Advanced Synthesis of 3-Alkyl Four-Membered Cycloenones for Commercial Scale-Up
The landscape of organic synthesis for strained ring systems is constantly evolving, driven by the need for more efficient and scalable pathways to complex intermediates. Patent CN102146028A introduces a groundbreaking methodology for the synthesis of various substituted 3-alkyl four-membered cycloenone compounds, a structural motif frequently encountered in the development of bioactive molecules and natural product analogs. This technology leverages a direct reaction between 2,3-allenoate esters and dialkylzinc reagents, bypassing the traditional reliance on unstable precursors. For R&D directors and procurement specialists in the fine chemical sector, this represents a significant opportunity to streamline the production of high-purity pharmaceutical intermediates. The method operates under relatively mild thermal conditions in common solvents like toluene or xylene, offering a robust alternative to legacy synthetic routes that often suffer from poor regioselectivity and difficult purification protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of four-membered cycloenone rings has been a formidable challenge in synthetic organic chemistry, primarily due to the inherent ring strain and the reactivity of the conjugated enone system. Conventional strategies predominantly rely on [2+2] cycloaddition reactions involving ketenes and enamine salts. However, these traditional precursors are notoriously unstable and difficult to handle on a large industrial scale, often requiring stringent anhydrous conditions and cryogenic temperatures to prevent decomposition. Furthermore, controlling the regioselectivity in these [2+2] reactions is problematic, frequently leading to complex mixtures of isomers that are arduous to separate. This lack of selectivity not only diminishes the overall yield but also drastically increases the cost of goods sold due to the extensive chromatographic purification required. For supply chain managers, the reliance on such finicky precursors introduces significant risk regarding batch-to-batch consistency and lead time reliability.
The Novel Approach
In stark contrast to the limitations of legacy methods, the technology disclosed in CN102146028A utilizes a highly efficient one-step protocol involving 2,3-allenoate esters and dialkylzinc. This novel approach eliminates the need for pre-forming unstable ketene intermediates, thereby simplifying the operational workflow significantly. The reaction proceeds through a concerted sequence of 1,4-addition, cyclization, 1,2-addition, and elimination, directly furnishing the desired 3-alkyl four-membered cycloenone skeleton. By employing readily available dialkylzinc reagents, the method ensures excellent regioselectivity, specifically introducing alkyl groups at the 3-position of the ring while maintaining the integrity of substituents at the 2 and 4 positions. This precision in molecular construction translates to cleaner reaction profiles, reduced solvent consumption during workup, and a substantial reduction in the environmental footprint associated with chemical manufacturing.
Mechanistic Insights into Dialkylzinc-Mediated Cyclization
The core of this synthetic breakthrough lies in the unique reactivity of the dialkylzinc species towards the allenoate ester system. The mechanism initiates with a 1,4-addition of the organozinc reagent to the conjugated allenoate, generating a zinc enolate intermediate. This intermediate subsequently undergoes an intramolecular cyclization, closing the four-membered ring structure. The process is completed by a 1,2-addition and elimination sequence that restores the carbonyl functionality and establishes the double bond within the ring. This cascade reaction is highly sensitive to the electronic nature of the substituents, yet the patent data demonstrates remarkable tolerance for various alkyl and aryl groups. The ability to introduce three different substituents at the 2, 3, and 4 positions of the cycloenone ring in a single operation provides medicinal chemists with unparalleled flexibility for structure-activity relationship (SAR) studies.
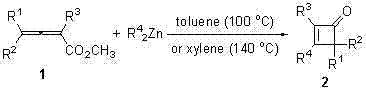
Furthermore, the impurity profile of this reaction is significantly improved compared to traditional methods. Because the reaction does not require transition metal catalysts that often leave toxic residues, the downstream purification process is simplified to basic aqueous workups and flash column chromatography. The absence of heavy metal contaminants is a critical advantage for pharmaceutical intermediate manufacturing, where strict regulatory limits on residual metals must be met. The high regioselectivity ensures that side products resulting from alternative addition pathways are minimized, leading to crude reaction mixtures that are easier to purify. This mechanistic efficiency not only enhances the chemical yield, which ranges from 50% to 92% across various substrates, but also ensures that the final product meets the stringent purity specifications required for downstream drug synthesis.
