Advanced Manufacturing of Hydroxynisone: A Scalable Copper-Catalyzed Route for Liver Fibrosis Therapeutics
Advanced Manufacturing of Hydroxynisone: A Scalable Copper-Catalyzed Route for Liver Fibrosis Therapeutics
The global pharmaceutical landscape is increasingly focused on developing effective treatments for chronic liver diseases, with liver fibrosis representing a critical unmet medical need. In this context, the compound Hydroxynisone has emerged as a promising candidate due to its favorable pharmacological profile in preventing and treating hepatic fibrosis and cirrhosis. A pivotal advancement in the manufacturing of this key pharmaceutical intermediate is detailed in patent CN116023327A, which outlines a novel preparation method characterized by simple operation, mild reaction conditions, and exceptional suitability for mass production. This technical insight report analyzes the transformative potential of this copper-catalyzed synthesis route, offering a strategic perspective for R&D directors, procurement managers, and supply chain leaders seeking to optimize their API intermediate sourcing strategies. By leveraging this innovative methodology, stakeholders can achieve significant improvements in process safety, yield efficiency, and overall cost structure without compromising on the stringent purity requirements demanded by modern regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Hydroxynisone and related analogues has been plagued by significant technical and economic inefficiencies that hinder large-scale commercial viability. Traditional pathways often rely on the use of 2-amino-5-methylpyridine as a starting material, necessitating complex protection and deprotection sequences that add unnecessary steps to the manufacturing workflow. Specifically, prior art methods frequently employ boron tribromide (BBr3) for the critical demethylation step, a reagent known for its extreme toxicity, high cost, and the generation of hazardous gaseous byproducts that require specialized scrubbing systems. Furthermore, these legacy processes typically suffer from low overall yields, often hovering around 40%, which drastically increases the cost of goods sold and creates substantial waste disposal challenges. The operational complexity associated with handling such dangerous reagents also introduces significant safety risks for plant personnel and complicates regulatory compliance, making these conventional routes increasingly obsolete in a modern, safety-conscious manufacturing environment.
The Novel Approach
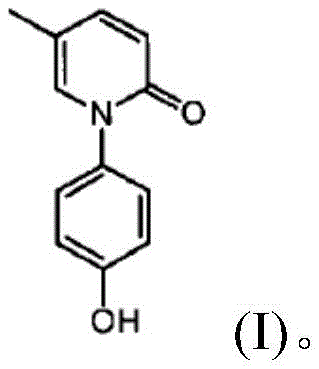
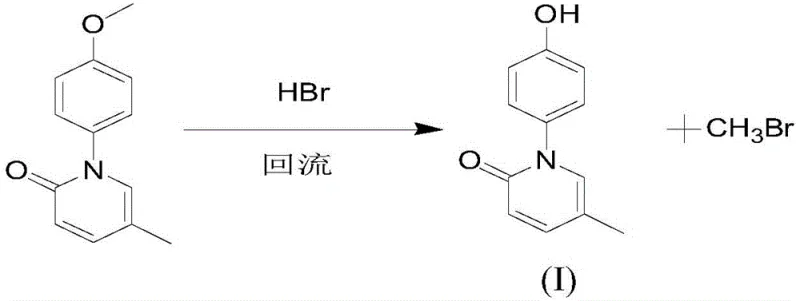
In stark contrast to these legacy methods, the novel approach disclosed in the patent data introduces a streamlined, copper-catalyzed carbon-nitrogen coupling strategy that fundamentally reshapes the production economics. This innovative route utilizes 2-hydroxy-5-methylpyridine and p-bromoanisole as readily available starting materials, bypassing the need for cumbersome protection groups and enabling a more direct assembly of the molecular scaffold. A key breakthrough lies in the substitution of hazardous boron tribromide with hydrobromic acid (HBr) for the demethylation step, which not only enhances operational safety but also simplifies the downstream purification process. The implementation of this method results in a dramatic improvement in yield efficiency, with the final demethylation step achieving yields as high as 98%, thereby maximizing raw material utilization. By eliminating toxic gas formation and reducing the number of unit operations, this new protocol offers a robust, safer, and significantly more cost-effective pathway for the industrial production of high-purity Hydroxynisone.
Mechanistic Insights into Cu-Catalyzed C-N Coupling
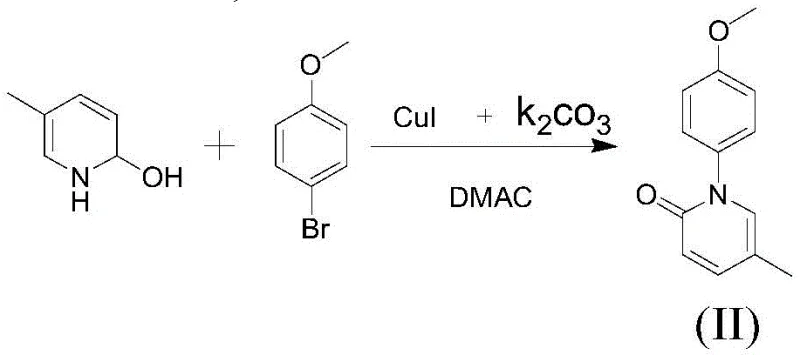
The core of this technological advancement rests on a highly efficient copper-catalyzed Ullmann-type coupling reaction, which facilitates the formation of the critical C-N bond between the pyridine and phenyl rings. In this mechanism, catalysts such as Cuprous Iodide (CuI) or Copper Sulfate Pentahydrate (CuSO4·5H2O) work in synergy with a base like Potassium Carbonate (K2CO3) to activate the aryl halide and promote nucleophilic attack by the hydroxypyridine species. The reaction is typically conducted in polar aprotic solvents such as DMAC or DMF at elevated temperatures ranging from 130°C to 150°C, conditions that ensure complete conversion while maintaining selectivity. The choice of catalyst system is crucial; for instance, the molar ratio of CuI to K2CO3 is optimized between 1:50 and 1:80 to balance catalytic activity with cost efficiency. This precise control over reaction parameters minimizes the formation of side products and ensures a clean reaction profile, which is essential for reducing the burden on downstream purification units and achieving the high purity standards required for pharmaceutical intermediates.
