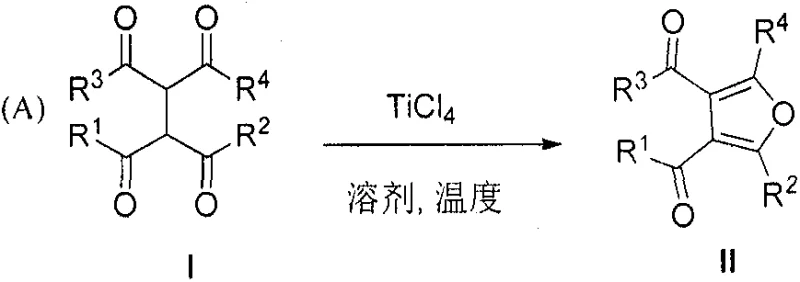
Advanced TiCl4-Catalyzed Paal-Knorr Cyclization for Scalable Tetra-Substituted Furan Production
Advanced TiCl4-Catalyzed Paal-Knorr Cyclization for Scalable Tetra-Substituted Furan Production
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for high-purity intermediates in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in patent CN107311963B, which details a novel and highly efficient method for synthesizing tetra-substituted furan compounds. This technology addresses long-standing challenges in the Paal-Knorr furan synthesis by utilizing titanium tetrachloride (TiCl4) as a dual-function Lewis acid and dehydrating agent. Unlike traditional methods that often rely on harsh Bronsted acids or energy-intensive microwave assistance, this approach offers a streamlined, one-pot solution that operates under remarkably mild conditions. For R&D directors and procurement specialists seeking a reliable tetra-substituted furan supplier, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing protocols. The ability to convert readily available tetracarbonyl precursors directly into complex furan scaffolds with high regioselectivity opens new avenues for the rapid assembly of bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted furans has been plagued by significant operational and chemical inefficiencies that hinder large-scale adoption. Traditional Paal-Knorr protocols frequently employ strong Bronsted acids such as concentrated sulfuric acid, hydrochloric acid, or p-toluenesulfonic acid, which necessitate rigorous corrosion-resistant equipment and generate substantial acidic waste streams that are costly to treat. Furthermore, alternative methods utilizing transition metal catalysts or ionic liquids often suffer from limited substrate scope, failing to accommodate sterically demanding groups without significant loss in yield. Techniques involving microwave or ultrasonic assistance, while effective on a milligram scale in academic settings, present formidable engineering barriers when attempting commercial scale-up of complex pharmaceutical intermediates due to issues with heat transfer uniformity and reactor penetration depth. These legacy methods often result in complex impurity profiles, requiring extensive and yield-eroding purification steps that drive up the final cost of goods.
The Novel Approach
In stark contrast, the methodology disclosed in CN107311963B leverages the unique oxophilicity of titanium tetrachloride to drive the cyclization forward with exceptional efficiency. By employing TiCl4 in common organic solvents like toluene or dichloromethane, the reaction proceeds smoothly at temperatures ranging from 20°C to 80°C, eliminating the need for extreme thermal inputs. This novel approach not only expands the substrate scope to include bulky aryl and alkyl groups but also ensures a cleaner reaction profile with fewer side products. The simplicity of the workup—merely requiring an aqueous ammonium chloride quench followed by standard extraction—drastically reduces the operational complexity typically associated with heterocycle synthesis. For supply chain managers, this translates to a robust process that minimizes downtime and maximizes throughput, effectively solving the bottleneck of cost reduction in fine chemical manufacturing while maintaining stringent quality standards required for downstream drug synthesis.
Mechanistic Insights into TiCl4-Catalyzed Paal-Knorr Cyclization
The efficacy of this synthesis lies in the powerful Lewis acidic nature of titanium tetrachloride, which interacts strongly with the carbonyl oxygen atoms of the tetracarbonyl precursor. Upon coordination, the electron density of the carbonyl group is significantly depleted, thereby enhancing the acidity of the adjacent alpha-protons and facilitating rapid enolization. This activated enol species then undergoes a nucleophilic attack on the neighboring carbonyl carbon, a process that is kinetically accelerated by the titanium center. Crucially, TiCl4 also acts as a potent dehydrating agent, sequestering the water molecule generated during the ring closure to form stable titanium-oxygen complexes. This dual role effectively pulls the reaction equilibrium towards the formation of the furan ring, preventing the reverse hydrolysis reaction that often plagues acid-catalyzed condensations in moist environments. The result is a highly driven thermodynamic pathway that ensures high conversion rates even for substrates that are traditionally unreactive.
From an impurity control perspective, the mechanism offers distinct advantages over Bronsted acid catalysis. Strong protic acids often promote polymerization or degradation of sensitive functional groups, leading to dark, tarry byproducts that are difficult to separate. However, the Lewis acid mechanism mediated by TiCl4 is more selective, primarily targeting the specific 1,4-dicarbonyl motifs required for furan formation. The mild reaction conditions further suppress thermal decomposition pathways, resulting in a crude reaction mixture that is significantly cleaner. This inherent selectivity means that the final high-purity tetra-substituted furan products can often be isolated with minimal chromatographic effort, preserving the overall yield. For quality assurance teams, this predictable impurity profile simplifies validation and ensures consistent batch-to-batch reproducibility, which is critical for regulatory compliance in pharmaceutical supply chains.
