Advanced Synthesis of Axially Chiral Indole Oxide Substituted Styrenes for High-Value Catalysis
Advanced Synthesis of Axially Chiral Indole Oxide Substituted Styrenes for High-Value Catalysis
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral intermediates that serve as the backbone for next-generation pharmaceuticals and advanced functional materials. A significant breakthrough in this domain is detailed in patent CN111848322A, which discloses a novel class of axially chiral indole oxide substituted styrene compounds and, crucially, an efficient resolution method for their production. This technology addresses a critical gap in the market where previous synthetic routes were either non-existent or plagued by low efficiency and narrow substrate scope. By leveraging a sophisticated chiral phosphoric acid catalytic system, this innovation enables the kinetic resolution of racemic precursors to yield structurally complex styrene derivatives with exceptional optical purity. For R&D directors and procurement specialists alike, this represents a paradigm shift towards more sustainable and cost-effective manufacturing of high-value chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has relied heavily on transition metal catalysis or harsh nucleophilic addition reactions, both of which present substantial drawbacks for modern industrial applications. Prior art, such as methods reported in Angew. Chem. Int. Ed. 2016 and J. Org. Chem. 2018, often necessitates the use of expensive palladium or titanium catalysts, which introduce significant challenges regarding heavy metal contamination and subsequent removal processes. Furthermore, these traditional pathways frequently suffer from limited substrate tolerance, meaning that even minor modifications to the aromatic rings can lead to drastic drops in yield or enantioselectivity. The reliance on cryogenic conditions or highly reactive reagents in some legacy methods also escalates operational costs and safety risks, making the commercial scale-up of complex pharmaceutical intermediates economically unviable for many manufacturers seeking reliable supply chain solutions.
The Novel Approach
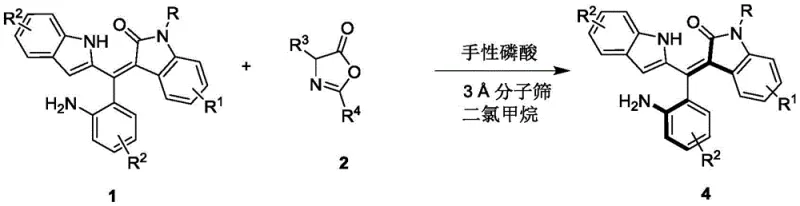
In stark contrast, the methodology outlined in the present patent utilizes an organocatalytic kinetic resolution strategy that operates under remarkably mild and controlled conditions. By employing a specific chiral phosphoric acid catalyst in conjunction with molecular sieves in a dichloromethane solvent system, the process achieves high levels of stereocontrol without the need for toxic transition metals. The reaction proceeds efficiently at temperatures ranging from 0°C to 10°C, significantly reducing energy consumption compared to cryogenic alternatives. This approach not only broadens the scope of compatible substrates, allowing for diverse substitutions on the indole and phenyl rings, but also ensures high atom economy. The result is a robust synthetic route that delivers the target axially chiral compounds with yields often exceeding 40% and enantiomeric excess (ee) values reaching up to 96%, establishing a new benchmark for efficiency in this chemical class.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
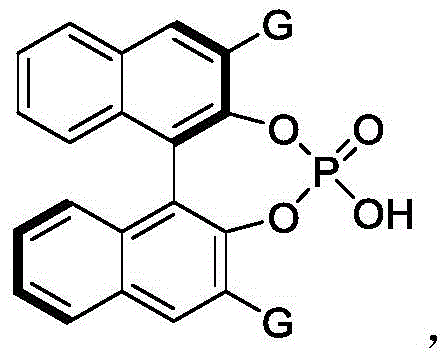
The core of this technological advancement lies in the precise interaction between the chiral phosphoric acid catalyst and the prochiral or racemic substrates. The catalyst, typically a binaphthyl skeleton derivative such as the one shown in Formula 3, acts as a bifunctional activator. Through a network of hydrogen bonding interactions, the phosphoric acid moiety simultaneously activates the electrophilic azlactone and organizes the nucleophilic styrene precursor within a well-defined chiral pocket. This dual activation lowers the activation energy for the desired transformation while imposing strict steric constraints that favor the formation of one enantiomer over the other. The presence of bulky substituents, such as the 2-naphthyl groups on the catalyst backbone, further enhances this discrimination, effectively blocking unfavorable reaction trajectories and ensuring that the resulting product possesses the desired axial chirality with high fidelity.
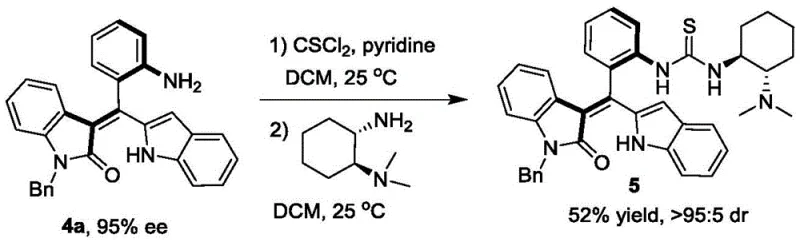
Beyond the primary resolution step, the utility of the resulting axially chiral styrene compounds extends into their application as precursors for even more complex catalytic systems. The patent details a seamless derivatization pathway where the resolved styrene (Formula 4a) is converted into a chiral tertiary amine catalyst (Formula 5). This transformation involves reacting the amino-substituted styrene with thiophosgene followed by coupling with a chiral diamine, preserving the axial chirality established in the initial step. The resulting thiourea-based catalyst demonstrates superior performance in asymmetric [4+2] cyclization reactions, outperforming commercially available counterparts. This downstream versatility underscores the strategic value of the initial resolution step, as it provides access to a library of potent organocatalysts that can drive further synthetic transformations with high stereoselectivity, thereby multiplying the value proposition for end-users in the fine chemical sector.