Beyond the coupling reaction itself, the patent highlights a sophisticated purification strategy that is critical for controlling the impurity profile of the final product. The intermediate compound (Formula II) is refined through a specialized process involving dissolution in ethyl acetate followed by treatment with activated carbon, a method that proves superior to simple recrystallization. This decolorization and purification step effectively removes trace metal catalysts, organic byproducts, and colored impurities that could otherwise persist through to the final API. The data indicates that this specific purification protocol yields an intermediate with purity exceeding 99.6%, setting a strong foundation for the subsequent demethylation step. By rigorously controlling the quality of the intermediate, the overall process ensures that the final Hydroxynisone product meets stringent specifications, with reported purity levels reaching up to 99.98%. This focus on intermediate quality control is a hallmark of a mature, GMP-ready manufacturing process designed to minimize batch-to-batch variability.
How to Synthesize Hydroxynisone Efficiently
The synthesis of Hydroxynisone via this patented route involves a logical sequence of coupling, purification, and demethylation steps that are optimized for industrial scalability. The process begins with the copper-catalyzed reaction to form the methoxy-intermediate, followed by a critical purification stage using ethyl acetate and activated carbon to ensure high purity. The final step involves acid-mediated demethylation to reveal the active phenolic group. For R&D teams looking to implement this technology, understanding the precise stoichiometry and thermal profiles is essential for successful technology transfer. The detailed standardized synthesis steps, including specific reagent ratios, temperature gradients, and workup procedures, are outlined in the technical guide below to facilitate immediate process adoption.
- Perform copper-catalyzed C-N coupling between 2-hydroxy-5-methylpyridine and p-bromoanisole using CuI or CuSO4 in DMAC/DMF at 130-150°C.
- Purify the intermediate compound II by dissolving in ethyl acetate and treating with activated carbon to remove impurities and color.
- Execute demethylation using hydrobromic acid (HBr) under reflux conditions to yield high-purity Hydroxynisone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound advantages for procurement and supply chain management, primarily driven by the simplification of the chemical process and the elimination of hazardous reagents. The shift away from expensive and toxic boron tribromide to hydrobromic acid represents a significant reduction in raw material costs and waste treatment expenses, directly impacting the bottom line. Furthermore, the use of common industrial solvents like DMF and ethyl acetate ensures that supply chain risks associated with specialty chemical sourcing are minimized, enhancing the reliability of raw material availability. The high yield and purity achieved through this method also mean that less raw material is required to produce the same amount of final product, leading to substantial cost savings in manufacturing. These factors combine to create a more resilient and cost-efficient supply chain capable of meeting the growing demand for liver fibrosis therapeutics without the bottlenecks associated with older, less efficient technologies.
- Cost Reduction in Manufacturing: The elimination of expensive protecting group reagents and the substitution of hazardous demethylation agents with hydrobromic acid drastically reduce the cost of goods sold. By avoiding the need for specialized equipment to handle toxic gases and reducing waste disposal volumes, the overall operational expenditure is significantly lowered. The high yield of the coupling and demethylation steps ensures maximum raw material efficiency, minimizing waste and further driving down production costs per kilogram of active ingredient.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as p-bromoanisole and 2-hydroxy-5-methylpyridine ensures a stable and secure supply chain, reducing the risk of production delays caused by raw material shortages. The simplified process flow, with fewer unit operations and milder reaction conditions, allows for faster batch turnover and increased production capacity. This operational agility enables suppliers to respond more quickly to market demand fluctuations, ensuring consistent delivery schedules for downstream pharmaceutical manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of highly toxic byproducts make this process inherently safer and easier to scale from pilot plant to commercial production volumes. The reduced environmental footprint, achieved through lower waste generation and the use of less hazardous reagents, simplifies regulatory compliance and permits easier approval for manufacturing in diverse global jurisdictions. This scalability ensures that the supply of Hydroxynisone can be expanded seamlessly to meet clinical and commercial needs without requiring massive capital investment in new safety infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Hydroxynisone, based on the specific advantages and data points presented in the patent literature. These answers are designed to provide clarity on process safety, yield expectations, and scalability for stakeholders evaluating this technology for their supply chains. Understanding these details is crucial for making informed decisions about sourcing strategies and technology partnerships in the competitive landscape of pharmaceutical intermediates.
Q: How does the new hydrobromic acid demethylation method compare to traditional boron tribromide methods?
A: The new method replaces expensive and highly toxic boron tribromide with hydrobromic acid, significantly improving operational safety and reducing waste treatment complexity while maintaining high yield.
Q: What is the achieved purity level for Hydroxynisone using this copper-catalyzed route?
A: Through optimized recrystallization and activated carbon decolorization steps, the process consistently achieves final product purity exceeding 99.9%, meeting stringent pharmaceutical standards.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions, readily available solvents like DMF and ethyl acetate, and avoids hazardous gas generation, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxynisone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthesis routes in the development of life-saving medications like Hydroxynisone. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to full-scale manufacturing. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We are uniquely positioned to leverage the efficiencies of the copper-catalyzed route described in CN116023327A, offering our clients a reliable source of high-purity intermediates that meet the exacting demands of global regulatory agencies.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the tangible benefits of partnering with a supplier who prioritizes both technical excellence and commercial value. Together, we can accelerate the delivery of effective liver fibrosis treatments to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
