Advanced Cyproconazole Manufacturing: A Novel Synthetic Route for Global Agrochemical Supply Chains
The global agricultural sector continuously demands high-efficiency fungicides to secure crop yields against evolving fungal pathogens, with triazole derivatives like Cyproconazole standing as a cornerstone in modern crop protection strategies. Patent CN101857576A introduces a transformative synthetic methodology that addresses critical bottlenecks in the manufacturing of this vital agrochemical intermediate. This technical insight report analyzes the novel pathway which utilizes p-chlorobenzyl chloride and cyclopropyl methyl ketone as primary starting materials, offering a streamlined alternative to legacy processes. The innovation lies not merely in the chemical transformation but in the strategic reconfiguration of the synthetic tree to enhance safety profiles and operational feasibility. For R&D Directors and Supply Chain Heads, understanding this shift is paramount as it represents a move towards greener chemistry without compromising the structural integrity or biological efficacy of the final active ingredient. The patent details a sequence involving Grignard addition, dehydration, hydroboration-oxidation, and Dess-Martin oxidation, culminating in a Darzens condensation that constructs the final epoxide precursor. This comprehensive approach mitigates the reliance on toxic heavy metals and hazardous alkylating agents that have historically plagued the production of high-purity Cyproconazole, thereby aligning with stringent international environmental regulations and corporate sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cyproconazole has relied heavily on two primary routes, both of which present significant challenges regarding safety, environmental impact, and cost efficiency. The first conventional method, often referred to as the p-chlorobenzaldehyde Grignard reaction method, necessitates the use of Simmons-Smith cyclopropanation to introduce the cyclopropyl ring. This specific step requires a zinc-copper couple and diiodomethane, reagents that are not only expensive but also generate substantial amounts of heavy metal waste that is difficult and costly to treat. Furthermore, subsequent steps in this traditional pathway often involve Swern oxidation and methylation using methyl iodide, a known carcinogen and potent alkylating agent that demands rigorous safety containment and specialized handling protocols. The second conventional route, based on the Friedel-Crafts reaction of p-chlorobenzene with cyclopropyl acetyl chloride, similarly struggles with the use of Lewis acids and the subsequent need for hazardous methylation reagents. These legacy processes create a complex impurity profile that requires extensive purification, driving up production costs and extending lead times for high-purity agrochemical intermediates. The reliance on such toxic reagents poses a continuous liability for manufacturers, complicating regulatory compliance and increasing the risk of supply chain disruptions due to environmental enforcement actions.
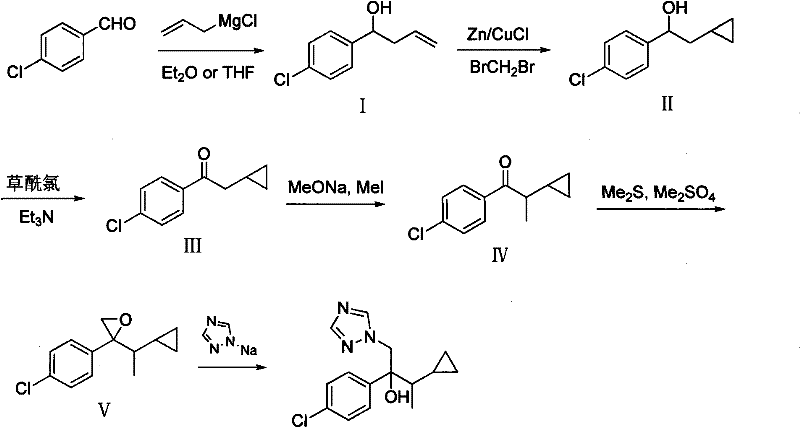
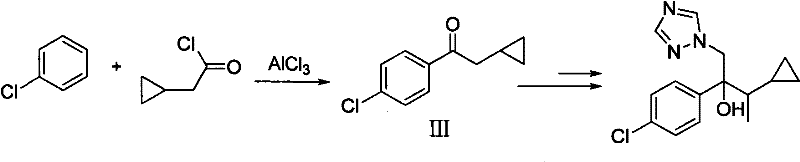
The Novel Approach
In stark contrast to the hazardous legacy pathways, the novel approach detailed in the patent data utilizes a more direct and atom-economical strategy starting from p-chlorobenzyl chloride. This method bypasses the need for Simmons-Smith cyclopropanation entirely by incorporating the cyclopropyl moiety early in the synthesis through the use of cyclopropyl methyl ketone. The initial Grignard reaction between p-chlorobenzyl magnesium chloride and cyclopropyl methyl ketone establishes the core carbon skeleton with high regioselectivity and yield. Subsequent dehydration using phosphorus oxychloride creates a conjugated olefin, which serves as an ideal substrate for hydroboration-oxidation. This sequence allows for the precise introduction of the hydroxyl group at the benzylic position without the need for harsh oxidizing conditions or heavy metal catalysts. The transformation of the resulting alcohol to the corresponding ketone via Dess-Martin periodinane oxidation ensures mild reaction conditions that preserve the integrity of the sensitive cyclopropyl ring. Finally, the Darzens condensation and triazole ring-opening steps are optimized to maximize conversion while minimizing the formation of difficult-to-remove byproducts. This reimagined synthetic route significantly simplifies the purification process, reduces the overall number of unit operations, and eliminates the most dangerous reagents from the manufacturing workflow, offering a robust solution for cost reduction in agrochemical manufacturing.
