Advanced Organocatalytic Resolution of Axially Chiral Oxindole Styrenes for Commercial Scale-Up
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral building blocks that serve as critical ligands and catalysts in pharmaceutical manufacturing. Patent CN111848322B introduces a groundbreaking methodology for the resolution of axially chiral oxindole-substituted styrene compounds, addressing a significant gap in current organic synthesis capabilities. Traditionally, the construction of axially chiral styrene skeletons has relied heavily on nucleophilic additions to alkynes or palladium-catalyzed coupling reactions, which often suffer from limited substrate scope and harsh reaction conditions. This new approach leverages a chiral phosphoric acid-catalyzed kinetic resolution strategy, utilizing readily available racemic precursors to access structurally diverse and complex chiral architectures with exceptional stereocontrol. The innovation lies not only in the novelty of the compounds themselves but in the robustness of the synthetic route, which operates under mild temperatures and avoids the use of toxic transition metals, aligning perfectly with modern green chemistry principles and regulatory requirements for API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral styrenes has been a formidable challenge for process chemists, primarily due to the reliance on transition metal catalysis which introduces significant downstream processing burdens. Conventional methods, such as those reported in Angew. Chem. Int. Ed. 2016, utilize palladium species to catalyze the coupling of aryl halides with hydrazones, a process that necessitates rigorous purification to remove trace metal contaminants that could poison subsequent catalytic cycles or violate strict pharmaceutical impurity limits. Furthermore, alternative strategies involving L-proline catalyzed nucleophilic additions often require specific substrate activation and may exhibit poor atom economy or limited functional group tolerance. These traditional pathways frequently demand elevated temperatures or specialized equipment, increasing energy consumption and operational complexity. The inability to efficiently resolve racemic mixtures directly has also meant that manufacturers often face a 50% theoretical yield loss unless dynamic kinetic resolution is employed, which adds another layer of catalytic complexity and cost to the manufacturing process.
The Novel Approach
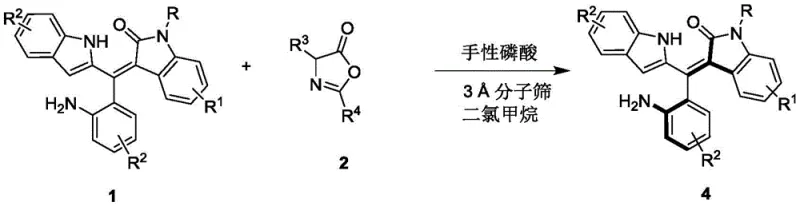
In stark contrast, the methodology disclosed in CN111848322B offers a streamlined, organocatalytic solution that bypasses the need for heavy metals entirely. By employing a specific chiral phosphoric acid catalyst, the process achieves a highly selective kinetic resolution of racemic oxindole-substituted styrenes through an asymmetric addition reaction with azlactones. This reaction proceeds efficiently in dichloromethane at temperatures as low as 0°C to 10°C, significantly reducing energy inputs compared to thermal coupling reactions. The use of 3A molecular sieves as an additive further enhances reaction efficiency by sequestering water, driving the equilibrium towards product formation without the need for exotic reagents. This approach not only simplifies the workup procedure—typically requiring only filtration and standard silica gel chromatography—but also expands the accessible chemical space, allowing for a wide variety of substituents on the indole and phenyl rings without compromising enantioselectivity. The result is a versatile platform technology capable of generating high-value chiral intermediates with yields ranging from 39% to 54% and enantiomeric excesses up to 96%.
