Electrocatalytic Sulfoxide Synthesis Revolutionizing Pharmaceutical Intermediate Manufacturing Scalability
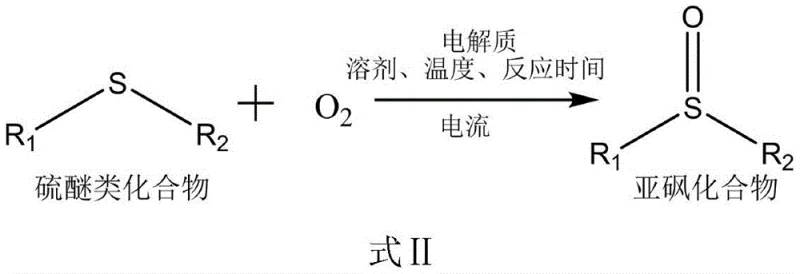
Patent CN111534832A introduces a transformative electrocatalytic methodology for synthesizing sulfoxide compounds—a critical class of intermediates in pharmaceutical manufacturing—by fundamentally reimagining oxidation chemistry through electron-driven processes rather than conventional catalytic systems. This innovation directly addresses longstanding industry challenges associated with transition metal catalysts including environmental contamination risks, complex purification requirements, and supply chain vulnerabilities that have historically constrained production scalability. Operating under exceptionally mild conditions at ambient temperature (25°C) with low electrical current (4 mA), the method leverages atmospheric oxygen as an oxidant source while eliminating toxic solvents through strategic use of 1,2-dichloroethane as reaction medium. The patent demonstrates remarkable efficiency across diverse substrates with yields consistently exceeding 90%, positioning this technology as a sustainable solution for producing high-purity intermediates essential to blockbuster drugs such as darunavir for antiviral therapy and omeprazole for gastrointestinal treatments. By replacing hazardous chemical oxidants with precisely controlled electrical parameters, this approach delivers both environmental compliance advantages and operational simplicity that align with evolving global regulatory frameworks for green pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional sulfoxide synthesis methodologies suffer from critical limitations that compromise both environmental sustainability and commercial viability within pharmaceutical supply chains. These processes predominantly rely on transition metal catalysts such as platinum, copper, or gold which generate significant hazardous waste streams requiring costly remediation procedures to meet regulatory standards for heavy metal residues in drug intermediates. The harsh oxidation conditions typically employed—including elevated temperatures above 80°C or strong chemical oxidants—frequently lead to poor reaction selectivity with substantial overoxidation byproducts that complicate purification workflows and reduce overall yields below acceptable commercial thresholds. Catalyst preparation itself involves multi-step syntheses that introduce additional cost layers and timeline dependencies while creating supply chain fragility due to geopolitical constraints on rare metal resources. Furthermore, residual metal contamination necessitates extensive post-reaction purification steps that increase both processing time and operational costs while introducing quality control variability that threatens batch consistency required for pharmaceutical applications where purity specifications must meet parts-per-billion standards.
The Novel Approach
The electrocatalytic methodology disclosed in CN111534832A overcomes these limitations through an elegant electron-driven oxidation mechanism that operates within an undivided cell configuration using graphite anodes and platinum cathodes at precisely controlled low current (4 mA). This approach eliminates transition metal catalysts entirely while maintaining exceptional reaction efficiency through tunable electrical parameters that prevent overoxidation side reactions common in conventional methods. By utilizing atmospheric oxygen as the oxidant source instead of hazardous chemical alternatives, the process achieves significant environmental benefits including near-zero waste generation and elimination of toxic byproducts that plague traditional oxidation pathways. The strategic selection of 1,2-dichloroethane as solvent replaces hazardous alternatives while optimizing ion mobility for consistent performance across diverse substrate types—from simple alkyl thioethers to complex aryl derivatives—demonstrating remarkable versatility without reoptimization requirements. Crucially, the absence of metal catalysts removes downstream purification burdens while ensuring inherent product purity that meets stringent pharmaceutical quality standards without additional processing steps.
Mechanistic Insights into Electrocatalytic Sulfoxide Formation
The core innovation resides in the electron-transfer mechanism where thioether substrates undergo single-electron oxidation at the anode surface to form radical cations that subsequently react with molecular oxygen sourced from ambient air through proton-coupled electron transfer sequences. This cascade occurs within an undivided cell configuration using graphite anodes and platinum cathodes operating at low current density (4 mA), where precise voltage control prevents overoxidation to sulfones while maintaining optimal reaction kinetics at ambient temperature (25°C). The electrolyte—typically tetrabutylammonium tetrafluoroborate at a molar ratio of 1:0.3 relative to substrate—stabilizes reactive intermediates through ion-pairing effects that enhance selectivity across diverse functional groups including electron-rich aryl systems and aliphatic derivatives. Solvent polarity plays a critical role in determining reaction efficiency; comparative studies show that dichloroethane provides ideal dielectric properties for charge separation while minimizing radical decomposition pathways that reduce yields in alternative solvents like DMSO or acetonitrile.
