Advanced Copper-Catalyzed Synthesis of Tetra-Substituted Furans for Commercial Pharmaceutical Applications
Advanced Copper-Catalyzed Synthesis of Tetra-Substituted Furans for Commercial Pharmaceutical Applications
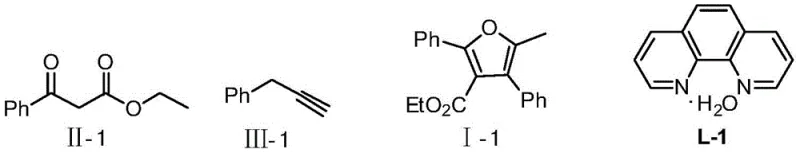
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for more efficient and cost-effective synthetic routes for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in Chinese Patent CN108976186B, which discloses a novel method for preparing tetra-substituted furan compounds based on carbon-hydrogen bond activation. This technology represents a paradigm shift from traditional noble metal catalysis to a more sustainable and economically viable copper-catalyzed system. By leveraging a [3+2] cycloaddition strategy between beta-keto esters and propargyl compounds, this invention enables the construction of complex furan scaffolds with exceptional efficiency. For R&D directors and procurement specialists in the global pharmaceutical sector, understanding the nuances of this patent is critical, as it offers a pathway to reduce manufacturing costs while ensuring high purity and structural diversity in key intermediates.

The core innovation lies in the specific catalytic system employed, which utilizes copper salts complexed with 1,10-phenanthroline ligands. This combination facilitates the activation of inert C-H bonds under relatively mild conditions, eliminating the need for harsh reagents often associated with older methodologies. The versatility of this approach is evident in its ability to tolerate a wide range of functional groups, allowing for the synthesis of diverse derivatives that are essential for drug discovery pipelines. As a reliable pharmaceutical intermediate supplier, recognizing the potential of such C-H activation technologies allows us to offer clients superior alternatives to legacy processes, ensuring that supply chains remain robust and cost-competitive in an increasingly regulated market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted furans has relied heavily on cyclization reactions involving non-cyclic precursors or the functionalization of existing furan rings. Traditional methods frequently necessitate the use of precious metal catalysts such as palladium, gold, or rhodium, which impose a substantial financial burden on large-scale manufacturing operations. Beyond the exorbitant cost of the catalysts themselves, these noble metal systems often require stringent reaction conditions, including high pressures or extreme temperatures, which complicate process safety and engineering controls. Furthermore, the removal of trace heavy metal residues from the final product to meet pharmacopeial standards adds additional downstream processing steps, such as specialized scavenging or recrystallization, thereby extending production lead times and increasing waste generation. These factors collectively hinder the economic feasibility of producing complex furan derivatives for commercial applications, creating a bottleneck for the cost reduction in API manufacturing that procurement teams desperately seek.
The Novel Approach
In stark contrast, the method described in CN108976186B introduces a streamlined copper-catalyzed protocol that addresses these inefficiencies head-on. By utilizing abundant and inexpensive copper salts alongside readily available 1,10-phenanthroline ligands, the process drastically reduces the raw material expenditure associated with catalysis. The reaction proceeds through an oxidative [3+2] cycloaddition mechanism that is highly atom-economical, minimizing the formation of unwanted byproducts and simplifying the purification workflow. The operational simplicity is further enhanced by the use of common organic solvents like ethanol or dichloromethane, which are easy to handle and recover on an industrial scale. This novel approach not only achieves high yields, reported up to 94% in specific embodiments, but also offers a broader substrate scope, enabling the incorporation of diverse substituents without compromising reaction efficiency. For supply chain heads, this translates to a more resilient production model with reduced dependency on volatile precious metal markets.
Mechanistic Insights into Copper-Catalyzed C-H Activation and Cyclization
From a mechanistic perspective, the success of this transformation hinges on the precise interplay between the copper center and the phenanthroline ligand, which modulates the electronic properties of the catalyst to facilitate C-H bond cleavage. The proposed catalytic cycle likely involves the initial coordination of the copper species to the alkyne moiety of the propargyl compound, followed by nucleophilic attack from the enol form of the beta-keto ester. This step is critical for forming the initial carbon-carbon bond that sets the stage for ring closure. Subsequent oxidative cyclization, driven by the presence of an oxidant such as silver carbonate, promotes the formation of the furan ring while regenerating the active copper species. The choice of ligand is paramount; electron-rich or sterically bulky phenanthroline derivatives can fine-tune the reactivity to prevent side reactions such as polymerization of the alkyne or over-oxidation of the sensitive furan core. Understanding these mechanistic details allows chemists to optimize reaction parameters, ensuring consistent quality and reproducibility across different batches of high-purity OLED material or pharmaceutical intermediates.
Impurity control is another vital aspect where this mechanistic understanding pays dividends. The specific selection of the base additive, such as DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene), plays a dual role in neutralizing acidic byproducts and promoting the final elimination step to aromatize the furan ring. By carefully controlling the stoichiometry of the oxidant and the base, the formation of regioisomers or partially cyclized intermediates can be suppressed. This level of control is essential for meeting the rigorous purity specifications required in the fine chemical industry. Moreover, the reaction's tolerance to various functional groups means that protecting group strategies can often be minimized, further streamlining the synthetic route. For R&D teams focused on process development, this mechanistic clarity provides a solid foundation for scaling up the commercial scale-up of complex heterocycles without encountering unexpected purification challenges.
