Advanced Hydroxynisone Synthesis: Scalable Technology for Global Pharmaceutical Supply Chains
Advanced Hydroxynisone Synthesis: Scalable Technology for Global Pharmaceutical Supply Chains
The global pharmaceutical landscape is increasingly demanding efficient, safe, and scalable synthesis routes for critical therapeutic intermediates, particularly in the realm of liver disease treatment. Patent CN116082225A introduces a groundbreaking preparation method for hydroxynisone, a compound showing significant promise in treating hepatic fibrosis and cirrhosis. This technology addresses the urgent market need for safe and effective medicines, leveraging a novel copper-catalyzed carbon-nitrogen coupling reaction that bypasses the limitations of prior art. The market demand for liver-protecting medicaments has increased year by year, with the liver fibrosis drug market estimated at substantial values in major regions like China and Japan. By adopting this innovative approach, manufacturers can achieve high yields and operational safety, positioning themselves as a reliable pharmaceutical intermediates supplier in a competitive global market. The core structure of the target molecule, hydroxynisone, is depicted below, highlighting the specific chemical architecture that this new process efficiently constructs.
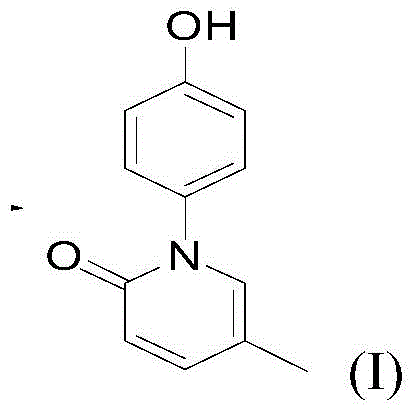
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxynisone and similar compounds has been plagued by significant technical and economic inefficiencies that hinder commercial viability. Prior art methods typically rely on the use of p-bromophenol with hydroxyl protection groups, necessitating complicated reaction procedures that add unnecessary steps to the industrial process. A major bottleneck in these traditional routes is the deprotection step, which often employs expensive reagents and generates highly toxic gases, creating severe environmental and safety hazards for production facilities. Furthermore, the sewage and waste treatment associated with these legacy methods are complex and costly, eroding profit margins and complicating regulatory compliance. Perhaps most critically, these conventional pathways suffer from low yields, often reported around 40%, which drastically increases the cost of goods sold and limits the ability to meet large-scale demand. The operational complexity and safety risks associated with these older methods make them unsuitable for modern, high-volume manufacturing environments.
The Novel Approach
In stark contrast, the method disclosed in patent CN116082225A offers a streamlined, robust, and economically superior alternative that resolves the痛点 of previous technologies. This novel approach eliminates the need for hydroxyl protection on p-bromophenol, significantly simplifying the operation and reducing the total number of synthetic steps. By utilizing ammonia water to treat the crude intermediate, the process effectively removes residual copper ions from the catalyst while simultaneously facilitating product separation, a dual benefit that enhances both purity and yield. The substitution of toxic boron tribromide with hydrobromic acid for demethylation represents a major safety advancement, reducing the risk of toxic gas formation and simplifying waste management. This method is characterized by mild reaction conditions, short reaction times, and high yields, making it ideally suitable for mass production. The result is a process that not only improves technical performance but also delivers substantial cost savings in API manufacturing through reduced material consumption and waste disposal costs.
Mechanistic Insights into Copper-Catalyzed C-N Coupling
The core of this technological breakthrough lies in the efficient copper-catalyzed carbon-nitrogen coupling reaction, which forms the critical bond between the pyridine and phenyl rings. The reaction involves 2-hydroxy-5-methylpyridine and p-bromoanisole reacting in a polar aprotic solvent such as DMAC or DMF in the presence of a copper catalyst system. The preferred catalyst system comprises CuI or CuSO4·5H2O combined with K2CO3 as a base, operating at reflux temperatures between 130°C and 150°C. This specific catalytic environment promotes the nucleophilic substitution necessary to form the intermediate compound of formula II with high selectivity. The molar ratio of reactants is carefully optimized, typically ranging from 1:1 to 1:1.5, to ensure maximum conversion while minimizing excess reagent waste. The choice of solvent and catalyst combination is critical, as it directly influences the reaction kinetics and the formation of by-products, ensuring that the process remains robust even when scaled to industrial volumes.
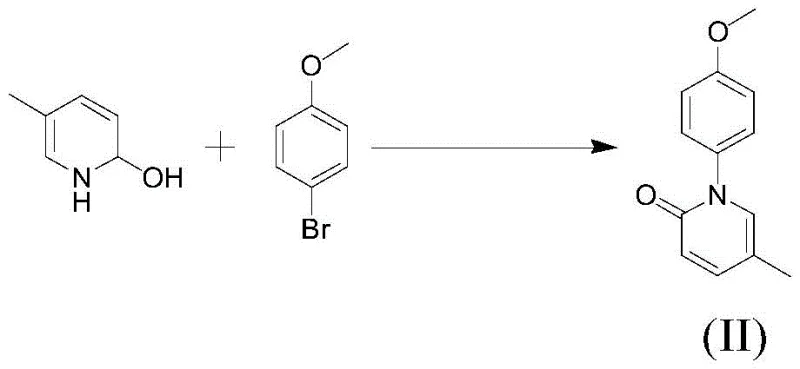
A pivotal innovation in this mechanism is the post-reaction purification strategy involving ammonia water washing. In traditional copper-catalyzed reactions, removing residual metal catalysts to meet pharmaceutical purity standards often requires expensive scavengers or complex chromatography. However, this patent reveals that washing the crude product with 15-28wt% ammonia water effectively complexes and removes copper ions. Comparative data demonstrates that water washing alone leaves detectable copper levels, whereas ammonia treatment renders the copper undetectable by UV-visible spectrophotometry. This step not only ensures the high purity of the intermediate (up to 99.66%) but also improves the isolation yield by promoting product precipitation. This mechanistic insight into impurity control is crucial for R&D directors focused on impurity profiles, as it guarantees a cleaner feed for the subsequent demethylation step, ultimately leading to a final product purity of 99.92% or higher.