Impurity control is inherently achieved through electrical parameter modulation where current density adjustments maintain consistent oxidation rates across batch scales while preventing side reactions that generate sulfone byproducts or unreacted starting materials. The absence of metal catalysts eliminates trace contamination risks that would otherwise require specialized analytical methods like ICP-MS for detection at parts-per-billion levels—a critical advantage for pharmaceutical applications where regulatory agencies mandate strict elemental impurity controls. Solvent optimization studies demonstrate how dichloroethane's moderate polarity prevents over-solvation of reactive intermediates compared to protic solvents like methanol which lead to hydrolysis side products reducing yields below acceptable thresholds. This mechanistic understanding directly translates to commercial benefits including simplified quality control protocols that reduce analytical costs while ensuring consistent compliance with pharmacopeial standards across all production scales from laboratory development through commercial manufacturing.
How to Synthesize Sulfoxide Compounds Efficiently
This electrocatalytic synthesis represents a paradigm shift in sulfoxide manufacturing by offering unprecedented operational simplicity combined with environmental sustainability through elimination of hazardous catalysts while maintaining high yields via precisely controlled electrical parameters ideal for both laboratory-scale development and commercial production environments. The methodology provides reproducible results across diverse substrate types including complex aryl derivatives essential for pharmaceutical applications requiring stringent purity specifications. Below is a standardized procedure derived from patent CN111534832A that ensures consistent implementation across industrial settings with minimal process validation requirements due to its inherent robustness.
- Prepare the reaction mixture by adding thioether compound and electrolyte in a molar ratio of 1: 0.3 to the reactor.
- Introduce solvent and apply electrocatalysis under constant current of 4 mA at 25°C for 7 hours.
- Concentrate crude product using rotary evaporator followed by silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative process directly addresses three critical pain points in pharmaceutical intermediate procurement: cost volatility from catalyst dependencies, supply chain fragility due to complex purification needs, and scalability limitations inherent in traditional oxidation methodologies. By replacing expensive transition metals with electricity—a universally accessible resource—the technology fundamentally restructures cost drivers while enhancing production resilience through simplified workflows that reduce both capital expenditure requirements and operational complexity across manufacturing sites globally. The elimination of metal catalysts removes associated supply chain risks from geopolitical instabilities affecting rare metal markets while enabling faster technology transfer between R&D facilities and production plants without extensive revalidation procedures.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts eliminates multiple cost layers including specialized procurement channels, hazardous material handling procedures, and extensive post-reaction purification systems required for metal residue removal—streamlining production into a single-step electrochemical process that reduces both capital expenditure on recovery infrastructure and operational costs from waste treatment protocols while maintaining high product quality standards without additional investments.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through utilization of air as the oxidant source combined with globally available electrolytes like tetrabutylammonium salts from multiple qualified suppliers—eliminating single-point failures associated with specialized catalyst supply chains while enabling seamless scale-up from kilogram quantities to multi-ton annual volumes without revalidation requirements due to consistent process performance across different substrate types.
- Scalability and Environmental Compliance: The technology demonstrates inherent scalability from laboratory (0.5 mmol) to commercial production scales through simple electrode surface area expansion without reoptimization needs—while minimizing environmental impact through near-stoichiometric oxygen utilization from air and potential solvent recycling capabilities that significantly reduce waste generation compared to conventional methods requiring strong chemical oxidants thus aligning with global regulatory trends toward sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN111534832A's implementation data and performance metrics reflecting real-world application scenarios encountered during process validation studies across multiple pharmaceutical intermediates manufacturing environments.
Q: How does this electrocatalytic method eliminate transition metal catalyst dependencies?
A: The process utilizes electrons as reagents through controlled electrical parameters instead of platinum or copper catalysts, removing both environmental hazards from metal waste streams and supply chain vulnerabilities associated with scarce metal resources.
Q: What ensures high purity without complex purification steps?
A: The inherent selectivity of electrochemical oxidation prevents overoxidation byproducts while eliminating metal contaminants, enabling straightforward purification via rotary evaporation and standard silica gel chromatography.
Q: How does solvent selection impact commercial viability?
A: Optimal solvent choice (e.g., DCE) maintains high yields above 97% while replacing toxic alternatives, reducing both waste treatment costs and regulatory compliance burdens across global manufacturing sites.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfoxide Compound Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities specifically designed for complex intermediate synthesis validation. This electrocatalytic technology exemplifies our commitment to developing sustainable manufacturing solutions that address both technical challenges and commercial imperatives in pharmaceutical intermediate production through deep process chemistry expertise combined with flexible manufacturing infrastructure capable of adapting to evolving client requirements across global markets.
Leverage our Customized Cost-Saving Analysis to evaluate how this technology can optimize your specific supply chain—contact our technical procurement team today to request detailed COA data and route feasibility assessments tailored to your manufacturing requirements.