How to Synthesize Tetra-Substituted Furan Efficiently
The practical implementation of this synthesis is designed for seamless integration into existing pilot and production facilities. The protocol begins with the preparation of an anhydrous environment, typically under a nitrogen blanket, to prevent the premature hydrolysis of the titanium reagent. The tetracarbonyl starting material is dissolved in a solvent such as toluene, followed by the controlled addition of TiCl4. The reaction progress is easily monitored via thin-layer chromatography (TLC), allowing operators to determine the precise endpoint without guesswork. Once complete, the reaction is quenched safely with aqueous ammonium chloride, separating the organic product from titanium salts. The detailed standardized synthesis steps, including specific molar ratios and temperature ramps optimized for different substrates, are outlined in the guide below to ensure successful replication.
- Under an inert nitrogen atmosphere, dissolve the tetracarbonyl compound in freshly distilled toluene or dichloromethane within a reactor equipped with a reflux condenser.
- Add titanium tetrachloride (TiCl4) to the solution at a molar ratio of 1: 1 to 1:5 relative to the substrate, ensuring strict moisture exclusion.
- Heat the mixture to 20-80°C and stir for 0.5 to 24 hours until TLC indicates complete consumption of the starting material.
- Quench the reaction with saturated aqueous ammonium chloride, separate the organic phase, extract the aqueous layer, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this TiCl4-mediated route offers transformative economic benefits that extend far beyond simple yield improvements. The primary advantage lies in the drastic simplification of the operational workflow, which directly correlates to reduced labor costs and increased facility utilization rates. By eliminating the need for specialized corrosion-resistant reactors required for strong mineral acids, capital expenditure for new production lines is significantly lowered. Furthermore, the use of commodity chemicals like toluene and TiCl4 ensures a stable and secure supply of raw materials, mitigating the risk of shortages that often plague exotic catalyst systems. This reliability is paramount for maintaining continuous production schedules and meeting the tight delivery windows demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of expensive transition metal catalysts and the reduction of waste disposal costs. Traditional methods often require precious metals or complex ligands that contribute heavily to the bill of materials, whereas TiCl4 is an inexpensive, bulk commodity chemical. Additionally, the simplified workup procedure reduces the consumption of solvents and silica gel during purification, leading to substantial savings in consumables. The high atom economy and reduced formation of tarry byproducts mean that less raw material is wasted, optimizing the overall material balance and driving down the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without compromising product quality. The use of standard solvents like dichloromethane and toluene ensures that sourcing is never a bottleneck, as these are widely available from multiple global vendors. Moreover, the short reaction times (often less than 2 hours for optimal substrates) allow for faster batch turnover, enabling manufacturers to respond more agilely to fluctuating market demands. This agility reduces the need for large safety stocks, freeing up working capital and improving the overall cash flow of the manufacturing operation.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage is straightforward due to the absence of hazardous high-pressure or high-temperature requirements. The mild conditions allow for the use of standard glass-lined or stainless steel reactors, facilitating easy technology transfer from R&D to production. From an environmental standpoint, the process generates less acidic waste compared to sulfuric acid methods, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The reduced environmental footprint not only lowers compliance costs but also enhances the sustainability profile of the supply chain, a key metric for modern ESG-conscious corporations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the TiCl4-mediated furan synthesis. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance. Understanding these nuances is critical for making informed decisions about process adoption and vendor selection. The following questions address substrate compatibility, safety considerations, and scalability factors that are top of mind for engineering and procurement stakeholders.
Q: What are the advantages of using TiCl4 over traditional Bronsted acids for furan synthesis?
A: Unlike corrosive Bronsted acids like sulfuric acid which require harsh conditions and generate significant waste, TiCl4 acts as a mild yet potent Lewis acid and dehydrating agent. This allows for lower reaction temperatures (20-80°C), reduces side reactions, and simplifies the workup procedure, leading to higher purity products suitable for pharmaceutical applications.
Q: Can this method accommodate sterically hindered substrates?
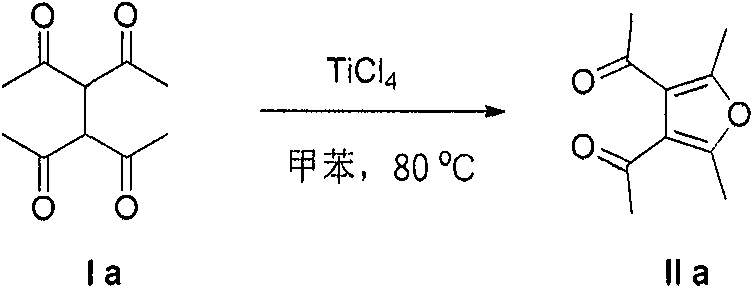
A: Yes, the patent data demonstrates excellent tolerance for bulky groups. For instance, tetracarbonyl compounds with phenyl substituents (Example 2) successfully cyclized to form tetra-phenyl substituted furans in 77% yield, indicating the method is robust against steric hindrance compared to many conventional protocols.
Q: Is the process scalable for industrial manufacturing?
A: Absolutely. The process utilizes common solvents like toluene and dichloromethane and operates at moderate temperatures without requiring specialized equipment like microwave reactors. The simple aqueous quench and extraction workup makes it highly amenable to large-scale production in standard chemical reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetra-Substituted Furan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a promising patent to a commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team of process chemists has extensively evaluated the TiCl4-catalyzed route described in CN107311963B and has optimized it for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of tetra-substituted furan meets the exacting standards required for pharmaceutical synthesis.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. Whether you require custom synthesis of novel furan derivatives or scale-up of existing intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and accelerate your time to market with our superior manufacturing solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