How to Synthesize Tetra-Substituted Furan Efficiently
The practical implementation of this copper-catalyzed methodology is designed to be accessible for both laboratory-scale optimization and pilot-plant production. The general procedure involves the in situ generation of the active copper catalyst, followed by the sequential addition of substrates and oxidants under an inert atmosphere. This modular approach allows for flexibility in adjusting reaction times and temperatures to suit specific substrate combinations. While the patent provides detailed experimental conditions, the key to success lies in maintaining strict exclusion of moisture and oxygen during the catalyst preparation phase to ensure maximum activity. The following guide outlines the standardized operational framework derived from the patent data, serving as a blueprint for technical teams aiming to replicate these results.
- Prepare the copper catalyst in situ by stirring copper salt and 1,10-diazaphenanthrene ligand in a reaction medium under nitrogen protection for 0.5 to 2 hours.
- Dissolve the beta-keto acid ester, propargyl compound, and oxidant in the reaction medium, then add this mixture to the catalyst solution under nitrogen.
- Reflux the mixture for 1-24 hours, add a base additive like DBU, continue refluxing for 0.5-8 hours, and finally isolate the product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this copper-catalyzed technology offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage is the dramatic shift in cost structure achieved by replacing scarce noble metals with earth-abundant copper. This substitution eliminates the price volatility associated with palladium or rhodium markets, allowing for more accurate long-term budgeting and contract negotiations. Additionally, the simplified workup procedure, which often requires only concentration and column chromatography, reduces the consumption of solvents and silica gel, contributing to substantial cost savings in waste disposal and material handling. These efficiencies directly impact the bottom line, making the production of high-value furan intermediates more economically attractive for generic drug manufacturers and specialty chemical producers alike.
- Cost Reduction in Manufacturing: The transition to a copper-based catalytic system fundamentally alters the cost equation for producing tetra-substituted furans. By removing the need for expensive ligands and precious metal precursors, the direct material cost per kilogram of product is significantly lowered. Furthermore, the high atom economy of the [3+2] cycloaddition minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into valuable product. This efficiency is compounded by the ability to use technical-grade solvents like ethanol, which are far cheaper than the anhydrous, high-purity solvents often required for sensitive organometallic reactions. Consequently, the overall cost of goods sold (COGS) is optimized, providing a competitive edge in pricing negotiations with downstream pharmaceutical clients.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the widespread availability of the key reagents used in this process. Copper salts and phenanthroline ligands are commodity chemicals produced by numerous global suppliers, reducing the risk of single-source bottlenecks. Unlike proprietary catalysts that may have long lead times or restricted distribution, the components of this system can be sourced rapidly to meet surging demand. This accessibility ensures continuity of supply, a critical factor for manufacturers committed to just-in-time delivery models. Moreover, the robustness of the reaction conditions means that production schedules are less likely to be disrupted by minor fluctuations in environmental parameters, further stabilizing the supply chain for critical API intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is facilitated by the use of standard reactor equipment and ambient pressure conditions. The absence of hazardous gases or extreme thermal requirements simplifies the engineering controls needed for large-scale operation, accelerating the timeline from lab bench to commercial plant. From an environmental perspective, the reduced reliance on toxic heavy metals aligns with green chemistry principles and increasingly stringent regulatory standards regarding metal residues in drug substances. This compliance reduces the regulatory burden on quality assurance teams and minimizes the environmental footprint of the manufacturing facility, enhancing the corporate sustainability profile of the producer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for stakeholders evaluating the technology for potential licensing or production partnerships. Understanding these specifics is crucial for making informed decisions about process integration and resource allocation.
Q: What are the primary advantages of this copper-catalyzed method over traditional noble metal routes?
A: This method replaces expensive noble metals like palladium or gold with inexpensive copper salts, significantly lowering raw material costs while maintaining high yields up to 94%.
Q: Is this synthesis suitable for large-scale commercial production of API intermediates?
A: Yes, the process utilizes common solvents like ethanol and operates under atmospheric pressure with simple workup procedures, making it highly scalable for industrial manufacturing.
Q: What is the substrate scope for the propargyl and beta-keto ester components?
A: The reaction demonstrates broad substrate tolerance, accommodating various substituted phenyl groups, alkyl chains, and heterocycles on both the beta-keto ester and propargyl starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetra-Substituted Furan Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in modernizing the production of heterocyclic building blocks. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN108976186B are fully realized in practical manufacturing settings. We are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of tetra-substituted furan meets the exacting standards required for pharmaceutical applications. Our commitment to quality and efficiency makes us a trusted partner for companies seeking to optimize their supply chains.
We invite you to explore how our capabilities can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current sourcing strategy or detailed technical data to validate a new route, our technical procurement team is ready to assist. Please contact us to request specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to drive innovation and efficiency in your chemical supply chain.