Mechanistic Insights into Grignard Addition and Hydroboration-Oxidation
The mechanistic elegance of this new synthesis lies in the careful orchestration of nucleophilic additions and oxidative transformations to construct the quaternary carbon center essential for Cyproconazole's biological activity. The initial Grignard reaction involves the nucleophilic attack of the p-chlorobenzyl magnesium species on the carbonyl carbon of cyclopropyl methyl ketone. This step is critical as it forms the tertiary alcohol intermediate, and the reaction conditions must be tightly controlled to prevent side reactions such as enolization or reduction. The presence of the cyclopropyl group adjacent to the carbonyl introduces steric constraints that influence the stereochemical outcome, although the patent indicates a racemic mixture is acceptable for the final fungicidal application. Following the formation of the alcohol, the dehydration step utilizing phosphorus oxychloride proceeds via an E2 elimination mechanism, driven by the stability of the resulting conjugated olefin system. This conjugation is vital for the subsequent hydroboration step, where borane-dimethyl sulfide complex adds across the double bond. The hydroboration occurs with anti-Markovnikov selectivity, placing the boron atom at the less substituted carbon, which upon oxidation with alkaline hydrogen peroxide, yields the primary alcohol at the benzylic position. This specific regiocontrol is a key advantage over other methods that might produce mixtures of isomers, thereby simplifying downstream processing and enhancing the overall purity of the intermediate.
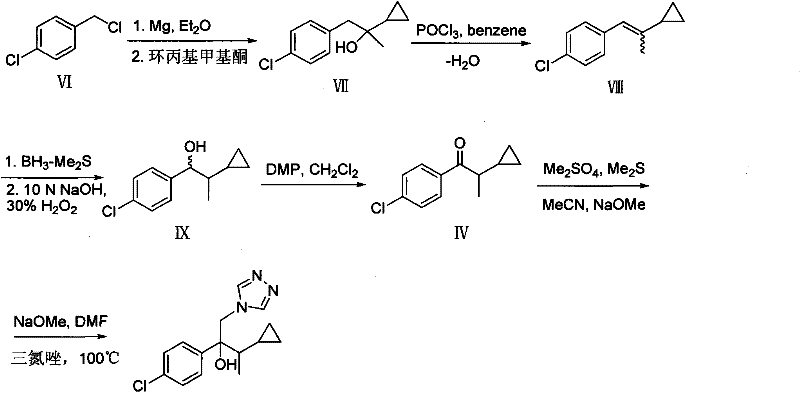
Impurity control in this novel pathway is inherently superior due to the avoidance of radical-based cyclopropanation and harsh methylation steps. In traditional methods, the use of methyl iodide and strong bases often leads to over-alkylation or the formation of elimination byproducts that are structurally similar to the target molecule and difficult to separate. By contrast, the Dess-Martin oxidation used in this new route is highly chemoselective, oxidizing the secondary alcohol to a ketone without affecting the cyclopropyl ring or the chlorophenyl moiety. The subsequent Darzens condensation, which forms the epoxide ring, is conducted under basic conditions using sulfonium ylides generated in situ. While this step does utilize dimethyl sulfate, the overall reduction in hazardous reagents in the earlier stages of the synthesis compensates for this, and the reaction conditions are optimized to minimize the formation of sulfonium salt byproducts. The final ring-opening with 1,2,4-triazole is a nucleophilic substitution that proceeds cleanly, yielding the final Cyproconazole structure with high fidelity. The cumulative effect of these mechanistic choices is a process that generates a cleaner crude product, reducing the burden on purification units and ensuring that the final active ingredient meets the stringent purity specifications required for registration in major agricultural markets.
How to Synthesize Cyproconazole Efficiently
The implementation of this synthetic route requires precise control over reaction parameters to ensure reproducibility and safety on a commercial scale. The process begins with the preparation of the Grignard reagent, where p-chlorobenzyl chloride is reacted with magnesium turnings in anhydrous ether or tetrahydrofunder strict temperature control to prevent runaway exotherms. Once the Grignard reagent is formed, it is added to a solution of cyclopropyl methyl ketone at low temperatures to manage the heat of reaction and ensure high conversion to the tertiary alcohol. The subsequent dehydration and hydroboration steps require careful monitoring of stoichiometry and reaction times to maximize yield while minimizing the formation of oligomeric byproducts. The oxidation steps, particularly the Dess-Martin oxidation, must be quenched properly to remove iodine-containing residues before proceeding to the Darzens condensation. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-efficiency pathway.