How to Synthesize Axially Chiral Indole Styrene Efficiently
The practical implementation of this synthesis route is designed for reproducibility and scalability, utilizing standard laboratory equipment and readily available reagents. The process begins with the preparation of a reaction mixture containing the racemic styrene precursor and the azlactone reactant in dry dichloromethane, with activated 3Å molecular sieves added to sequester trace moisture that could deactivate the catalyst. Upon the addition of the chiral phosphoric acid catalyst, the mixture is stirred at a controlled low temperature, typically 0°C, to maximize enantioselectivity. Reaction progress is monitored via thin-layer chromatography (TLC) until the starting material is consumed, after which the mixture is filtered to remove the solid sieves and concentrated under reduced pressure. The crude product is then subjected to silica gel column chromatography using a petroleum ether and dichloromethane eluent system to isolate the pure axially chiral compound. For a comprehensive, step-by-step standardized operating procedure including specific molar ratios and workup details, please refer to the guide below.
- Prepare the reaction mixture by combining the racemic styrene precursor (Formula 1) and the azlactone reactant (Formula 2) in dichloromethane solvent with molecular sieves.
- Add the chiral phosphoric acid catalyst (Formula 3) at a loading of 10 mol% and maintain the reaction temperature strictly between 0°C and 10°C.
- Monitor reaction progress via TLC until completion, then filter off the molecular sieves, concentrate the filtrate, and purify the residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of cost, reliability, and scalability in the supply of high-purity pharmaceutical intermediates. By shifting from metal-dependent catalysis to organocatalysis, manufacturers can eliminate the costly and time-consuming steps associated with heavy metal scavenging and validation, which are critical for meeting stringent regulatory standards in API production. The mild reaction conditions reduce the energy load on production facilities and minimize the risk of thermal runaway incidents, thereby enhancing overall plant safety and operational continuity. Furthermore, the broad substrate scope implies that a single catalytic platform can be adapted to produce a wide variety of analogues, reducing the need for extensive process redevelopment when switching between different product SKUs.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as palladium or titanium results in a direct reduction in raw material costs, while the simplified purification workflow decreases solvent usage and waste disposal expenses. The high enantioselectivity achieved minimizes the loss of material to unwanted isomers, effectively improving the overall mass balance and yield of the valuable chiral intermediate. Additionally, the use of common solvents like dichloromethane and standard purification techniques like silica chromatography ensures that the process can be integrated into existing infrastructure without requiring capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including the substituted indole derivatives and azlactones, are chemically stable and commercially accessible, mitigating the risk of supply bottlenecks often associated with exotic reagents. The robustness of the reaction conditions means that production is less susceptible to variations in ambient temperature or minor fluctuations in reagent quality, leading to more consistent batch-to-batch performance. This reliability is crucial for maintaining just-in-time inventory levels and ensuring that downstream drug development timelines are not compromised by delays in intermediate availability.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic profile being manageable under standard cooling conditions, allowing for safe transition from gram-scale laboratory synthesis to multi-kilogram pilot production. The absence of toxic heavy metals simplifies the environmental compliance landscape, reducing the burden of wastewater treatment and hazardous waste handling. This aligns with global trends towards greener chemistry and sustainability, potentially qualifying the manufacturing process for various eco-friendly certifications that are increasingly valued by major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these axially chiral compounds. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring that the information reflects the actual capabilities and limitations of the technology. Understanding these details is essential for evaluating the feasibility of integrating this synthetic route into your current development pipeline or supply chain strategy.
Q: What is the primary advantage of this resolution method over traditional metal-catalyzed approaches?
A: This organocatalytic method eliminates the need for expensive and toxic transition metals like palladium or titanium, significantly reducing downstream purification costs and environmental impact while achieving enantiomeric excess values up to 96%.
Q: Can the resulting axially chiral styrene compounds be used directly as catalysts?
A: Yes, the resolved compounds (Formula 4) serve as versatile precursors that can be derivatized into highly effective chiral tertiary amine catalysts (Formula 5) for asymmetric [4+2] cyclization reactions.
Q: What are the typical reaction conditions required for industrial scale-up?
A: The process operates under remarkably mild conditions, typically requiring temperatures between 0°C and 25°C and standard atmospheric pressure, which simplifies reactor requirements and enhances safety profiles for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Styrene Supplier
The successful translation of this academic innovation into a commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of axially chiral intermediate meets the exacting standards required for pharmaceutical applications. We understand the critical nature of chirality in drug efficacy and are committed to delivering products with consistent optical purity and minimal impurity profiles.
We invite potential partners to engage with our technical team to explore how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this organocatalytic route. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