Mechanistic Insights into Chiral Phosphoric Acid Catalysis
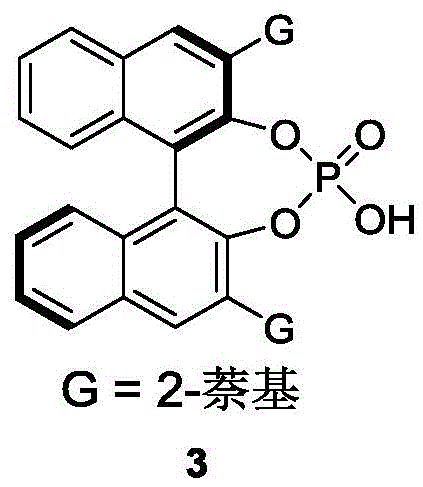
The success of this resolution strategy hinges on the precise three-dimensional architecture of the chiral phosphoric acid catalyst, specifically the BINOL-derived scaffold substituted with bulky 2-naphthyl groups (Formula 3). This catalyst functions as a dual hydrogen-bond donor, simultaneously activating the electrophilic azlactone and organizing the nucleophilic styrene substrate within a well-defined chiral pocket. The steric bulk of the 2-naphthyl groups at the 3,3'-positions of the BINOL backbone creates a confined environment that effectively differentiates between the two enantiomers of the racemic starting material. Through a network of non-covalent interactions, including hydrogen bonding and pi-pi stacking, the catalyst lowers the activation energy for the reaction of one enantiomer while sterically hindering the other, leading to high kinetic resolution. This mechanism ensures that the unreacted starting material and the product possess distinct stereochemical configurations, allowing for the isolation of the desired axially chiral styrene with high optical purity. The robustness of this hydrogen-bonding activation mode allows the reaction to tolerate a broad range of electronic and steric variations on the substrate, making it a powerful tool for library synthesis.
Controlling the impurity profile in such complex molecular assemblies is critical for their subsequent use as ligands or catalysts. The mechanism inherently suppresses the formation of side products by enforcing a rigid transition state geometry. Unlike metal-catalyzed processes where beta-hydride elimination or oxidative addition side reactions can generate difficult-to-remove impurities, this organocatalytic pathway is highly chemoselective. The mild acidic nature of the phosphoric acid prevents the decomposition of sensitive functional groups such as esters or halides, which might otherwise degrade under basic or strongly Lewis acidic conditions. Furthermore, the reaction produces minimal waste; the primary byproduct is the ring-opened form of the azlactone or unreacted starting material, both of which are easily separated via standard chromatographic techniques. This high level of selectivity translates directly to reduced purification costs and higher overall process efficiency, as fewer recrystallization steps are needed to achieve the stringent purity specifications required for fine chemical applications.
How to Synthesize Axially Chiral Oxindole Styrene Efficiently
The practical implementation of this synthesis is designed for scalability and ease of operation, making it highly attractive for industrial adoption. The process begins with the preparation of a reaction mixture containing the racemic styrene precursor and the azlactone reagent in dry dichloromethane, supplemented with activated 3A molecular sieves to maintain anhydrous conditions. The chiral phosphoric acid catalyst is then introduced at a loading of 10 mol%, and the mixture is stirred at a controlled temperature of 0°C. Reaction progress is monitored via thin-layer chromatography (TLC), typically reaching completion within 5 hours. Upon completion, the molecular sieves are removed by simple filtration, and the filtrate is concentrated under reduced pressure. The crude residue is purified using silica gel column chromatography with a petroleum ether and dichloromethane eluent system to afford the target axially chiral compound as a yellow solid. For detailed standardized operating procedures and safety protocols, please refer to the technical guide below.
- Prepare the reaction mixture by combining the racemic styrene precursor (Formula 1) and the azlactone reagent (Formula 2) in dichloromethane solvent with 3A molecular sieves.
- Add the chiral phosphoric acid catalyst (Formula 3, specifically the 2-naphthyl substituted BINOL derivative) at a loading of 10 mol% under inert atmosphere.
- Stir the reaction at 0°C to 10°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography to isolate the target chiral styrene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift towards this organocatalytic resolution method represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with metal-dependent syntheses. By eliminating the need for precious metal catalysts like palladium or titanium, manufacturers can avoid the volatile pricing and supply chain disruptions often associated with these commodities. Moreover, the removal of heavy metals from the process flow drastically simplifies the purification train, reducing the consumption of specialized scavenging resins and solvents required to meet residual metal specifications. This streamlining of the downstream processing directly contributes to significant cost reduction in pharmaceutical intermediate manufacturing, as it shortens cycle times and reduces waste disposal costs associated with metal-contaminated effluents. The mild reaction conditions also imply lower energy consumption for heating or cooling, further enhancing the economic viability of large-scale production runs.