- Preparation of Grignard reagent from p-chlorobenzyl chloride and magnesium, followed by nucleophilic substitution with cyclopropyl methyl ketone.
- Dehydration of the resulting alcohol using phosphorus oxychloride to form a conjugated olefin intermediate.
- Hydroboration-oxidation to introduce a hydroxyl group, followed by Dess-Martin oxidation and Darzens condensation to finalize the structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic pathway offers tangible benefits that extend beyond simple chemical yield improvements. The primary advantage lies in the significant reduction of operational risks associated with handling highly toxic reagents. By eliminating the need for heavy metal couples like zinc-copper and avoiding the use of methyl iodide in the early stages of synthesis, manufacturers can drastically lower their expenditure on safety equipment, waste treatment, and regulatory compliance. This shift translates directly into cost reduction in agrochemical intermediate manufacturing, as the overhead associated with hazardous material management is substantially decreased. Furthermore, the simplified workflow reduces the number of isolation and purification steps required, leading to shorter cycle times and increased throughput capacity. This efficiency gain allows suppliers to respond more rapidly to market demand fluctuations, enhancing supply chain reliability and reducing lead time for high-purity agrochemical intermediates. The robustness of the process also means fewer batch failures and less variability in product quality, ensuring a consistent supply of material for formulation partners.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as diiodomethane and methyl iodide removes a significant cost burden from the raw material bill. Additionally, the removal of heavy metal catalysts negates the need for complex and costly wastewater treatment processes designed to remove copper and zinc residues. This qualitative shift in the cost structure allows for substantial cost savings that can be passed down the supply chain or reinvested into capacity expansion. The higher overall yield of the process, driven by fewer side reactions and cleaner transformations, further amplifies these economic benefits by maximizing the output per unit of input material. Consequently, the total cost of ownership for this manufacturing route is significantly lower than that of conventional methods, providing a competitive edge in price-sensitive global markets.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like p-chlorobenzyl chloride and cyclopropyl methyl ketone ensures a stable supply base that is less susceptible to the volatility often seen with specialized organometallic reagents. By simplifying the synthesis tree, the process becomes less fragile, reducing the risk of production stoppages due to reagent shortages or quality issues. This stability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of large-scale agrochemical formulators. The improved safety profile also reduces the likelihood of regulatory interventions or plant shutdowns due to safety incidents, thereby guaranteeing supply continuity. For supply chain heads, this means a more predictable and resilient sourcing strategy that can withstand external pressures and market disruptions.
- Scalability and Environmental Compliance: The novel route is explicitly designed with industrial scale-up in mind, featuring reaction conditions that are easily manageable in large reactors. The avoidance of pyrophoric reagents and highly toxic gases simplifies the engineering requirements for the production facility, making it easier to scale from pilot plant to commercial production volumes. From an environmental perspective, the reduction in heavy metal waste and hazardous organic byproducts aligns with increasingly strict global environmental regulations, such as REACH in Europe. This compliance reduces the risk of fines and facilitates faster regulatory approval for new manufacturing sites. The greener profile of the process also enhances the brand reputation of the manufacturer, appealing to end-users who prioritize sustainability in their supply chain. This combination of scalability and compliance ensures long-term viability and market access for the produced Cyproconazole.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Cyproconazole synthesis route. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data, providing clarity on the process advantages. Understanding these details is essential for technical procurement teams evaluating the feasibility of switching to this newer manufacturing method. The insights provided here cover aspects of safety, yield, and regulatory compliance, which are critical for decision-making.
Q: How does this new synthesis route improve environmental safety compared to traditional methods?
A: The novel route described in patent CN101857576A eliminates the need for heavy metal catalysts such as copper and zinc used in Simmons-Smith cyclopropanation, significantly reducing toxic waste generation and simplifying effluent treatment protocols.
Q: What are the key cost drivers reduced in this manufacturing process?
A: By avoiding expensive and hazardous reagents like methyl iodide and heavy metal couples, the process reduces raw material costs and lowers the operational expenses associated with safety containment and waste disposal.
Q: Is this synthetic pathway suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is designed for industrial production, offering high yields and operational simplicity that facilitate commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyproconazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global agrochemical market. Our CDMO expertise allows us to seamlessly integrate innovations like the pathway described in CN101857576A into our production lines, ensuring that our clients benefit from the latest advancements in chemical manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that the transition from lab to plant is smooth and efficient. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which ensure that every batch of Cyproconazole meets the highest international standards. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient and compliant with the most demanding environmental regulations.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthetic technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate the tangible benefits of our advanced manufacturing capabilities. Let us help you secure a reliable supply of high-quality Cyproconazole that drives your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
