Advanced Catalytic Synthesis of Quinoline Derivatives for Commercial Pharmaceutical Applications
Advanced Catalytic Synthesis of Quinoline Derivatives for Commercial Pharmaceutical Applications
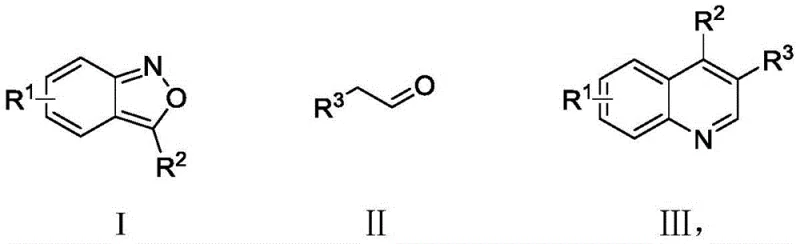
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for constructing bioactive heterocyclic scaffolds, particularly quinoline derivatives which serve as core structures in numerous therapeutic agents. Patent CN110204486B discloses a groundbreaking methodology for synthesizing these valuable compounds using 2,1-benzisoxazole derivatives and phenylacetaldehyde derivatives as key starting materials. This innovative approach leverages a dual copper and silver catalyst system under an oxygen atmosphere, offering a green and efficient alternative to traditional methods that often rely on harsh conditions or toxic reagents. For R&D directors and procurement specialists, this technology represents a significant opportunity to streamline supply chains for high-purity pharmaceutical intermediates while adhering to increasingly strict environmental regulations. The versatility of this reaction allows for the introduction of various functional groups, making it highly adaptable for the synthesis of diverse drug candidates and bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the quinoline skeleton frequently involve multi-step sequences that require expensive noble metal catalysts or stoichiometric amounts of hazardous oxidants. These conventional methods often suffer from poor atom economy, generating substantial quantities of chemical waste that necessitate complex and costly disposal procedures. Furthermore, many existing protocols demand extreme reaction conditions, such as very high temperatures or strong acidic environments, which can limit the tolerance of sensitive functional groups on the substrate. This lack of functional group compatibility often forces chemists to employ additional protection and deprotection steps, thereby increasing the overall production time and reducing the final yield. From a supply chain perspective, reliance on scarce precious metals introduces volatility in raw material costs and potential bottlenecks in sourcing, posing risks to long-term manufacturing stability.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes readily available and inexpensive 2,1-benzisoxazole derivatives coupled with phenylacetaldehyde derivatives to achieve direct construction of the quinoline core. This process employs a cost-effective copper powder and silver trifluoromethanesulfonate catalyst system, which significantly lowers the barrier to entry for large-scale production compared to palladium or rhodium-based systems. The reaction proceeds smoothly under an oxygen atmosphere, utilizing molecular oxygen as a clean oxidant that produces water as the only byproduct, aligning perfectly with green chemistry principles. The mild reaction conditions, typically ranging from 80°C to 110°C, ensure excellent functional group tolerance, allowing for the synthesis of complex derivatives without the need for extensive protective group chemistry. This streamlined approach not only enhances operational safety but also drastically simplifies the downstream processing requirements.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between the copper catalyst and the organic substrates, initiating with the oxidation of zero-valent copper to a monovalent species. This active copper species then inserts into the 2,1-benzisoxazole ring, forming a key organometallic intermediate that activates the substrate for subsequent nucleophilic attack. The coordination of phenylacetaldehyde to this copper-intermediate facilitates a precise alignment of the reacting centers, promoting the cleavage of the weak nitrogen-oxygen bond within the benzisoxazole moiety. This ring-opening event is critical as it generates a reactive imine species that is primed for intramolecular cyclization. The entire cycle is driven by the continuous regeneration of the active catalyst species through oxidation by molecular oxygen, ensuring high turnover numbers and sustained catalytic activity throughout the reaction duration.
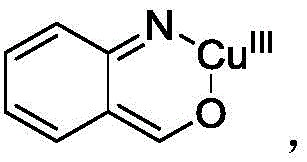
Following the initial activation and ring-opening steps, the reaction proceeds through an isomerization and cyclization sequence to form the dihydroquinoline intermediate. This intermediate subsequently undergoes an oxidative aromatization step, catalyzed by the silver co-catalyst, to yield the final stable quinoline derivative. The presence of the silver salt is crucial for facilitating the final dehydration and aromatization, ensuring that the reaction drives to completion with high selectivity for the aromatic product. Impurity control is inherently managed by the specificity of the copper insertion step, which minimizes non-specific radical reactions that often plague free-radical oxidative processes. This controlled mechanistic pathway results in a clean reaction profile, reducing the burden on purification teams and ensuring that the final product meets the rigorous purity standards demanded by regulatory bodies for pharmaceutical ingredients.
