Advanced Base-Mediated Cyclization for High-Purity Quinolizinone Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN111116576A introduces a groundbreaking methodology for the preparation of quinolizinone compounds, a class of scaffolds with demonstrated potential in treating Alzheimer's disease, type II diabetes, and immunoglobulin E-related disorders. This innovation addresses critical bottlenecks in existing synthetic strategies by utilizing a direct cyclization approach between alkynyl ester propiolates and pyridine derivatives. Unlike traditional multi-step sequences that often require harsh conditions and expensive catalysts, this novel protocol operates under remarkably mild thermal parameters, typically between 60°C and 80°C. The strategic design of this reaction pathway not only simplifies the operational workflow but also significantly enhances the overall atom economy and process safety profile. By leveraging readily available starting materials and common inorganic bases, this technology represents a paradigm shift towards more sustainable and cost-efficient manufacturing of complex pharmaceutical intermediates.
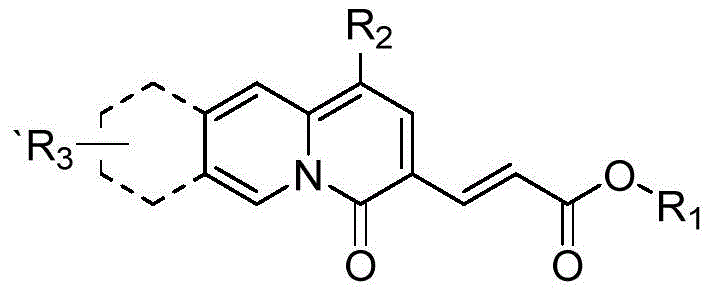
Historically, the construction of the quinolizinone core has been fraught with synthetic challenges that impede large-scale production. Conventional methods, such as the condensation of ethyl 2-pyridine acetate with diethyl ethoxymethylene malonate, often necessitate rigorous pre-activation of substrates and involve multiple isolation steps that accumulate waste and reduce overall throughput. Furthermore, alternative approaches like the Horner-Wadsworth-Emmons olefination frequently demand specialized reagents and strict anhydrous conditions, driving up equipment costs and limiting scalability. These legacy processes are characterized by long production cycles and energy-intensive heating requirements, which collectively inflate the cost of goods sold (COGS) for the final active pharmaceutical ingredient. In contrast, the novel approach detailed in the patent data circumvents these limitations by employing a one-pot cyclization strategy. This method utilizes a simple base-mediated mechanism that proceeds efficiently in common solvents like ethyl acetate, thereby eliminating the need for exotic catalysts or complex protection-deprotection sequences.
The mechanistic elegance of this transformation lies in the base-promoted nucleophilic attack of the pyridine derivative on the electron-deficient alkyne of the propiolate. The reaction initiates with the deprotonation of the active methylene group adjacent to the nitrile or ester functionality, generating a nucleophilic species that attacks the triple bond. This is followed by an intramolecular cyclization that constructs the fused bicyclic quinolizinone system with high regioselectivity. The use of mild bases such as potassium carbonate or cesium carbonate ensures that sensitive functional groups on the substrate remain intact, minimizing the formation of degradation byproducts. For instance, in the optimization of specific embodiments, the reaction consistently delivers yields exceeding 80% when conducted at 80°C for 3 hours. This high level of conversion is attributed to the favorable kinetics of the cyclization step under these specific thermal conditions, which strike an optimal balance between reaction rate and thermal stability of the intermediates.
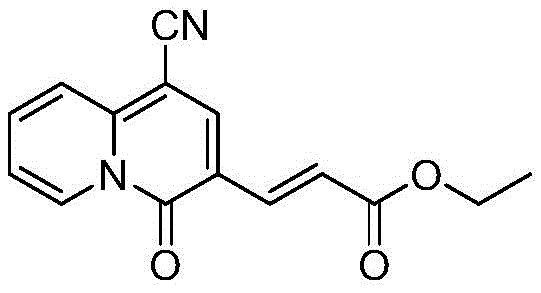
To facilitate the practical adoption of this technology, the patent outlines a streamlined operational procedure that is amenable to both laboratory and pilot-scale execution. The synthesis begins with the precise mixing of alkynyl ester propiolate compounds and pyridine derivatives in a defined molar ratio, typically favoring an excess of the alkyne component to drive the equilibrium forward. Following the addition of the solvent and the base, the mixture is subjected to controlled heating, after which the workup involves simple aqueous washing and distillation. This simplicity reduces the burden on technical teams regarding process validation and operator training. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the specific reagent quantities and purification parameters required to replicate these high-yielding results consistently across different batches.