- Cost Reduction in Manufacturing: The transition to a metal-free organocatalytic process fundamentally alters the cost equation by removing the expense of precious metal catalysts and the associated purification steps. Traditional methods often require expensive ligands and rigorous metal scavenging protocols to ensure product safety, which adds substantial operational expenditure. In contrast, the chiral phosphoric acid catalyst used in this method is robust and can be sourced efficiently, while the absence of metals means that standard silica gel chromatography is sufficient for purification. This simplification reduces solvent usage and waste generation, leading to substantial cost savings in raw materials and environmental compliance. Additionally, the high atom economy of the addition reaction ensures that a greater proportion of the input mass is converted into valuable product, minimizing material loss.
- Enhanced Supply Chain Reliability: Relying on transition metals introduces a layer of supply chain vulnerability, as the availability of elements like palladium can be subject to geopolitical instability and mining constraints. By adopting a synthetic route based on abundant organic elements (carbon, hydrogen, nitrogen, oxygen, phosphorus), the supply chain becomes more resilient and less susceptible to external shocks. The reagents required for this process, such as the azlactones and the BINOL-derived catalysts, are synthesized from commodity chemicals with stable global supply networks. This stability ensures consistent lead times for high-purity intermediates, allowing procurement teams to plan inventory more effectively and reduce the need for safety stock. Furthermore, the broad substrate scope of the reaction means that a single catalytic platform can produce a diverse array of derivatives, reducing the need to qualify multiple distinct synthetic routes for different analogs.
- Scalability and Environmental Compliance: The mild conditions and simple workup procedures of this resolution method make it inherently scalable from gram to multi-kilogram quantities without significant re-optimization. The use of dichloromethane, while requiring careful handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols. More importantly, the absence of heavy metal waste simplifies environmental compliance, as the effluent does not require specialized treatment for metal removal before discharge. This aligns with increasingly stringent global environmental regulations and corporate sustainability goals. The process generates less hazardous waste overall, reducing the carbon footprint of the manufacturing operation. The ability to scale this process efficiently ensures that supply can meet demand spikes without the long lead times typically associated with scaling complex metal-catalyzed reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these axially chiral compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production pipelines or R&D programs. We encourage technical teams to review these insights to fully appreciate the versatility and robustness of this synthetic platform.
Q: What are the advantages of using chiral phosphoric acid over metal catalysts for this resolution?
A: The use of chiral phosphoric acid eliminates the need for expensive transition metals like palladium or titanium, thereby removing the costly and complex heavy metal removal steps required for pharmaceutical compliance. Additionally, the reaction proceeds under significantly milder conditions (0°C) compared to traditional coupling methods.
Q: What is the typical enantiomeric excess (ee) achieved with this method?
A: The patented method consistently achieves high enantioselectivity, with reported ee values reaching up to 96% for various substrates, ensuring the production of high-purity chiral intermediates suitable for sensitive catalytic applications.
Q: Can the resolved styrene compounds be further functionalized?
A: Yes, the resolved axially chiral styrenes serve as versatile precursors. They can be derivatized into chiral tertiary amine catalysts (Formula 5), which have demonstrated superior performance in asymmetric [4+2] cyclization reactions compared to commercial alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Styrene Supplier
The development of efficient routes to axially chiral oxindole-substituted styrenes marks a significant advancement in the field of asymmetric catalysis, offering new possibilities for the synthesis of complex bioactive molecules. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring these cutting-edge intermediates to the market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch meets the exacting standards required for pharmaceutical and fine chemical applications. We understand the critical nature of chiral purity in downstream synthesis and are committed to delivering materials with consistent optical rotation and enantiomeric excess values.
We invite you to collaborate with us to explore how this technology can enhance your product portfolio and streamline your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data for our available batches or to discuss route feasibility assessments for your target molecules. Together, we can accelerate the development of next-generation chiral catalysts and intermediates, driving efficiency and innovation in the global chemical industry.
