehydration strategies often necessitate the use of harsh dehydrating agents, strong oxidants, or expensive transition metal catalysts that can contaminate the final product with heavy metal residues. These conventional pathways typically operate under high temperature and high pressure conditions, demanding energy-intensive autoclaves and inert gas protection, which drastically inflate capital expenditure and operational overhead. The cumulative effect of these factors creates a fragile supply chain vulnerable to regulatory scrutiny and production bottlenecks, making cost reduction in pharmaceutical intermediate manufacturing increasingly difficult to achieve without compromising safety standards.
The Novel Approach
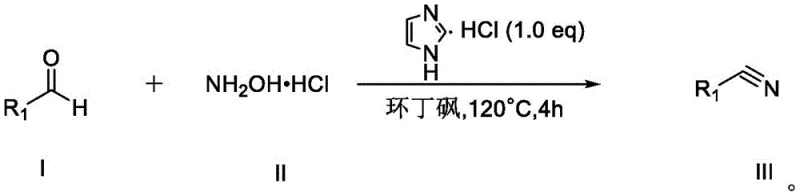
In stark contrast to these legacy methods, the novel approach detailed in the patent introduces a streamlined, one-pot synthesis that fundamentally reshapes the production landscape. By utilizing readily available aldehyde precursors and hydroxylamine hydrochloride in the presence of imidazole hydrochloride, the reaction proceeds smoothly under mild thermal conditions without the need for external pressure or inert atmospheres. This methodology eliminates the reliance on corrosive concentrated hydrochloric acid and toxic metal catalysts, thereby simplifying the reactor setup and reducing the burden on waste management systems. The process exhibits remarkable robustness, tolerating a wide array of functional groups including nitro, halo, hydroxy, and alkoxy substituents, which allows for the direct synthesis of complex intermediates without extensive protecting group strategies. This operational simplicity translates directly into enhanced process reliability and reduced lead time for high-purity nitrile compounds, offering a compelling value proposition for supply chain managers focused on continuity and efficiency.
Mechanistic Insights into Imidazole Hydrochloride-Promoted Dehydration
The core of this technological advancement lies in the unique catalytic role of imidazole hydrochloride, which acts as a potent promoter for the dehydration of the intermediate aldoxime species. Unlike traditional acid-catalyzed dehydrations that might require stoichiometric amounts of strong mineral acids leading to side reactions, the imidazole salt facilitates a controlled elimination of water under moderate thermal conditions. The reaction mechanism likely involves the activation of the hydroxyl group of the aldoxime intermediate through hydrogen bonding or proton transfer interactions mediated by the imidazolium cation, lowering the energy barrier for the elimination step. This precise control over the reaction pathway minimizes the formation of undesired byproducts such as amides or carboxylic acids, which are common impurities in less selective dehydration processes. The use of sulfolane as the solvent further enhances this selectivity due to its high boiling point and excellent solvating power for polar intermediates, ensuring a homogeneous reaction environment that maximizes molecular collisions and conversion rates.
From an impurity control perspective, the absence of transition metals is a critical advantage for pharmaceutical applications where residual metal limits are strictly regulated. The metal-free nature of this catalytic system ensures that the crude product stream is free from heavy metal contaminants, significantly reducing the complexity of downstream purification. Additionally, the mild reaction conditions prevent the degradation of sensitive functional groups on the aromatic ring or heterocyclic core, preserving the structural integrity of the molecule. This high level of chemoselectivity means that the final product profile is cleaner, requiring fewer crystallization steps or chromatographic separations to meet stringent quality specifications. For R&D directors, this implies a more predictable scale-up trajectory where the impurity profile established at the gram scale can be reliably maintained at the multi-ton commercial level, mitigating the risk of batch failures during technology transfer.
How to Synthesize Nitrile Compounds Efficiently
Implementing this synthesis route in a production environment requires adherence to specific operational parameters to maximize yield and purity. The protocol is designed for simplicity, utilizing commercially available starting materials that do not require specialized storage or handling beyond standard chemical safety practices. The reaction is typically conducted by charging the aldehyde substrate, hydroxylamine hydrochloride, and the imidazole hydrochloride promoter into a reaction vessel containing sulfolane. The mixture is then heated to an optimal temperature range of 110°C to 130°C, with 120°C identified as the sweet spot for balancing reaction rate and selectivity. Monitoring the reaction progress via thin-layer chromatography (TLC) allows operators to determine the precise endpoint, usually achieved within 4 hours, ensuring that resources are not wasted on unnecessary extended reaction times. Detailed standardized synthetic steps see the guide below.