For procurement and supply chain executives, the implications of adopting this synthetic route are profound, offering tangible advantages in cost structure and logistical reliability. The elimination of transition metal catalysts, which are often subject to volatile market pricing and stringent residual limits in final drug products, translates directly into substantial cost savings in manufacturing. Additionally, the reliance on commodity chemicals like ethyl acetate and potassium hydroxide ensures a stable supply chain that is less susceptible to geopolitical disruptions compared to specialized reagents. The mild reaction temperatures also imply lower energy consumption and reduced wear on reactor vessels, contributing to a lower total cost of ownership for production facilities. From an environmental compliance perspective, the simplified workup procedure generates less hazardous waste, aligning with increasingly strict global regulations on chemical discharge and sustainability. This process enables the commercial scale-up of complex pharmaceutical intermediates with a significantly reduced environmental footprint.
The robustness of this methodology further enhances supply chain reliability by shortening the overall production lead time. Traditional multi-step syntheses often span several days due to intermediate isolations and drying steps, whereas this one-pot cyclization can be completed within a single shift of 3 to 5 hours. This acceleration allows manufacturers to respond more agilely to fluctuating market demands and reduces the inventory holding costs associated with work-in-progress materials. Moreover, the high purity achieved through standard column chromatography minimizes the need for extensive recrystallization or additional purification stages, streamlining the path from raw material to finished intermediate. These factors collectively contribute to a more resilient and responsive supply network, ensuring that critical pharmaceutical ingredients are available when needed without compromising on quality standards or regulatory compliance requirements.
- Mix alkynyl ester propiolate compounds with pyridine derivatives in a molar ratio of approximately 3: 1 to form the initial reaction mixture.
- Add ethyl acetate as a solvent and introduce an inorganic or organic base such as potassium hydroxide or cesium carbonate, then heat the mixture to 60°C–80°C for 3 to 5 hours.
- Cool the reaction to room temperature, remove solvents via reduced pressure distillation, wash with water to remove inorganic salts, and purify the final product using column chromatography.
Frequently Asked Questions (FAQ)
Q: What are the primary advantages of this quinolizinone synthesis method?
A: This method offers significant advantages including mild reaction conditions (60°C–80°C), short reaction times (3–5 hours), and high yields ranging from 70% to 95%. It eliminates the need for complex pre-activation steps required in conventional methods.
Q: What types of substituents are compatible with this cyclization process?
A: The process is highly versatile, accommodating various R1 groups such as methyl, ethyl, tert-butyl, and allyl, as well as electron-withdrawing R2 groups like cyano and ester functionalities, allowing for diverse structural modifications.
Q: How is product purity ensured in this manufacturing process?
A: High purity is achieved through a straightforward workup procedure involving water washing to remove inorganic salts followed by column chromatography purification, ensuring the removal of side products and unreacted starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolizinone Supplier
As the global demand for advanced therapeutic agents continues to rise, the ability to produce high-quality intermediates efficiently becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in organic synthesis to bring innovative patents like CN111116576A to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and risk-mitigated. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinolizinone intermediate meets the exacting standards required by top-tier pharmaceutical companies worldwide. Our commitment to technical excellence ensures that complex chemical architectures are delivered with consistency and reliability.
We invite potential partners to engage with our technical procurement team to explore how this advanced synthesis technology can optimize your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic benefits of switching to this more efficient manufacturing route. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project requirements. Together, we can accelerate the development of life-saving medications by ensuring a stable, cost-effective, and high-quality supply of essential chemical building blocks for the next generation of therapies.