How to Synthesize Hydroxynisone Efficiently
The synthesis of hydroxynisone via this patented route involves a logical sequence of coupling, purification, and demethylation steps designed for industrial robustness. The process begins with the copper-catalyzed coupling to form the methoxy-protected intermediate, followed by the critical ammonia wash to ensure metal compliance. The final step involves acid-mediated demethylation to reveal the active phenolic group. This streamlined workflow eliminates unnecessary protection groups and hazardous reagents, offering a direct path from raw materials to the high-purity final API intermediate. For technical teams looking to implement this route, the detailed standardized synthesis steps are provided in the guide below, ensuring reproducibility and safety at scale.
- Perform copper-catalyzed carbon-nitrogen coupling between 2-hydroxy-5-methylpyridine and p-bromoanisole in DMAC or DMF solvent.
- Execute a critical ammonia water wash step to remove residual copper ions and isolate the refined intermediate compound II.
- Conduct demethylation using hydrobromic acid (HBr) followed by recrystallization to yield the final high-purity hydroxynisone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible strategic advantages beyond mere technical feasibility. The elimination of expensive and hazardous reagents like BBr3 directly contributes to cost reduction in pharmaceutical intermediates manufacturing by lowering raw material costs and reducing the need for specialized safety infrastructure. The simplified operational procedure, which omits complex protection steps, reduces labor hours and equipment occupancy time, thereby increasing overall plant throughput. Furthermore, the use of readily available starting materials such as 2-hydroxy-5-methylpyridine and p-bromoanisole ensures supply chain stability, reducing the risk of production delays caused by scarce reagents. The high yield and purity achieved minimize waste generation, aligning with environmental compliance goals and reducing disposal fees. These factors collectively enhance the reliability of the supply chain, ensuring consistent delivery of high-purity pharmaceutical intermediates to downstream partners.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing expensive deprotection reagents with cost-effective hydrobromic acid and eliminating the need for hydroxyl protection groups entirely. This reduction in chemical consumption, combined with the high yield of the coupling and demethylation steps, drastically lowers the cost per kilogram of the final product. Additionally, the simplified purification process reduces solvent usage and energy consumption during distillation and crystallization, further driving down operational expenditures without compromising quality standards.
- Enhanced Supply Chain Reliability: By utilizing common industrial solvents like DMAC and DMF and widely available catalysts like copper iodide or copper sulfate, the process mitigates the risk of raw material shortages. The robustness of the reaction conditions allows for flexible scheduling and reduces the likelihood of batch failures, ensuring reducing lead time for high-purity pharmaceutical intermediates. The ability to source materials from multiple vendors and the stability of the reaction pathway provide a secure foundation for long-term supply agreements with global pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of highly toxic gases make this process inherently safer and easier to scale from pilot plant to commercial production. The effective removal of copper ions via ammonia washing simplifies wastewater treatment, reducing the environmental footprint and ensuring compliance with strict regulatory standards. This scalability supports the commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to respond quickly to market demand surges for liver fibrosis treatments while maintaining a sustainable production profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydroxynisone synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in patent CN116082225A, providing clarity on purity, safety, and scalability. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The insights provided here reflect the consensus of technical analysis regarding the process advantages over conventional methods.
Q: How does the new ammonia wash step improve product purity compared to traditional water washing?
A: The patent data indicates that using ammonia water (15-28wt%) instead of pure water significantly enhances the removal of residual copper catalyst ions. Comparative examples show that water washing leaves detectable copper levels (0.05 μg/50 ml), whereas the ammonia treatment ensures the product meets stringent heavy metal specifications required for pharmaceutical intermediates.
Q: What are the safety advantages of using HBr over BBr3 for the demethylation step?
A: Traditional methods often utilize boron tribromide (BBr3), which is highly toxic and generates complex hazardous waste. This novel process substitutes HBr, which is safer to handle, reduces toxic gas formation risks, and simplifies the sewage treatment process, thereby lowering overall environmental compliance costs.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for mass production. It operates under mild reaction conditions (130-150°C), avoids complex protection-deprotection sequences, and achieves high yields (up to 90% in final steps), making it economically viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxynisone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes like the one described in patent CN116082225A for the future of liver disease therapeutics. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of hydroxynisone meets the exacting standards required by global regulatory bodies. We are committed to supporting our partners in bringing safe and effective medicines to market through superior manufacturing capabilities.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your production efficiency and product quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