- Charge a reaction vessel with the aldehyde substrate, hydroxylamine hydrochloride (1.0 equivalent), and imidazole hydrochloride (1.0 equivalent) in sulfolane solvent.
- Heat the reaction mixture to 120°C and stir for approximately 4 hours, monitoring progress via TLC until the starting material is consumed.
- Cool the mixture, quench with saturated brine, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers profound strategic benefits that extend beyond simple unit cost metrics. The primary advantage lies in the drastic simplification of the raw material portfolio; by replacing hazardous cyanide salts and expensive metal catalysts with benign, commodity-grade chemicals, companies can significantly reduce their exposure to volatile specialty chemical markets and strict regulatory controls. This shift not only lowers the direct cost of goods sold but also diminishes the administrative and compliance costs associated with handling scheduled substances. Furthermore, the robustness of the reaction conditions means that production can be executed in standard glass-lined or stainless steel reactors without the need for high-pressure autoclaves, thereby increasing asset utilization rates and freeing up specialized equipment for other critical processes. This flexibility enhances overall plant throughput and responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive scavenging resins and complex filtration steps typically required to meet heavy metal specifications in API manufacturing. This streamlining of the downstream processing workflow results in substantial cost savings by reducing solvent consumption, filtration media usage, and labor hours associated with purification. Additionally, the high atom economy of the one-pot transformation minimizes waste generation, leading to lower disposal costs and a smaller environmental footprint. The use of sulfolane, a recoverable solvent, further contributes to long-term economic efficiency by allowing for solvent recycling loops that decrease fresh solvent procurement requirements.
- Enhanced Supply Chain Reliability: Sourcing hydroxylamine hydrochloride and imidazole hydrochloride is far less prone to disruption compared to sourcing specialized cyanation reagents or proprietary metal catalysts, which often have limited supplier bases. The reliance on bulk commodity chemicals ensures a stable and continuous supply of raw materials, mitigating the risk of production stoppages due to vendor shortages. Moreover, the mild reaction conditions reduce the wear and tear on production equipment, extending the lifecycle of capital assets and minimizing unplanned maintenance downtime. This reliability is crucial for maintaining consistent delivery schedules to downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from gram to multi-kilogram scales without loss of yield or selectivity. The absence of toxic cyanide waste streams simplifies environmental compliance, as wastewater treatment becomes less complex and costly. This aligns perfectly with green chemistry principles, enhancing the corporate sustainability profile of the manufacturer. The ability to operate without inert gas protection or high-pressure vessels also reduces the energy intensity of the process, contributing to lower carbon emissions and supporting corporate net-zero initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this nitrile synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the safety advantages of this nitrile synthesis method compared to traditional cyanation?
A: Unlike traditional methods that rely on highly toxic cyanide reagents like sodium cyanide or potassium cyanide, this protocol utilizes hydroxylamine hydrochloride and imidazole hydrochloride. This eliminates the severe safety hazards and regulatory burdens associated with handling剧毒 cyanide salts, making it significantly safer for large-scale industrial operations.
Q: Does this method require expensive transition metal catalysts?
A: No, the process described in patent CN114409571B is entirely metal-free. It uses imidazole hydrochloride as a reaction auxiliary instead of costly transition metal catalysts. This not only reduces raw material costs but also simplifies downstream purification by removing the need for rigorous heavy metal scavenging steps to meet pharmaceutical purity standards.
Q: What is the substrate scope for this dehydration reaction?
A: The method demonstrates excellent functional group tolerance. It is effective for a wide range of aromatic aldehydes containing electron-withdrawing or electron-donating groups, heterocyclic aldehydes such as pyrrole and thiophene derivatives, and even aliphatic aldehydes, delivering high yields across diverse structural motifs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrile Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for producing high-value nitrile intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to delivering consistent quality and supply continuity for our global clientele.
We invite you to collaborate with us to optimize your supply chain for nitrile-based intermediates. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific target molecules, evaluating the economic viability of switching to this greener methodology. Please contact our technical procurement team to request specific COA data for relevant reference standards and comprehensive route feasibility assessments. Let us help you engineer a more resilient and cost-efficient supply chain for your critical building blocks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