How to Synthesize 3-Alkyl Four-Membered Cycloenones Efficiently
Implementing this synthesis route requires careful attention to reaction conditions to maximize yield and safety. The process begins by charging a reaction vessel with the 2,3-allenoate ester substrate and an aromatic solvent such as toluene or xylene under an inert nitrogen atmosphere. The dialkylzinc reagent, typically used in a molar excess to drive the reaction to completion, is then added dropwise to the stirred mixture. The reaction temperature is carefully controlled, usually maintained between 100°C and 140°C depending on the specific solvent and substrate reactivity. Following the reaction period, the mixture is cooled and quenched with a saturated ammonium chloride solution to decompose any remaining organozinc species. The detailed standardized synthesis steps are provided in the guide below.
- Under nitrogen protection, combine 2,3-allenoate ester substrate with toluene or xylene solvent in a reaction vessel.
- Dropwise add dialkylzinc reagent to the mixture under stirring, maintaining a molar ratio of approximately 3: 1.
- Heat the reaction to 100°C or 140°C, then quench with saturated ammonium chloride and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers profound benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for complex intermediates. The elimination of unstable precursors and transition metal catalysts directly correlates to a reduction in raw material costs and waste disposal expenses. By simplifying the synthetic sequence to a one-step process, manufacturers can significantly reduce the operational time and labor required per batch. This efficiency gain allows for a more responsive supply chain capable of meeting tight production schedules without compromising on quality. Additionally, the use of common solvents like toluene and xylene ensures that the process is compatible with existing standard chemical infrastructure, avoiding the need for specialized equipment investments.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the avoidance of complex multi-step precursor synthesis lead to substantial cost savings. The high atom economy of the dialkylzinc addition means that a larger proportion of the starting materials are incorporated into the final product, minimizing waste. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents during chromatography, which are often significant cost drivers in fine chemical production. These factors combined result in a more economically viable production model for high-value intermediates.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as 2,3-allenoate esters and dialkylzinc, mitigates the risk of supply disruptions associated with custom-synthesized precursors. The robustness of the reaction conditions, which do not require extreme cryogenic temperatures, ensures consistent batch-to-batch performance. This reliability is crucial for maintaining continuous production lines and meeting the just-in-time delivery requirements of global pharmaceutical clients. The predictable nature of the chemistry allows for more accurate forecasting and inventory management.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful synthesis of various derivatives with consistent yields. The absence of heavy metal catalysts simplifies the environmental compliance landscape, reducing the burden of wastewater treatment and hazardous waste disposal. The reaction generates fewer byproducts, aligning with green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations. This scalability ensures that the technology can be seamlessly transitioned from laboratory discovery to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The clarity provided here aims to facilitate informed decision-making regarding process adoption and supplier selection.
Q: What are the advantages of this dialkylzinc method over traditional [2+2] cycloaddition?
A: Traditional methods often rely on unstable ketene or enamine salt precursors which are difficult to control regioselectively. This patent describes a direct reaction using stable allenoate esters and dialkylzinc, eliminating the need for unstable precursors and significantly improving regioselectivity and yield.
Q: What solvents and temperatures are required for this synthesis?
A: The process utilizes common aromatic solvents such as toluene or xylene. The reaction temperatures are moderate, typically ranging between 100°C and 140°C, which facilitates easier thermal management during commercial scale-up compared to cryogenic conditions.
Q: What is the expected yield range for these cycloenone compounds?
A: According to the experimental data in patent CN102146028A, the reaction yields range from 50% to 92% depending on the specific substituents. This high efficiency reduces raw material waste and downstream purification costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkyl Four-Membered Cycloenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation pharmaceuticals. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN102146028A can be effectively translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest quality standards. Our commitment to technical excellence allows us to navigate the complexities of strained ring synthesis with precision and reliability.
We invite you to collaborate with us to optimize your supply chain for these valuable intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can support your R&D and commercial goals. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technically superior and compliant with global regulatory standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