How to Synthesize Quinoline Derivatives Efficiently
To implement this synthesis effectively, one must carefully control the stoichiometry of the reactants and the concentration of the catalyst system to maximize yield and minimize side reactions. The patent outlines a preferred protocol where the molar ratio of 2,1-benzisoxazole to phenylacetaldehyde is maintained between 1:1.5 and 1:2.5 to drive the equilibrium towards product formation. The reaction is typically conducted in hexafluoroisopropanol, a solvent that stabilizes the charged intermediates and enhances the reaction rate, although other polar aprotic solvents can also be employed depending on substrate solubility. Detailed standardized synthesis steps are provided below to guide process chemists in replicating these results reliably in a pilot or production setting.
- Prepare the reaction mixture by dissolving 2,1-benzisoxazole derivatives and phenylacetaldehyde derivatives in hexafluoroisopropanol solvent with copper and silver catalysts.
- Conduct the reaction under an oxygen atmosphere at temperatures between 80°C and 110°C for 18 to 35 hours to facilitate oxidative cyclization.
- Purify the crude product using silica gel column chromatography with a petroleum ether and ethyl acetate mixture to isolate high-purity quinoline derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits regarding cost stability and operational efficiency. The reliance on base metal catalysts like copper, rather than precious metals, insulates the manufacturing process from the volatile price fluctuations often seen in the commodities market for palladium or platinum. Additionally, the use of molecular oxygen as the terminal oxidant eliminates the need for purchasing and storing hazardous chemical oxidants, thereby reducing both material costs and safety compliance overheads. The simplicity of the workup procedure, which involves standard silica gel chromatography, means that existing purification infrastructure can be utilized without significant capital investment in new equipment. These factors combine to create a resilient supply chain model that is both economically advantageous and environmentally sustainable.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and hazardous oxidants leads to substantial cost savings in raw material procurement and waste management. By utilizing abundant copper powder and silver salts in catalytic amounts, the overall cost of goods sold is significantly reduced compared to processes requiring stoichiometric noble metals. Furthermore, the high atom economy of the reaction minimizes waste generation, lowering the expenses associated with environmental compliance and disposal fees. This economic efficiency allows for more competitive pricing strategies in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials, 2,1-benzisoxazole derivatives and phenylacetaldehyde derivatives, are commercially available and can be sourced from multiple suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is critical for maintaining steady supply flows to downstream drug manufacturers. The mild operating parameters also reduce equipment wear and tear, leading to higher uptime and reliability of the production facilities. This stability is essential for meeting tight delivery schedules and maintaining strong relationships with key stakeholders in the pharmaceutical value chain.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common solvents and straightforward reaction setups that can be easily transferred from laboratory to pilot and full-scale production. The green nature of the chemistry, producing water as the primary byproduct, aligns with corporate sustainability goals and helps companies meet increasingly stringent environmental regulations. Reduced solvent usage and simplified purification steps further decrease the environmental footprint of the manufacturing process. This compliance advantage not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible and forward-thinking partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinoline synthesis technology. These answers are derived directly from the patent data and practical considerations for industrial application, providing clarity for decision-makers evaluating this route. Understanding these details is crucial for assessing the feasibility of integrating this method into existing production portfolios.
Q: What are the primary advantages of this quinoline synthesis method over conventional routes?
A: This method utilizes cheap and readily available raw materials like 2,1-benzisoxazole and phenylacetaldehyde, avoiding expensive transition metal catalysts often required in traditional methods. The process operates under mild conditions with oxygen as the oxidant, significantly reducing environmental impact and operational costs associated with waste disposal.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the protocol demonstrates excellent substrate scope and high yields across various substituted derivatives, indicating robust scalability. The use of common solvents and simple purification via column chromatography suggests that the process can be adapted for commercial scale-up with minimal engineering modifications.
Q: How does the catalyst system impact the purity of the final pharmaceutical intermediate?
A: The dual copper and silver catalyst system promotes high selectivity, minimizing the formation of side products and complex impurity profiles. This inherent selectivity simplifies downstream purification, ensuring that the resulting quinoline derivatives meet stringent purity specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our team of expert chemists is well-versed in optimizing complex catalytic cycles like the one described in CN110204486B to meet stringent purity specifications required for clinical and commercial use. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of quinoline derivatives conforms to the highest quality standards. Our commitment to technical excellence and supply chain reliability makes us the ideal partner for your long-term pharmaceutical intermediate needs.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how this advanced synthesis method can benefit your supply chain. By collaborating with us, you gain access to a reliable network of resources and expertise dedicated to accelerating your drug development timeline. Reach out today to discuss how we can support your next breakthrough in pharmaceutical innovation.
